| |
The Natural History and Treatment of Chronic Hepatitis B: A Critical Evaluation of Standard Treatment Criteria and End Points (HBV-DNA, ALT)
|
| |
| |
Ching-Lung Lai, MD, and Man-Fung Yuen, MD, PhD
From the University of Hong Kong, Hong Kong, China.
Potential Financial Conflicts of Interest: Grants received: C.L. Lai (Bristol-Myers Squibb).
Annals of Internal Medicine
3 July 2007 | Volume 147 Issue 1 | Pages 58-61
The definite indications for the treatment of chronic hepatitis B are serum hepatitis B virus (HBV) DNA levels greater than 105 copies/mL and alanine aminotransferase (ALT) levels more than 2 times the upper limit of normal. If cirrhosis is present, an HBV DNA level greater than 105 copies/mL is the sole criterion for treatment. Treatment end points include hepatitis B e antigen (HBeAg) seroconversion for HBeAg-positive patients, reduction of HBV DNA levels to less than 105 copies/mL, and normalization of ALT values. These guidelines may apply to patients who acquire the hepatitis B infection during adolescence or adulthood but are less suitable for most hepatitis B carriers, who are infected in early life. Cirrhosis complications, including hepatocellular carcinoma, often occur in this latter group despite HBeAg seroconversion, HBV DNA levels less than 104 copies/mL, or ALT levels between 0.5 and 2 times the upper limit of normal. Therefore, HBeAg seroconversion may not be an adequate end point for these patients; the ideal treatment end points are permanent suppression of HBV DNA to levels undetectable by polymerase chain reaction and reduction of ALT levels to less than 0.5 times the upper limit of normal. (Anna Lok says in her Commentary below: Until treatments that are safe and affordable for long-term use can achieve permanent suppression of HBV replication in all or most patients, we recommend treatment only for patients who have high serum HBV DNA levels and active or advanced liver disease and deferral of treatment for patients with normal ALT levels who have a low likelihood of progressive liver disease (those who are young, are HBeAg- positive, and have persistently normal ALT levels) until it is indicated).
Approximately 350 to 400 million people worldwide have chronic hepatitis B virus (HBV) infection, with roughly 1.25 million cases in the United States and 180 000 in Britain. More than 70% of all people with chronic HBV infection are Asian. Although white persons in the U.S. and British populations are usually infected in adolescence or adulthood (by means of sexual contact or sharing of intravenous needles), nearly every Asian person with this infection (whether born in or outside Asia) acquires it at birth from an infected mother or within 2 years of birth by means of close contact with infected relatives.
There are important differences between these 2 groups. Patients infected in adolescence or adulthood immediately enter an immune clearance phase; their disease is of short duration and tends to become quiescent after they seroconvert from hepatitis B e antigen (HBeAg) to antibodies against HBeAg (anti-HBe). Such patients have been termed "healthy" carriers (1). In contrast, patients infected early in life have a prolonged immune tolerance phase followed by a prolonged immune clearance phase before HBeAg seroconversion, and strong evidence indicates that their disease progresses after HBeAg seroconversion.
Guidelines for treatment and treatment end points from 3 regional liver associations (the American Association for the Study of Liver Diseases, the European Association for the Study of the Liver, and the Asian Pacific Association for the Study of the Liver) suggest that treatment should be initiated for patients who are HBeAg seropositive if their HBV DNA levels are 105 copies/mL or greater (≥20 000 IU/mL) and their alanine aminotransferase (ALT) levels are greater than 2 times the upper limit of normal (Table) (2-4). Liver biopsy may be considered for patients with ALT levels between 1 and 2 times the upper limit of normal, particularly those older than 40 years, and treatment may be initiated if there are abnormal histologic findings. They also suggest that treatment can be stopped after seroconversion to anti-HBe and a decrease in HBV DNA to less than 105 copies/mL (<20 000 IU/mL) and in ALT to normal levels.
