 |
 |
 |
| |
GSK625433: A Novel and Highly Potent Inhibitor of the HCV NS5B Polymerase
|
| |
| |
Reported by Jules Levin
EASL, april 11-15, 2007
Barcelona, Spain
Fraser Gray, Liz Amphlett, Helen Bright, Lynn Chambers, Anne Cheasty, Rebecca Fenwick, David Haigh, David Hartley, Peter Howes, Richard Jarvest, Fereshteh Mirzai, Fabrizio Nerozzi, Nigel Parry, Martin Slater, Stephen Smith, Pia Thommes, Claire Wilkinson and Emily Williams
Infectious Disease Centre for Excellence in Drug Discovery, GlaxoSmithKline R&D, Stevenage, UK
Author Conclusions
The APs are a novel series of inhibitors that bind in the palm region of the HCV polymerase.
GSK625433 is a highly potent and selective inhibitor of genotype 1 HCV polymerases.
GSK625433 is synergistic with interferon-_ in vitro
Changes at amino acid positions 414 and 447 appear to be the key resistance mutations in vitro.
Resistance studies support the use of GSK625433 in combination with other polymerase/protease inhibitors in the clinic.
The biological and pre-clinical PK profiles support progression of GSK625433 into the clinic
Introduction
The NS5B RNA-dependent RNA polymerase is a critical enzyme in the lifecycle of the hepatitis C virus (HCV). It is well conserved within genotypes and between
subtypes and has become an attractive target for the development of antiviral agents. Different classes of HCV polymerase inhibitors are in the clinic that target different binding sites on the NS5B molecule and suppress HCV replication. We report here on the pre-clinical profile of GSK625433, a potent and selective inhibitor of the HCV NS5B polymerase from the acyl pyrrolidine series (APs)
that bind to the palm region of the NS5B polymerase.
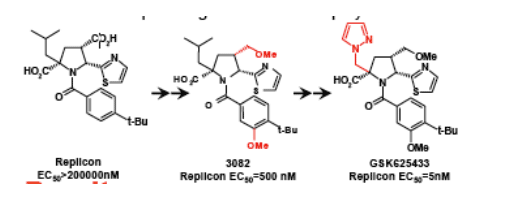
Results
The APs were identified by screening the GSK compound collection against the genotype 1b delta21 NS5B enzyme (2). Improvement of potency, cell penetration
and PK lead to the discovery of 3082 as the first candidate molecule from this series (9). Further iterations of SAR identified GSK625433 as the best molecule combining excellent potency and selectivity with good PK deemed suitable for clinical development. GSK625433 demonstrated highly potent and selective
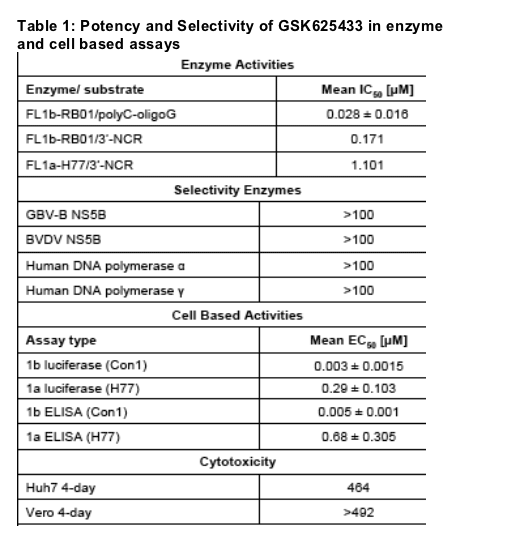
inhibition of the NS5B polymerase of HCV genotype 1 using both full length (Table 1) and delta21 enzymes (Table 2). The compound did not inhibit polymerases of other members of the Flaviviridae family, GBV-B and BVDV,
and did not inhibit human DNA polymerases (Table 1). In the HCV replicon system GSK625433 is highly potent against HCV genotype 1 in a variety of assay formats. In the presence of 40% human serum, its EC50 is shifted 3-
4-fold (data not shown). No significant cytotoxicity was observed.
