 |
 |
 |
| |
48 Weeks & High Ribavirin Dose May Improve SVR Rates for Genotype 2/3, French study by Bernard Willems
|
| |
| |
Reported by Jules Levin
EASL, April 11-15, 2007
Barcelona, Spain
Several HCV studies presented with oral talks were of particular interest at EASL. Bernard Willems from France presented data suggesting that for genotype 2 & 3 patients who do not achieve a Rapid Viral Response (week 4), 48 weeks Peg/RBV with 1000/1200 mg RBV attains higher SVR rates and lower relapse rates than 24 weeks peg/RBV and than using 800 mg RBV. Willems presented data from a retrospective look at 2 early pivotal Pegasys/RBV studies to see if 48 weeks peg/RBV would be better than 24 weeks in genotype 2/3 patients if they do not achieve a RVR (week 4 response). In one study the SVR and relapse rates were better for genotype 2 & 3 patients who received 48 weeks and RBV 1000/1200 than for patients who received 24 weeks with either 800 or 1000/1200 mg RBV. But, in the second study the SVR was not better with 48 weeks. However, the results were not statistically significant, the numbers of patients in the analysis was small, and this examination was retrospective, so Willems concluded "formal prospective studies should be done before 48 weeks treatment duration can be recommended in these patients". I will report Willems full data set. (see slides from Willems talk below).
"Should treatment with peginterferon plus ribavirin be intensified in patients with HCV genotype 2/3 without a rapid virologic response?"
Bernard Willems1, S.J. Hadziyannis2, T.R. Morgan3,
M. Diago4, P. Marcellin5, D. Bernstein6, P.J. Pockros7, A. Lin8,
M.L. Shiffman9, S. Zeuzem10
1Hôpital Saint-Luc-Centre Hospitalier de l'Universite de Montreal, Montreal, Quebec, Canada; 2Henry Dunand Hospital, Athens, Greece, 3VA Medical Center, Long Beach, USA; 4Hospital General Valencia, Spain; 5Hôpital Beaujon, France; 6North Shore University Hospital, Manhasset, USA; 7Scripps Clinic, La Jolla, USA; 8Roche, Nutley, USA; 9Virginia Commonwealth University Medical Center, Richmond, USA;
10J.W. Goethe-University Hospital, Frankfurt, Germany
BACKGROUND
In the treatment of chronic hepatitis C, genotypes 2 and 3 are considered as "easier to treat".
Patients with G2 or G3 who have a Rapid Virologic Response (RVR) achieve high rates of Sustained Virologic Response (SVR) with 12 to 16 weeks of treatment with pegylated interferon plus ribavirin.
However, genotype 2/3 patients who do not achieve an RVR have a much lower SVR.1-3
(RVR = HCV RNA <50 IU/mL at week 4)
In the ACCELERATE trial (n=1469), the SVR rate was 90% in patients with RVR and only 49% in patients without RVR.4
These patients may benefit from a more intensified treatment regimen (longer duration, higher ribavirin dose).
1. Mangia A, et al. N Engl J Med 2005; 352: 2609
2. von Wagner M, et al. Gastroenterology 2005; 129: 522
3. Yu M-L, et al. Gut 2007; 56: 553
4. Shiffman M, et al. 41st EASL 2006; Abstract 734
In patients infected with genotype 1:
-- Patients with RVR (undetectable HCV RNA at week 4) achieve high rates of SVR (89%) with a shorter duration of 24 weeks1
--Patients with cEVR (complete Early virologic Response: no RVR but undetectable HCV RNA at week 12) achieve high rates of SVR (67%) with the currently recommended regimen of 48 weeks2
--Patients with pEVR (partial Early virologic Response: no RVR and detectable HCV RNA but a >2 log10 drop at week 12) achieve improved rates of SVR (>50%) with an extended treatment duration of 72 weeks 3-5
1. Jensen D, et al. Hepatology 2006; 43: 954
2. Marcellin P, et al. 42nd EASL 2007 Abstract 613
3. Sanchez-Tapias J, et al. Gastroenterology 2006; 131: 451
4. Berg T, et al. Gastroenterology 2006; 130: 1086
5. Ferenci P, et al. 57th AASLD 2006; Abstract 390
STUDY OBJECTIVE
To determine whether an intensified regimen of peginterferon alfa_2a (40KD) plus ribavirin may be beneficial in HCV genotype 2/3 patients who do not achieve an RVR.
