 |
 |
 |
| |
Resistance to New HCV Antivirals
|
| |
| |
Reported by Jules Levin
42nd Annual Meeting of the European Association for the Study of the Liver
(EASL)
April 11-15, 2007, Barcelona. Spain
As in HIV, drug resistance to the new HCV antivirals in development is an important consideration. HCV has high levels of virus replication and there are a number of pre-existing resistance mutations which can emerge after therapy is started. So far the VX950 studies have shown that the addition of Peginterferon plus ribavirin is able to suppress HCV to undetectable. One question is what about patients who don't respond well to interferon? Several researchers have expressed concern privately about patients who don't respond to peginterferon. There is some research suggesting that HCV protease inhibitors may reverse this inability to respond to interferon but we will need to see this in clinical studies in patients. But Vertex has reported that monotherapy with VX950 can result in the quick development of drug mutations. Additional oral agents, polymerase inhibitors, may be needed in a regimen for some patients to achieve undetectable HCV RNA. Another question is what is the degree of cross-resistance between HCV protease inhibitors in development and will cross-resistance impair serial use of the HCV protease inhibitors now in development. As well, resistance related to HCV polymerase inhibitors also is an important question.
Tara Kieffer from Vertex discussed HCV protease inhibitor resistance at today's opening day symposium at EASL: "New Insights Into Antiviral Drug resistance in Chronic Viral Hepatitis Treatment".
Kieffer started off by saying "mutations in the HCV RNA genome occur spontaneously and frequently as a natural result of viral replication: the HCV polymerase has a high error rate, there are high levels of HCV replication (much more than in HIV). Therefore, drug resistant variants most likely exist at a low frequency prior to drug treatment: resistant variants tend to be less fit than wild-type. Treatment with a specifically-targeted antiviral drug selects for drug resistant virus, eg, HCV, HBV, HIV. Resistant variants confer different levels of resistance: resistant variants can exist with single, double, or more mutations on the same viral genome. These can differ significantly in their level of resistance and fitness. Drug resistance can result in clinical viral rebound. Replication of resistant variants can be inhibited by additional HCV inhibitors". Although it appears as there is no known mechanism in HCV for resistance to be archived as in HIV, at the EASL pre-conference resistance symposium Dr Jean-Michel Pawlotsky said HCV resistance mutations can build up when HCV is replicating and may emerge again after restarting therapy with drugs associated with the same mutations. I do not think we know yet how resistance mutations that emerge on therapy will act in HCV. Will they fade & re-emerge upon challenge in patients or will they just fade? In the third picture below triple therapy with VX950/peg/RBv suppressed resistant HCV in the study conducted by Vertex.
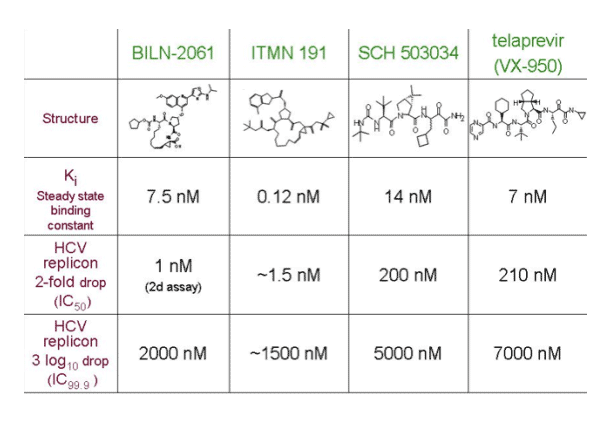
Kieffer presented this table displaying resistance mutations associated with various HCV protease inhibitors in development. Apparently several drug mutatyions are common to the HCV PIs in development but will that impair serial use? It is possible that a potent PI and one able to achieve high drug levels can overcome cross-resistance but it is too soon to know if that can be achieved. The developers of ITMN-191 think that may be possible with their drug but we don't know yet. But this is an important issue.
