 |
 |
 |
| |
HCV Protease Inhibitor ITMN-191 Resistance
|
| |
| |
Reported by Jules Levin
EASL, April 2007, Barcelona, Spain
SEQUENCE VARIATION OF NS3/4A IN HCV REPLICONS EXPOSED TO ITMN-191 CONCENTRATIONS ENCOMPASSING
THOSE LIKELY TO BE ACHIEVED FOLLOWING CLINICAL DOSING
S. D. Seiwert, J. Hong, S. R. Lim, T. Wang, P.T. Ravi Rajagopalan, K. Kossen, H. Tan, and L. M. Blatt
InterMune, Inc., Brisbane, CA
"....These data suggest the genetic barrier to resistance in patients upon clinical dosing may be high with ITMN-191....." note from Jules: there are 17 substantive new HCV drugs in pre-clinical and clinical development (in patients). We are at the beginning of trying to understand drug resistance. Drug resistance will be an issue, but it is likely to be different than in HIV...will we be able to sequence protease inhibitors, perhaps...but we don't know yet. Will we be able to eliminate peginterferon...perhaps never, we don't know but it will take 6 years to find out...will we be able to eliminate ribavirin...we don't know and this should take at least 2 years or more to find out...clearly, combination therapy with oral HCV drugs is the therapeutic regimen for the future, whichfor at least a number of years will contain also peginterferon plus likely also ribavirin.
ABSTRACT
Background: Approximately one half of patients with chronic Hepatitis C do not achieve a Sustained Virologic Response (SVR) with pegylated interferon alpha and ribavirin which constitute the standard of care (SoC). Direct antiviral agents may improve the rate SVR when used in combination with SoC or in novel regimens. ITMN-191 is a potent HCV NS3/4A protease inhibitor in clinical development. The identity of escape mutations in NS3/4A that may be observed in patients following clinical dosing of ITMN-191 will likely reflect drug exposure. Because drug exposure in patients may be variable, NS3 sequence variation was characterized in vitro at ITMN-191 concentrations ranging from an order of magnitude below the 12 hr postdose liver concentration (Cmin) in primate to the approximate liver Cmax in rat (20 nM to 25,000 nM).
Results: In confirmation of previous work, HCV replicons exposed to ITMN-191 concentrations approximately equivalent to primate liver Cmin (≦320 nM) carry the single substitution D168A. At concentrations encompassing rat liver Cmin (≥320 nM), D168A, D168V or D168E are observed in combination with additional substitutions within 10A of the ITMN-191 binding site. Variants carrying both D168V and A156S/V predominate. At concentrations near rat liver Cmax (25 _M), the most predominant variant carries a total of 6 substitutions within 10A of the ITMN-191 binding site. Besides D168V and A156S/V, these changes include Q41R and S138T in the P1/P1' region, S489L in a portion of the helicase domain that buttresses the P2 pocket, and a substitution at a site in the NS4A peptide cofactor that borders the P1' pocket (V23A). None of the predominant amino acid substitutions in NS3 are observed in the Los Alamos HCV sequence database, suggesting these substitutions would not be immediately accessible for viral escape in naïve patient populations.
Conclusions: NS3 sequence undergoes very dynamic changes when subject to increasing concentrations of ITMN-191. Substitution at D168 appears fundamental to resistance but is not sufficient at most of the ITMN-191 liver concentrations observed in animals. Rather, substitution at multiple sites is required. These data suggest the genetic barrier to resistance in patients upon clinical dosing may be high.
AUTHOR SUMMARY & CONCLUSIONS:
Two independent selections suggest substitution at D168 is fundamental to ITMN-191 resistance in vitro
-- Substitution at position 168 alone is sufficient at primate liver C12hr (mid-nM)
-- Additional substitution at A156 or F43 required at approximate rat liver C12hr
(single digit _M)
-- At rat liver Cmax (25 _M), a variant carrying 6 substitutions predominates
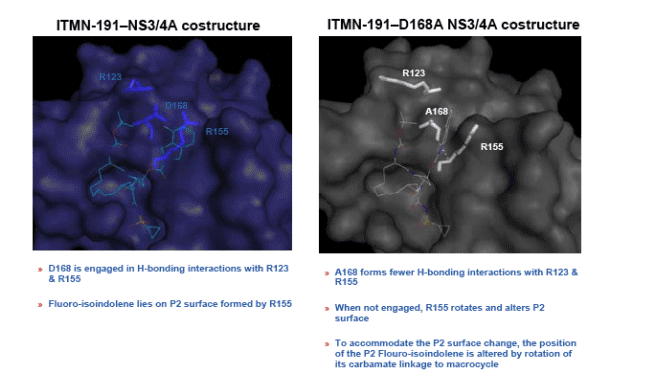
D168A reduces potency of ITMN-191 by altering its binding mode to NS3/4A with a consequent increase in the decay rate of an ITMN-191-NS3/4A complex.
