 |
 |
 |
| |
14 Weeks vs 24 Weeks Peg/RBV for Genotypes 2/3
|
| |
| |
Reported by Jules Levin
EASL, April 15, 2007
Barcelona, Spain
This was the very last oral presentation at the conference so by this time many attendees had left to go home but I attended this interesting session where there was a presentation on a new therapy celgosivir, which I reported on, and this study. I returned home the next morning but if you recall there was a noreaster storm so many of us returning on the same flight to Newark were concerned about being able to land. Howver, we landed on time as apparently the rain and storm had stopped already in NYC.
"Peginterferon Alfa-2b and Ribavirin for 14 or 24 Weeks in Patients with HCV Genotype 2 or 3 and Rapid Virological Response (RVR): the North-C Trial"
Olav Dalgard et al, Infectious Disease Dept, Ulleval University Hospital, Oslo, Norway
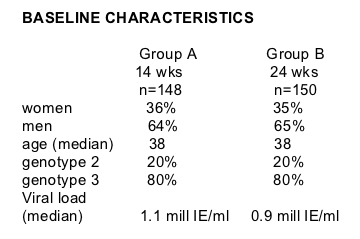
The aim of the study is to compare the virological response of 14 and 24 weeks of treatment to patients with genotype 2 or 3 infection and rapid virological response (RVR). This is a randomized, multicenter study: 49 hospitals in Sweden, Denmark, and Norway. It is a non-inferiority trial. Patients were required to have elevated ALT, no biopsy required, and were treatment-naïve. Patients received pegylated interferon alfa-2b s.c. 1.5 ug/kg/weekly, and ribavirin 800-1400 mg/daily weight-based dose. Rapid viral response was defined as HCV RNA <50 IU/mL at week 4. Sustained viral response (EVR) was HCV RNA <50 IU/mL 24 weeks after end of treatment. If patients were HCV RNA negative after 4 weeks they were randomized to an additional 10 or 20 weeks treatment. Patients who were HCV RNA positive received 24 weeks treatment. Dalgard had previously published results of a pilot study of 94 patients (Dalgard, Hepatology 2004) where the SVR rate was 91% and relapse rate was 9% with 14 weeks treatment.
The author concluded that "90% with genotype 2/3 and RVR obtain SVR after 14 weeks treatment; reduced side effects, saved costs, and good response to retreatment, make shorter treatment a rational approach for these patients, in our opinion; even though longer treatment may provide better response". 95% receiving 24 weeks therapy achieved SVR. These results were apparently "per protocol", that is with >80/80/80 adherence.
This was a non-inferiority trial but due to patient dropouts they did not have enough patients remaining for the final analysis to draw a conclusion that 14 weeks was as good as 24 weeks. In the final per protocol analysis 90% of patients in the 14 week group had SVR (10% relapse rate) and 95% in the 24 week group had SVR (5% relapse rate). Based on an intent-to-treat analysis 81% in the 14 week group had SVR and 91% in the 24 week group had SVR, with a 19% relapse rate in the 14 week group and 9% relapse rate in the 24 week group, but the author said "the majority of the relapsers are lost to followup".
They identified a few subgroups of patients that had a lower SVR rate. Genotype 3 patients had 85% SVR rate with 14 weeks compared to 92% with 24 weeks. They were concerned about patients with high viral loads but surprisingly patients with low viral load (<400,000 IU/mL) had a lower response rate with 14 weeks than patients with low viral load receiving 24 weeks (86% vs 100%), "there is a big difference although the numbers of patients were small": 42 in the low viral load group/14 weeks and 55 in the low viral load/24 weeks group. The author said "it seems that patients when they start treatment with low viral load they more often relapse with short treatment duration, and this seems logical, we can speculate on why that happens. Patients with genotype 3 and low viral load <400,000 IU/ml) "more often had relapse", SVR rate of 81% after shorter duration treatment of 14 weeks (n=36) compared to 100% SVR rate for patients with genotype 3/low viral load who received 24 weeks treatment. The author also said age was "an important determinant for response" with younger patients achieving high rates of SVR and the SVR rates declining as age increases. In Scandanavia "we often see young patients who seek treatment". Patients younger than 30 years had 97% SVR rate with 14 weeks and 100% SVR rate with 24 weeks (n=65), while patients >50 years had 81% SVR rate with 14 weeks and 83% SVR rate with 24 weeks treatment. There were only 39 patients in the subgroup who were older than 50 years so the author said "is it safe to offer short duration of therapy to older patients? It would seem so from this trial although we have only 39 patients in this group". Of note the age groups of 30-39 yrs (84% vs 96%, n=103) and 40-49 (83% vs 90%, n=78) yrs also had lower SVR rates with 14 weeks therapy compared to 24 weeks therapy. The author added however age could be a surrogate marker for stage of disease and they did not do biopsies.