METHODS
Data were retrospectively analysed from two randomised, multicentre phase III studies in which patients were treated with peginterferon alfa-2a (40KD) 180 mg/wk plus ribavirin
In NV15942, patients were randomised to 24 or 48 weeks of peginterferon alfa-2a (40KD) plus ribavirin 800 or 1000/1200 mg/day1
In NV15801, patients received 48 weeks of peginterferon alfa-2a (40KD) plus ribavirin 1000/1200 mg/day2
1. Hadziyannis S, et al. Ann Intern Med 2004; 140: 346
2. Fried M, et al. N Engl J Med 2002; 347: 975
PATIENTS
-- Treatment naive
-- Age ≥18 years
-- Quantifiable HCV RNA (>600 IU/mL) in serum
-- Elevated serum ALT levels
-- Liver biopsy consistent with a diagnosis of chronic hepatitis C
Compensated liver disease
PROPORTION OF GENOTYPE 2/3 PATIENTS WITH AN RVR
A high proportion of genotype 2/3 patients achieved an RVR in each study:
-- 74% (362/492) in NV15942 (HADZIYANNIS et al.)
-- 58% (81/140) in NV15801 (FRIED et al.)
In a multiple logistic regression analysis, significant independent factors associated with achieving an RVR were:
-- Genotype 2 versus 3 (p<0.001)
-- Non-US region versus US region (p<0.001)
-- Low baseline viral load (HCV RNA <400 000 IU/mL) (p<0.005)
Genotype 2/3 patients with an RVR: SVR and relapse rates
(RVR = HCV RNA <50 IU/mL at week 4)
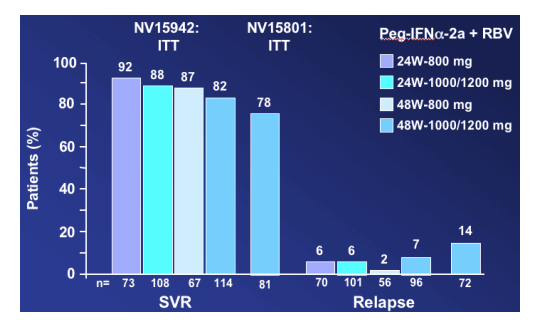
In the NV15942 Hadziyannis study, among patients with an RVR, the SVR rates are 82% to 92%: 92% for 24w-800mg; 88% for 24w-1000/1200mg; 87% for 48w-800mg; 82% for 48w-1000/1200mg. The relapse rates were low, 2% to 7%. Willems said "there was no benefit in longer treatment or higher dose of RBV". In the NV15801 study SVR rate is 78% and the relapse rate was 14% "even if the patients received 48 weeks and high dose of RBV".
Genotype 2/3 patients without an RVR: SVR and relapse rates
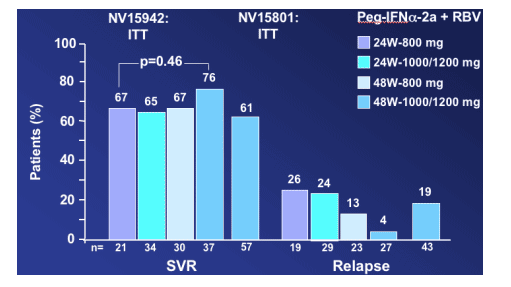
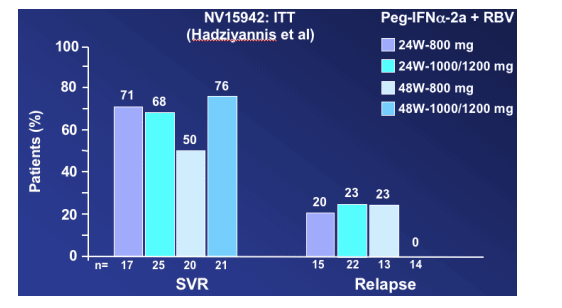
In the Hadziyannis study NV15942, the highest response rate was 76% SVR (4% relapse rate, the lowest) for patients receiving 48 w-1000/1200mg compared to 65% to 67% for patients receiving 24w-800mg, 24w-1000/1200mg, or 48w-800mg, but the difference was not statistically significant (p=0.46) "with the available sample size". Willems said he calculated that almost 400 patients would have had to be in each treatment group to demonstrate a difference of 9%. In the second study (Fried NV15801), the SVR rate was 61% and the relapse rate was 19%, "even if those patients received 48 weeks and high dose of ribavirin.