While clinical resistance studies will be driven by virologic breakthrough, these studies provide a guide for potential mechanisms of resistance.
ITMN-191 (R7227) background
--NS3 protease active site inhibitor
--Displays preferential liver exposure in preclinical species
--In phase I clinical development
-- Genotype 1b replicon IC50 = 1.8 nM
-- Replicon elimination by 45 nM
Biochemical characteristics inhibition
-- ITMN-191 binds to NS3/4A through a two-step mechanism and displays a slow off rate (like diketoamides). No current evidence suggests formation of a covalent adduct (unlike di-keto amides).
-- Because of the two step binding mechanism, potency increases when ITMN-191 and NS3/4A are preincubated.
- IC50 without preincubation of ITMN-191 and NS3/4A 800 pM
- IC50 with preincubation of ITMN-191 and NS3/4A 120 pM
At ITMN-191 concentrations achieved in the liver of preclinical species, the more stable complex is rapidly formed.
The stable complex has a t1/2 ≥ 5 hr
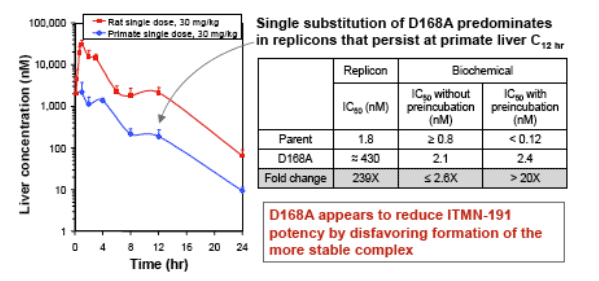
Selection 1: NS3 variation at ITMN-191 concentrations achieved in primate liver 12 hours post dose
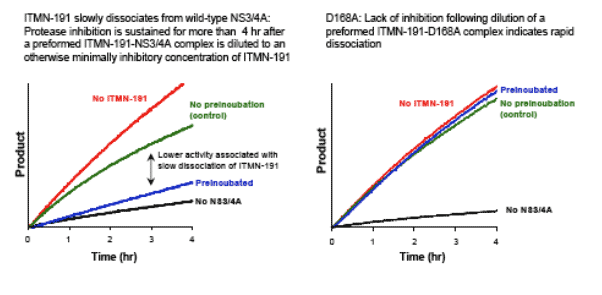
Reduced potency of ITMN-191 against D168A bearing NS3 is associated with
faster compound dissociation
Altered position of P2 group of ITMN-191 and/or altered position of NS3/4A residues is associated with faster compound dissociation and a loss of potency against D168A