Table. Guidelines for Treatment and Treatment End Points from the 3 Regional Liver Associations
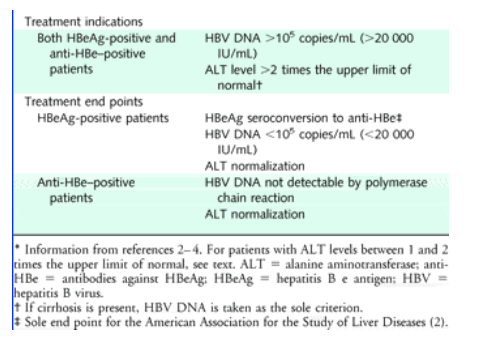
These guidelines assume that chronic HBV infection is likely to progress to cirrhosis and hepatocellular carcinoma only in patients with these markers of active infection and that treatment can be stopped once a patient achieves a healthy hepatitis B carrier state (5), defined as seronegativity for HBeAg (or seropositivity for anti-HBe) and HBV DNA and ALT levels below the indicated thresholds. This concept of a healthy hepatitis B carrier state may apply to patients infected in adolescence or adulthood, but the following evidence suggests that it does not apply to patients infected earlier in life.
Hepatitis B e Antigen
In a 30-year follow-up of 296 blood donors from northern Italy, there was no clinically significant liver illness or mortality compared with 157 uninfected control participants (6). The investigators attribute this favorable prognosis to the absence of comorbid conditions, inactive HBV infection, and abstinence from alcohol. Another study, a long-term follow-up of white, HBeAg-positive, interferon-treated patients and untreated control participants, showed that patients who cleared HBeAg survived much longer than those who remained HBeAg positive (7). The essential contributor to the favorable outcome in these groups is probably that the patients were infected in adolescence or adulthood.
In contrast, other studies show that the disease continues to progress after HBeAg seroconversion in patients infected early in life. A study from Taiwan of 683 patients showed that new cirrhosis developed at similar annual rates (1.3% vs. 2.4%) in HBeAg-positive and anti-HBe-positive patients (8). In a recent study of 3233 Asian patients without cirrhosis, the median age at HBeAg seroconversion was 35 years, whereas the age at development of cirrhosis complications was 57.2 years (9). More than 73% of the patients were anti-HBe positive when cirrhosis complications and hepatocellular carcinoma developed. In a follow-up of 92 Chinese carriers of hepatitis B surface antigen (HBsAg) with HBsAg seroclearance, 37% still had intrahepatic HBV DNA. Furthermore, 5.4% of these patients developed hepatocellular carcinoma during follow-up compared with 8.7% of 92 control patients who did not lose HBsAg (10), a statistically insignificant difference.
These findings establish that hepatocellular carcinoma can develop even after HBsAg seroclearance. A study from Taiwan of 11 893 men in which HBeAg positivity was associated with increased risk for hepatocellular carcinoma (11) seems to contradict this conclusion; however, the participants' HBeAg and anti-HBe status were examined only at the time of enrollment. Because the participants were between 30 and 65 years of age, a substantial proportion of them would have HBeAg seroconversion during the 9 to 10 years of follow-up, and the effect of HBeAg seroconversion subsequent to enrollment on the development of hepatocellular carcinoma is unknown. In addition, the relative risk for hepatocellular carcinoma was still 9.6 times higher in participants who were anti-HBe positive at enrollment than in noncarriers.
Hepatitis B Virus DNA
In a retrospective study of 165 Chinese patients, 45% of HBeAg-negative patients with active hepatitis (defined as abnormal ALT levels) had HBV DNA levels less than 105 copies/mL (12). In another study, 67.1% of 79 Chinese carriers with cirrhosis-related complications were anti-HBe positive, with 37.7% having HBV DNA levels less than 105 copies/mL (<20 000 IU/mL) and 24.5% having levels less than 104 copies/mL (<2000 IU/mL) (13).
