 |
 |
 |
| |
Four Year Assessment Of Entecavir Resistance In Nucleoside Naive and Lamivudine Refractory Patients
|
| |
| |
Reported by Jules Levin
EASL, April 11-15, 2007
Barcelona
Richard Colonno reported the study results today in the Late Breaker session at EASL.
R.J. Colonno, R.E. Rose, K. Pokornowski, C.J. Baldick, B. Eggers, D. Xu, A. Cross and D.J. Tenney
Bristol-Myers Squibb Research and Development
Author Summary
ETV resistance is very rare in nucleoside naive patients and distinct from other HBV antivirals1 (LVD = 71%, ADV = 18%, TBV = 9-22% [Yr 2])
Enrichment/Selection of any pre-existing LVDr variants present at the time of study entry decreases over time
Cumulative probabilities of genotypic ETVr alone and virologic breakthrough due to genotypic ETVr through 4 years
- Naive Patients - ETVr = 1.2%, ETVr w/ breakthrough = 0.8%
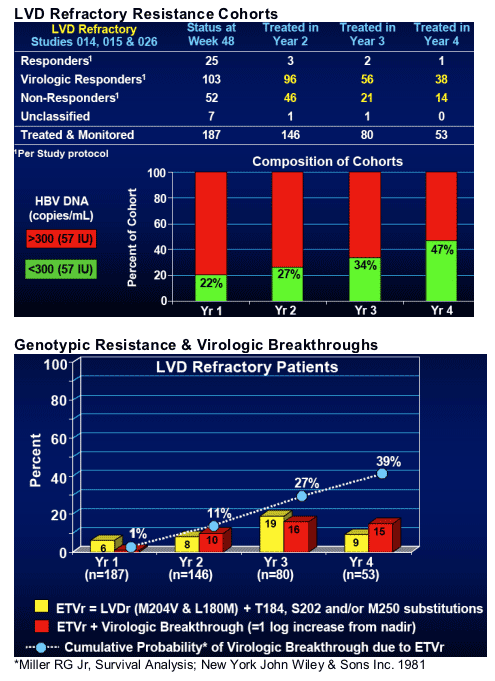
- LVD Refractory - ETVr = 43%, ETVr w/ breakthrough = 39%
ETVr requires M204V & L180M plus additional substitution(s) at residues S202, T184 or M250, all can be selected by prior LVD treatment
Not all ETVr variants lead to virologic breakthrough
*********************
Colonno started off by describing the "Primary Factors in Preventing Resistance".
"Potency" - Degree to which viral replication is suppressed to avoid generation of new mutations
"Genetic Barrier" - Number of substitutions required, concentrations achieved and impact on viral fitness
In nucleoside naive WT HBV in HBeAg+ HBV DNA reduction with entecavir is 6.9 logs (0.5mg dose). % PCR undetectable at week 48 in HBeAg+ is 67%. 3 genotypic substitutions in nucleoside naive patients are required for resistance (M204V & L180M + T184, S202 or M250.

Monitoring of Resistance
Clinical Studies: Naive (022, 027 and 901), LVD Refractory (014, 015, 026 and 901).
Genotyped paired samples from ALL ETV-treated patients with HBV DNA levels >300 copies/mL at Wks 48, 96, 144, 192 or End of Dosing (n=749 paired samples
analyzed)
--Phenotype determined for all novel emerging substitutions.
Phenotyped ALL patients experiencing a virologic breakthrough (=1 log increase from nadir), including those with no observed genotypic changes.
Genotype/Phenotype of HBV from Patients with Virologic Breakthroughs
--LVD Refractory Patients
37 virologic breakthroughs due to ETVr substitutions over 4 years
-- All had LVDr substitutions M204V + L180M
-- Frequency of additional ETVr substitutions
+ Single substitution (n =30): T184 (n=15) or S202 (n=15)
+ Double substitutions (n=6): T184 + S202 (n=4) or T184/S202 + M250 (n=2)
+ Triple substitutions (n=1): T184 + S202 + M250
-- ETVr detected (=0.1%) at baseline in 11 (30%) patients
-- Median ETV susceptibility decreased 19-fold from baseline and 283-fold from WT virus
-- ETVr isolates remain susceptible to adefovir
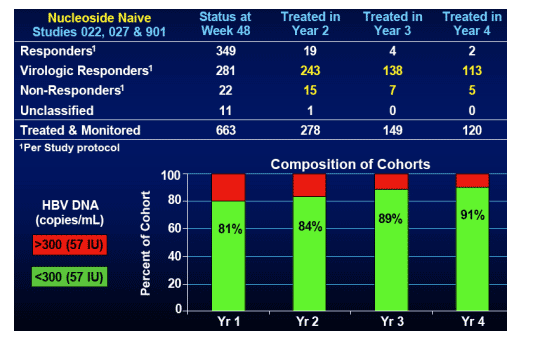
Nucleoside Naive Resistance Cohorts
In these 3 naive studies (022, 027, 901) used for the nuke naive resistance cohort analysis the total patients treated & monitored were 663, status at week 48: 349 responders, 281 virologic responders, 22 non-responders, 11 unclassified;
--treated in year 2: treated & monitored 278; responders 19; virologic responders: 243; non-responders: 15; unclassified: 1.