Biochemical Assays: Performed using the purified enzymes (full-length or with C-terminal 21 amino acid truncation, delta21) and polyC-oligoG or 3'NCR substrates in the presence of _-33P-GTP. Incorporated radioactivity was determined by SPA or by adsorption to DEAE filtermat.
Replicon Assays: Conducted in Huh-7 cells containing subgenomic HCV RNA (8). ELISAs were performed on acetone:methanol fixed cells using an antibody against HCV NS5A and an HRP conjugated secondary antibody. Luciferase assays (5) were developed using Steady-Glo (Promega) after assessing cell viability with Resazurin.
Cytotoxicity assays: Performed on exponentially growing Huh7 or Vero cells
using MTT as a vital dye.
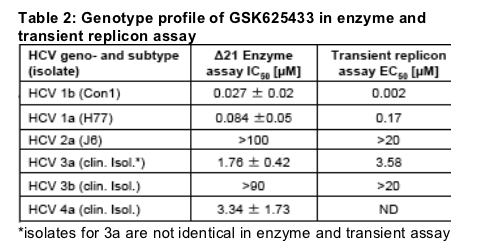
To establish a wider genotype profile of GSK625433 we purified delta21 enzymes from genotypes 1-4. In parallel we also exchanged the NS5B gene in the transient replicon for the respective one from other genotypes. All enzymes
showed good activity and a good replication signal in the transient assay. The genotype profile for GSK625433 in enzyme and transient replicon assay is comparable, with good potencies against genotype 1, reduced potencies
against genotypes 3a and 4a and inactivity against genotypes 2a and 3b.
Structural Studies
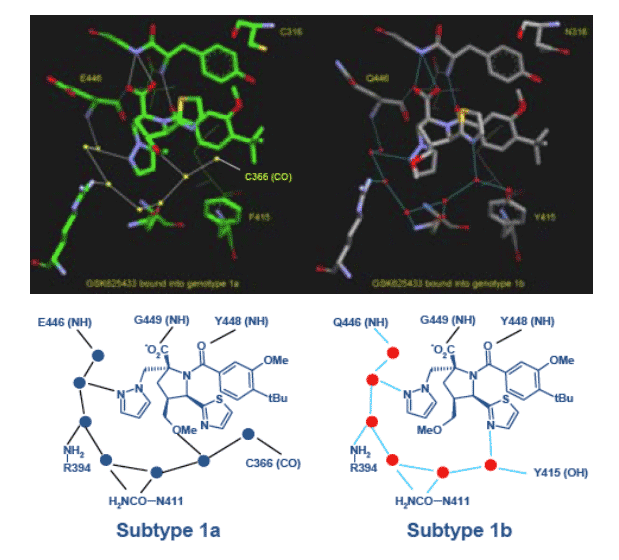
Through X-ray structural analysis of GSK625433 soaked into crystals of NS5B enzymes of subtypes 1a or 1b we have established that the compound binds into the palm region of the NS5B polymerase. However, the binding is subtly but critically different between subtypes 1a and 1b (Figure 1). Residue 415 is key to the interaction and the Y415F change between subtypes 1a and 1b affects the
water network around the ligand (6). This probably accounts for the different potencies against subtypes 1a and 1b of this class of molecules.
Resistance Studies
To generate replicon cell lines resistant to GSK625433 Huh-7 cells containing the HCV genotype 1b subgenomic RNA were passaged in the presence of compound (20x EC50). Discrete colonies grew through in 5-6 weeks and replicon RNA was isolated and sequenced. Mutations from the wild-type sequence were
individually introduced into the transient assay vector (5) and mutant replicons were tested for sensitivity against GSK625433. In vitro selection of resistant replicon cell lines in the presence of GSK625433 resulted in the identification of two mutations, within 5 A of the binding site, M414T and I447F. Each of these mutations individually confers resistance against GSK625433 (Table 3).