A study of 3653 carriers from Taiwan concludes that an HBV DNA level of at least 104 copies/mL (≥2000 IU/mL) is a strong risk predictor for hepatocellular carcinoma (14). During follow-up, the hazard ratio for the development of hepatocellular carcinoma was 10.1 (95% CI, 6.3 to 16.2) for persons whose HBV DNA level remained greater than 105 copies/mL (>20 000 IU/mL) compared with those who had levels less than 104 copies/mL (<2000 IU/mL) at enrollment. However, the hazard ratio was still substantial at 3.8 (CI, 1.7 to 8.4) for persons whose HBV DNA levels had decreased from greater than 105 copies/mL (>20 000 IU/mL) on enrollment to less than 104 copies/mL (<2000 IU/mL) during follow-up.
These studies suggest that a clinically significant proportion of Asian patients continue to be at risk for active hepatitis and its complications after their HBV DNA levels decrease to less than 105 copies/mL (<20 000 IU/mL) or even 104 copies/mL (<2000 IU/mL).
Alanine Aminotransferase
In a prospective cohort study of 94 533 men and 47 522 women with no known liver disease, mortality from liver disease was greater in patients with ALT levels between 0.5 and 1 times the upper limit of normal than in those with ALT levels less than 0.5 times the upper limit of normal (15). We suggest that adjustment of the normal range for ALT may be necessary.
For Asian patients with chronic hepatitis B, the previously mentioned study of 3233 patients found that patients with ALT levels less than 0.5 times the upper limit of normal had the lowest risk for complications, patients with ALT levels of 0.5 to 1 times the upper limit of normal had a statistically significantly increased risk for complications, and patients with ALT levels greater than 1 to 2 times the upper limit of normal had the highest risk.
Implications
These studies suggest that the current treatment criteria and end points are not satisfactory for most patients with chronic hepatitis B who are infected early in life. The disease continues to progress in a substantial proportion of patients who achieve standard milestones, such as HBeAg seroconversion, HBsAg clearance, HBV DNA levels less than 105 copies/mL (<20 000 IU/mL) or even 104 copies/mL (<2000 IU/mL), and ALT levels between 0.5 and 2 times the upper limit of normal. For these patients, HBeAg seroconversion may be an inappropriate end point (16), because immune damage to the liver may continue after HBeAg seroconversion even with relatively low levels of viral replication. To prevent the development of complications, patients with ALT levels between 0.5 and 2 times the upper limit of normal should be treated; placebo-controlled trials are needed to confirm effectiveness. In addition, HBV DNA suppression should continue until the level decreases to less than 104 copies/mL (<2000 IU/mL).
Two studies demonstrated the effectiveness of long-term suppression of HBV replication with nucleoside or nucleotide treatment. A study of 63 HBeAg-positive patients who took lamivudine for 3 years (17) and a study of 185 HBeAg-negative patients who took adefovir for 96 weeks (18) showed reversal of fibrosis and even cirrhosis. However, the emergence of the tyrosine, methionine, aspartate, aspartate (YMDD) mutation in the virus during lamivudine therapy blunts the improvements (17).
Two other studies investigated the effects of nucleoside and nucleotide analogues on the development of cirrhosis complications and hepatocellular carcinoma. In 651 patients with confirmed advanced fibrosis or cirrhosis, lamivudine treatment reduced cirrhosis complications and hepatocellular carcinoma compared with placebo (by 7.8% and 17.7%, respectively; P = 0.001) after a median of 32.4 months (19). Patients who developed YMDD viral mutations were more likely than those without these mutations to have an increase in their Child-Pugh score (P < 0.001) but were still less likely than those receiving placebo to develop complications (P > 0.05).
A similar reduction in the development of cirrhosis or hepatocellular carcinoma was found in 142 HBeAg-positive Asian patients without cirrhosis who received lamivudine for a median of 89.9 months compared with 124 untreated control patients (P = 0.005) (20). Patients with YMDD viral mutations after lamivudine treatment (76.3% after 6 years of treatment) had more cases of cirrhosis and hepatocellular carcinoma than those without YMDD mutations (P = 0.394); however, there were still fewer cases than in the untreated patients (P = 0.024).
Finally, long-term suppressive therapy may reduce covalently closed circular HBV DNA, a major transcriptional template for HBV replication and the probable source of HBV DNA rebound after treatment is stopped prematurely. One year of treatment with nucleoside and nucleotide analogues may decrease serum and intrahepatic covalently closed circular DNA by 1 to 2 logs (21, 22).