--treated in year 3: treated & monitored, 149; responders: 4; virologic responders: 138; non-responders: 7; unclassified: 0;
--treated in year 4: treated & monitored, 120; responders: 2; virologic responders: 113; non-responders: 5; unclassified: 0.
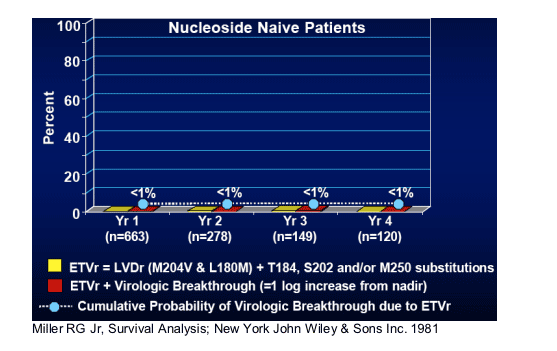
Genotypic Resistance & Virologic Breakthroughs
This table shows cumulative probability of virologic breakthrough due to ETV/r: <1% genotypic resistance & viral breakthrough in each of 4 years: yr 1: n=663; yr 2: n=278; yr 3: n=149; yr 4: n=120.
Virologic Profiles of Patients Experiencing a Virologic Breakthrough
Nucleoside Naive Patients
Year 1 (n = 14)
- 11 with no genotypic or phenotypic evidence of resistance
- 1 LVDr at baseline, becomes undetectable at Wk 102
- 1 with emerging LVDr (11 substitutions arise simultaneously)
- 1 LVDr at baseline, with emerging ETVr
Year 2 (n = 8)
- 7 with no genotypic or phenotypic evidence of resistance
- 1 LVDr at baseline, becomes undetectable at Wk 148
Year 3 (n = 2)
- 1 with no genotypic or phenotypic evidence of resistance
- 1 with simultaneous emergence of LVDr + ETVr substitutions
Year 4 (n = 1)
- No genotypic or phenotypic evidence of resistance
during the Q&A after the presentation Colonno was asked by someone at the microphone why some viral failures did not have resistance and he said perhaps non-compliance.
Virologic Profile of Virologic Breakthrough
Nucleoside Naive Patients
Study 027 HBeAg neg patient with LVDr (M204I + L80I) at the
time of study entry
- Subsequent emergence of LVDr M204V + L180M + ETVr S202G while on 0.5 mg ETV
- 273-fold decrease in ETV susceptibility and breakthrough at Week 48
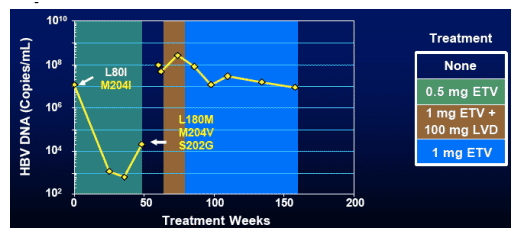
Virologic Profile of Virologic Breakthrough
Nucleoside Naive Patients
Study 022 HBeAg pos patient with undetectable (=0.1%) LVDr + ETVr substitutions at the time of study entry
- Received 16 weeks LVD+ETV at Week 100 in study 901
- Simultaneous appearance of L199V, L180M & M204V (LVDr) and S202G (ETVr) substitutions at Wk 139, and breakthrough at Wk 148
- Pattern of emergence suggestive of pre-existing variant
Genotypic Surveillance
Nucleoside Naive Patients
1 patient (Study 022 HBeAg pos) identified who had emerging LVDr & ETVr substitutions in absence of a virologic breakthrough
- Simultaneous appearance of genetically-linked Y54H, L180M, M204V (LVDr) & S202G (ETVr) substitutions at Wk 84
- Left study at Wk 96 without a virologic breakthrough
- Pattern of emergence suggestive of pre-existing variant
|
| |
|
 |
 |
|
|