Genotype 2/3 patients without an RVR: SVR and relapse rates
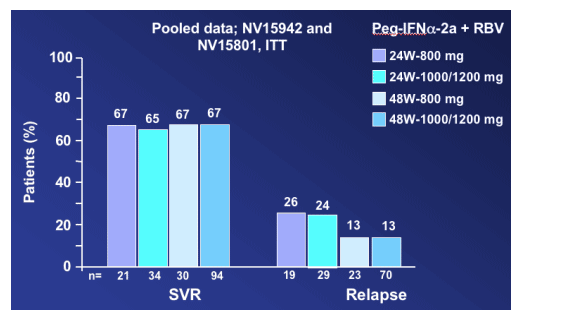
The data is pooled from the 2 studies in this slide and then there is no difference in SVR rate (65-67%). The relapse rate is lower (13%) for patients receiving 48 weeks compared to 24 weeks treatment (24-26%).
Genotype 2 patients without an RVR: SVR and relapse rates
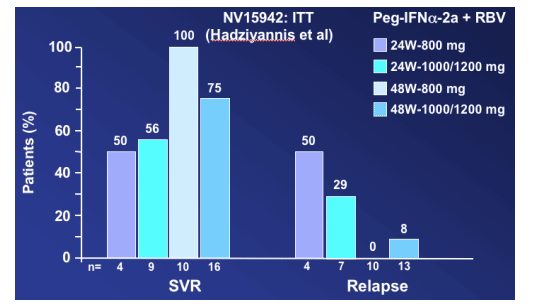
In this analysis of NV15942, "the best SVR rate and the lowest relapse rate was for patients receiving 48 weeks but the numbers are small".
Genotype 3 patients without an RVR: SVR and relapse rates
The best SVR rate and lowest relapse rate was with 48 weeks and the highest RBV dose, and again the numbers are small.
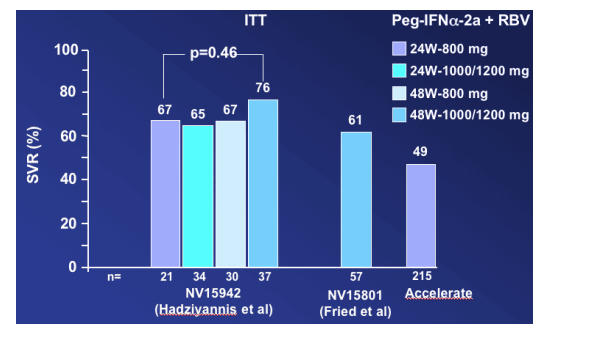
Genotype 2/3 patients without an RVR: SVR rates across studies: Summary
In the Accelerate trial patients received 24 weeks treatment and 800mg RBV. Patients without an RVR had 41% SVR.
Willems concluded:
Genotypes 2 and 3 are not necessarily "easy-to-treat" genotypes.
Patients who do not achieve an RVR demonstrate disappointing SVR rates with the recommended 24 weeks of treatment.
This retrospective cross-study analysis does not permit a recommendation for intensifying treatment in all patients.
However, an overall decrease in relapse rates with a longer duration could be observed.
Formal prospective studies should be done before 48 weeks treatment duration can be recommended in these patients.
|
| |
|
 |
 |
|
|