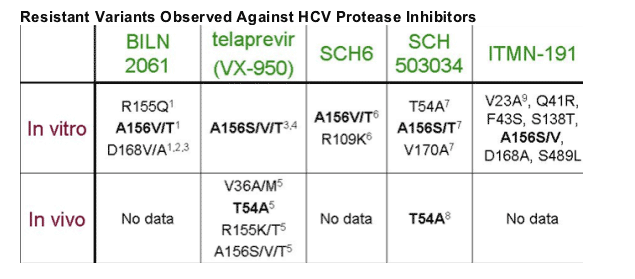
Therefore, prudence and caution is suggested in deciding when to begin therapy and in when & how one uses the new oral drugs as they become available. VX950 may become available in 2009 to be used in combination with peginterferon and ribavirin but availability of additional oral agents also in development now, polymerase inhibitors, are likely to be at a later time. If using VX950 plus Peg/RBV it may be important to consider how much response a patient will receive from the peg/RBV component in the regimen in suppressing HCV. It is still early in development for VX950 so lets see what the phase II and phase III studies tell us. I think Vertex is planning a study in interferon non-responders and such a study could tell us a lot about the use of VX9500 in individuals who failed peginterferon.
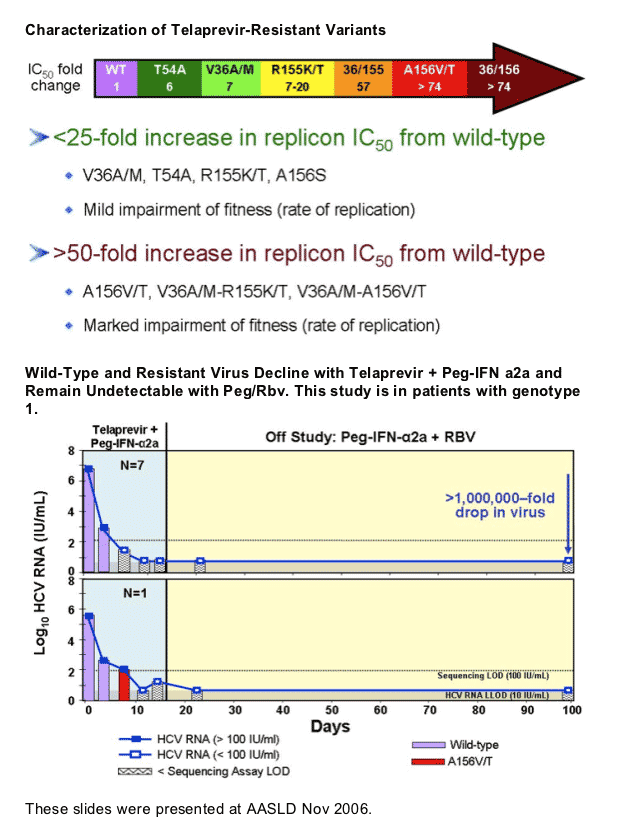
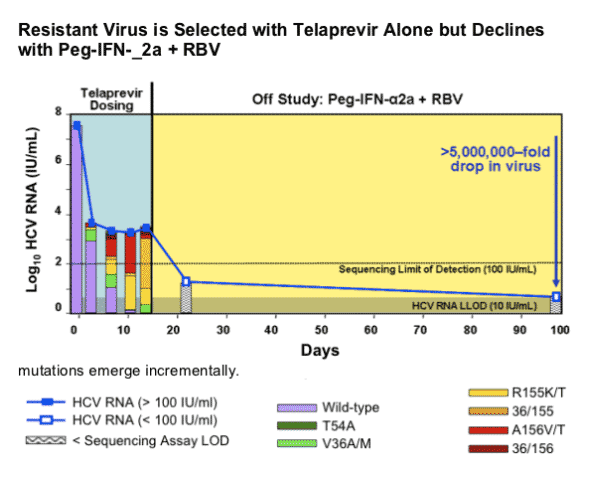
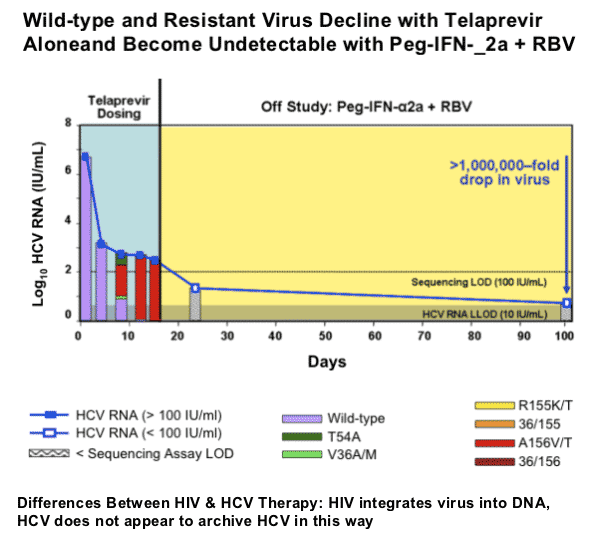
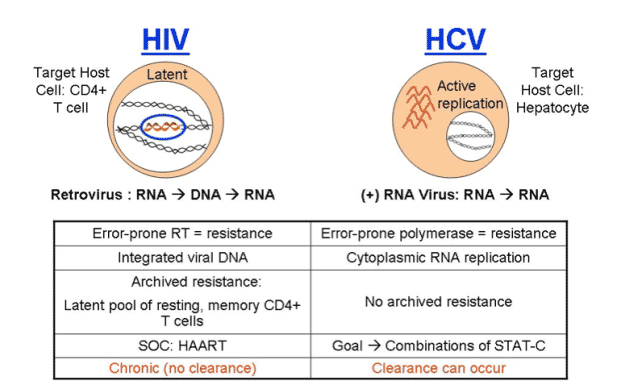