There were a number of patients lost to follow-up in the study because they were "former drug users". As well, some patients had less than 80/80/80 adherence; in the long treatment arm, 24 weeks, patients tended to stop treatment early, 48 patients (author said "1 in 3 patients stopped early). Because they had previously conducted a pilot trial with similar results and clinicians and patients knew the results and because of side effects these patients apparently decided to stop treatment earlier. While in the 14 week treatment arm only 11 patients stopped treatment early out of 148 patients between weeks 4 and 40. After the database was locked they discovered another problem, "these were former drug users' and I think he said young patients, as there were 9 patients lost to follow-up in the 14 week arm and 4 in the 24 week arm. But since the SVR was so high it's a big problem to call them treatment failures. So they decided to base their conclusions on the per protocol population: patients who received more than 80% of both drugs and with follow-up data.
COSTS
Mangia et al published in the NEJM 2004: 9/10 relapsers following 12 weeks treatment obtained SVR after 24 weeks retreatment. Retreatment is a "very good option" although 24 weeks may provide better response. The authors calculated that even after retreatment of 1 in 10 patients 14 weeks treatment will reduce costs by 30-35% (approximately Euro 3,500, per patient).
SAMPLE SIZE
Non-inferiority margin; 10%
Significance level: 2.5%
Power: 80%
Sample size: 435 (300 randomized)
PATIENT FLOW
428 patients were enrolled.
HCV RNA <50 IU/mL at week 4 (n=302) (71%).
Group A: 14 weeks treatment, ITT population, n=148
<80/80/80, n=11
Lost to follow-up, n=9
PER PROTOCOL POPULATION, n=128 (>80% of both drugs and followup data)
Group B: 24 weeks treatment, ITT population, n=150
<80/80/80 n=48
Lost to follow-up, n=4
PER PROTOCOL POPULATION: n=98 (>80% drugs and followup data)
SVR PER PROTOCOL
Group A: 90% SVR, 10% relapse (n=128)
Group B: 95% SVR, 5% relapse (n=98)
5.1% difference (95% CI: -2.4, +12.4)
Non-inferiority margin: 10%
Author said: "the 95% confidence interval overlaps 10% probably because we lost power as we only have 98 patients in the control group. Therefore, we cannot conclude non-inferiority".
SUB-GROUP ANALYSIS
SVR and Genotype
Genotype 2: 93% SVR 14 weeks (n=29), 97% SVR 24 weeks (n=31)
Genotype 3: 85% SVR 14 weeks (n=110), 92% 24 weeks (n=1150
Viral Load
<400,000 IU/mL: 86% SVR 14 weeks (n=42), 100% 24 weeks (n=55)
>400,000 IU/mL: 88% 14 weeks (n=88), 88% 24 weeks (n=85)
Genotype & Viral Load
Genotype 3 and low viral load (<400,000 IU/mL): 81% SVR 14 weeks (n=36), 100% 24 weeks (n=44)
Genotype 3 and high viral load (>400,000 IU/mL): 86% 14 weeks (n=66), 86% 24 weeks (n=66)
ADHERENCE
>80/80/80: 90% SVR 14 weeks (n=128), 95% SVR 24 weeks (n=98)
<80/80/80: 57% SVR weeks (n=11), 90% 24 weeks (n=44)
|
| |
|
 |
 |
|
|