Selection 2: characterization of NS3/4A sequence dynamics over a 1,000-fold concentration range encompassing animal liver exposure
NS3/4A cloned and sequenced from replicons that persisted at each concentration
Substitutions considered significant if:
- Present in more than 1 clone at a given concentration OR
- Present at more than drug concentration AND
- Were within 10A of compound binding site
NS3 sequence undergoes very dynamic changes as selection pressure increases:
- 11 substituted positions
- 24 amino acid identity changes
- 46 different NS3/4A genotypes
- Substitutions in P1', P1, P2, P3 and P4
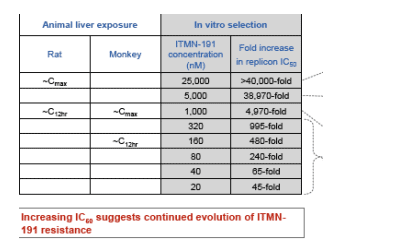
Most prevalent variants across concentrations achieved in animal liver
--Corresponds to >40,000 fold increase in replicon IC50
D168V, A156S/V in combination with changes in P1' (Q41R), NS4A (V23A), P1 (S138T) and in helicase domain that buttresses P2 (S489L) dominates at rat liver Cmax
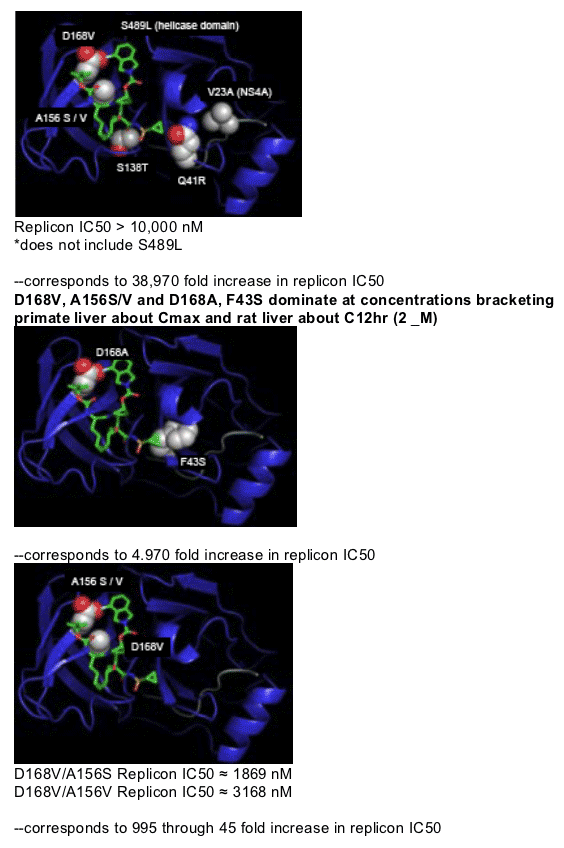
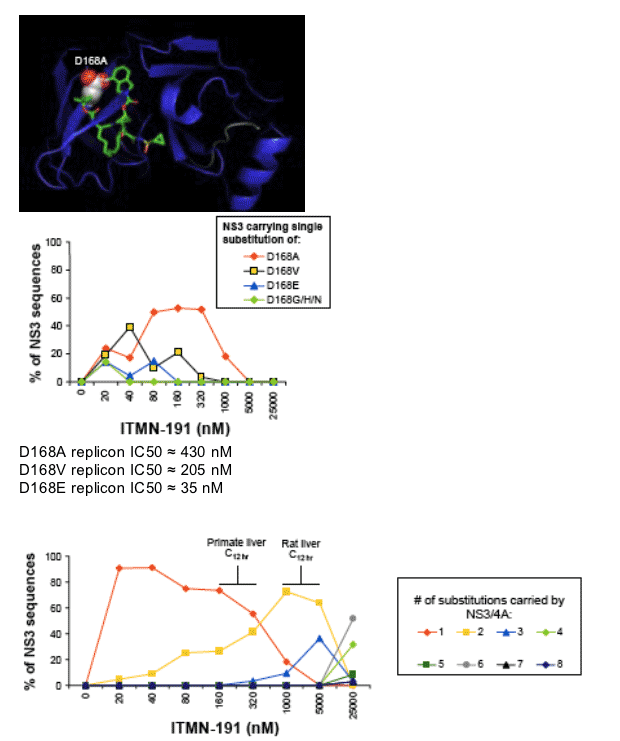
Substitution of a single amino acid is sufficient at primate liver C12 hr
Multiple substitutions favored at ≥ rat liver C12 hr
Viral escape in humans may require multiple substitutions depending
upon liver exposure
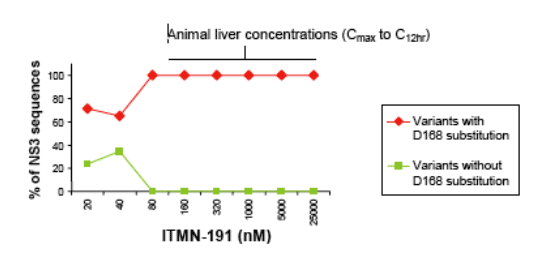
D168 appears to be an obligatory substitution at ITMN-191 concentrations
observed in the liver of animals
Additional substitutions added to this "base" at higher concentrations
|
| |
|
 |
 |
|
|