Unlike in HIV, HCV is "curable". This means that for patients who achieve undetectable viral load and sustain this (SVR) for 2 years after stopping HCV therapy HCV does not return and recent studies have shown for many patients liver damage can reverse or at least not progress any further, and HCV RNA is essentially gone. There have been several studies finding some residual HCV in reservoirs in patients with SVR but there is no evidence that viral rebound has occurred as a result of this; and there have been questions around whether the studies finding residual HCV were conducted properly. If a patient, however, has cirrhosis upon starting therapy if they achieve SVR they will have to monitor the liver with MRIs or Ct-Scans forever to assure that liver cancer does not develop. The risk of this occurring decreases over time if it has not occurred and is very low.
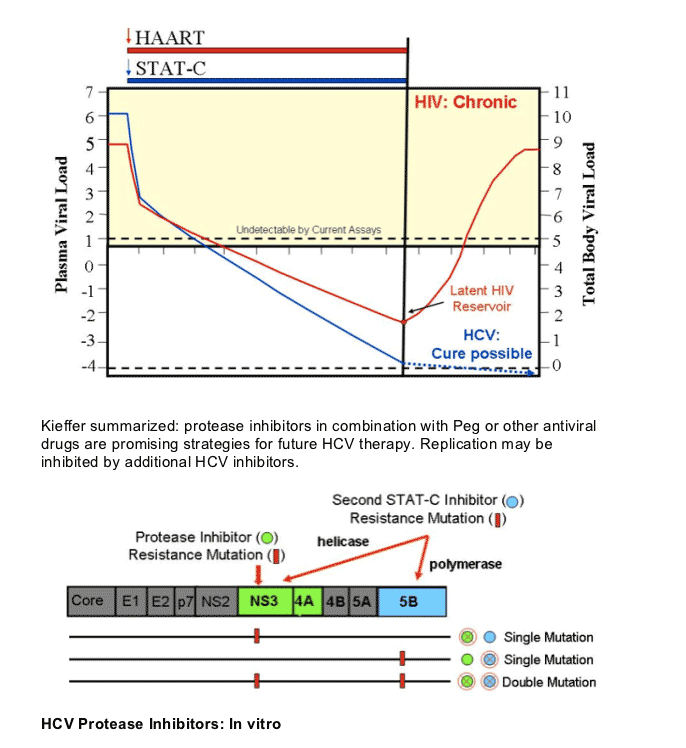
Kieffer summarized: protease inhibitors in combination with Peg or other antiviral drugs are promising strategies for future HCV therapy. Replication may be inhibited by additional HCV inhibitors.
|
| |
|
 |
 |
|
|