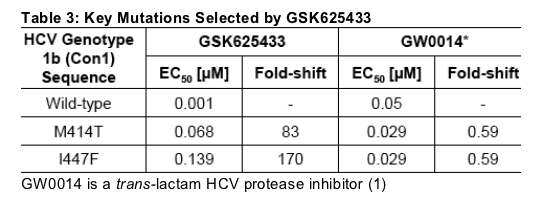
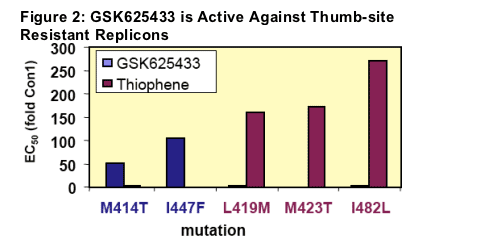
We tested the potency of different classes of NS5B inhibitors against the transient replicon containing the M414T or I447F mutations. A thiophene (3), which binds in the thumb region was equipotent against the M414T and wild-type replicons (Figure 3), while a thiadiazine (4) which also binds in the palm region had a >30-fold shift in potency (not shown). In addition, when GSK625433 was tested against transient replicons containing mutations selected in the presence of the thiophene (L419M, M423T and I482L; Ref12) it retained activity. Similarly, GSK625433 retained activity against enzymes containing mutations selected in the presence of compounds binding to the hinge (10) and finger-loop site
(7). This suggests that APs probably can be combined for therapy with either protease inhibitors or polymerase inhibitors binding to different sites.
IFN Combination Studies
It is likely that GSK625433 will be used clinically in combination with IFN-_. Combination assays between GSK625433 and IFN-_2a were performed in a two-dimensional chequerboard format, using three replicate plates for each assay. The plates were incubated and developed as per the standard ELISA assay. In these combination studies synergy between GSK625433 and IFN-_2a could be demonstrated (Figure 4). GSK625433 was also synergistic with ribavirin in vitro (not shown).
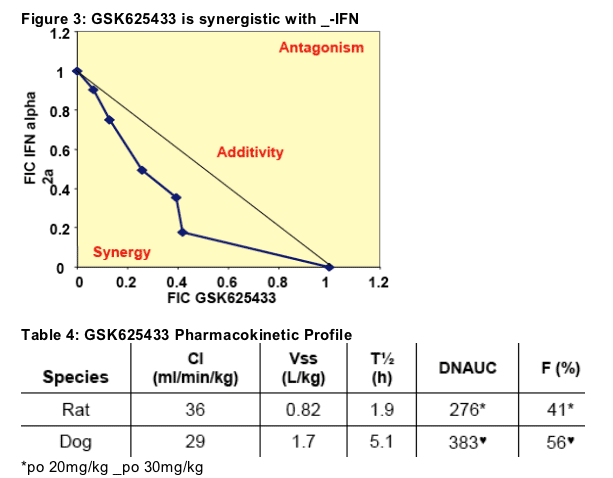
GSK625433 has been evaluated in both the rat and the dog (Table 4) and has good PK in both species to support its progression into pre-clinical toxicology
studies. In the rat, GSK625433 was preferentially distributed to the target organ and gave a liver:plasma ratio of 21:1 based on AUC24h.
References
1. Andrews D. et al (2003). Eur. J. Med. Chem 38:339-343
2. Burton G. et al. (2005) Bioorg. Med. Chem. Lett. 15:1553 - 1556
3. Chan L. et al (2004) Bioorg. Med. Chem. Lett. 14:797-800
4. Dhanak D. et al (2002) J. Biol. Chem. 277:38322-38327
5. Friebe P et al (2005) J.Virology 75:12047-12057
6. Haigh, D. et al (2006) 13th Int. Symposium on HCV and related viruses.
7. Kukolj, G. et al. (2005) J. Biol. Chem. 280, 39260-39267
8. Lohmann V. et al. (1999) Science, 285: 110-113
9. Slater M. et al. (2007) J. Med. Chem. 50: 897 - 900
10.Thommes, P. et al (2006) 13th Int. Symposium on HCV and Related Viruses.
11.Vrolijk JM et al (2003) J. Virological Methods, 110: 201-209
12.Zuercher T et al. (2005) 12th Int. Symposium on HCV and Related Viruses.
|
| |
|
 |
 |
|
|