Concerns about Early and Prolonged Hepatitis B Virus Therapy
Concerns about both early and prolonged suppressive therapy for chronic HBV include development of HBV resistance, adverse drug effects, the benefits of early treatment versus the desirability of waiting until the disease is active, the efficacy of treatment when ALT levels are normal, and the cost of treatment. We respond to each of these concerns below.
Although resistance to nucleoside and nucleotide analogues develops with prolonged treatment, patients receiving lamivudine who develop resistant viral mutations still fare better than untreated patients (19, 20). Furthermore, newer, more potent drugs are associated with much lower rates of resistance.
Extensive preclinical safety tests have been conducted on all nucleoside and nucleotide analogues that are licensed or in phase III studies. No evidence of carcinogenicity was seen with lamivudine, adefovir, and telbivudine. Lung and liver adenoma and carcinomas were seen in mice exposed to levels of entecavir 3 to 40 times greater than that recommended for humans, but none were seen in rats, dogs, or monkeys. For comparison, the observation that lipid-lowering drugs, such as fibrates and the statins, have caused cancer in some rodents at levels of exposure similar to those prescribed for humans (23) has not prevented their widespread use.
The consensus to wait until the disease is active before starting treatment raises the question of when the disease becomes active. Some patients with normal ALT levels have clinically significant fibrosis and disease morbidity and mortality (10, 24). It is surely advisable to initiate treatment before fibrosis and cirrhosis set in.
Therapeutic agents are ineffective when ALT levels are normal only if the desired end point is HBeAg seroconversion; this reasoning does not apply to HBeAg-negative patients whose disease is still progressing. Taking viral suppression as the chief end point of treatment, nucleoside and nucleotide analogues are equally effective in patients with both normal and elevated ALT levels.
Finally, the cost of prolonged treatment should be balanced against the substantially higher medical cost incurred when the disease progresses from chronic hepatitis to compensated cirrhosis, decompensated cirrhosis, and hepatocellular carcinoma, or when it progresses so far that liver transplantation is needed (25).
Conclusions
A new algorithm for the treatment of chronic hepatitis B, proposed by an independent group of hepatologists in the United States, deals with some of the issues we have raised (26). The ideal end points of treatment, at least for patients infected in early life, should be permanent suppression of HBV DNA to very low levels (preferably below the threshold of detectability by polymerase chain reaction) and ALT levels below 0.5 times the upper limit of normal. These end points are achievable with existing therapeutic tools. Therapy with nucleoside and nucleotide analogues may also reduce covalently closed circular HBV DNA, making clearance of this substance (and thus of nonintegrated HBV) a distinct possibility with prolonged treatment.
COMMENTARY
When to Start and Stop Hepatitis B Treatment: Can One Set of Criteria Apply to All Patients Regardless of Age at Infection?
Bulent Degertekin, MD, and Anna S.F. Lok, MD
From the University of Michigan Health System, Ann Arbor, MI
Annalsof Internal Medicine
3 July 2007 | Volume 147 Issue 1 | Pages 62-64
Existing guidelines for the treatment of hepatitis B virus (HBV) infection recommend that only patients with active or advanced liver disease and high serum HBV DNA levels be treated and that, in patients who are initially seropositive for hepatitis B e antigen (HBeAg), treatment can be stopped 6 months after HBeAg seroconversion (1-3). In this issue, Lai and Yuen (4) raise concern that those recommendations are inappropriate for patients with perinatal HBV infection. They propose that patients with normal alanine aminotransferase (ALT) levels who acquire infection early in life should also be treated and that HBeAg-positive patients should continue treatment after HBeAg seroconversion. The 2007 update of the American Association of the Study of Liver Disease guidelines (5) address some of Lai and Yuen's concerns by recommending that treatment also be considered for patients with intermittent or mildly elevated ALT levels; those who remain HBeAg- positive with high serum HBV DNA levels after 40 years of age; and HBeAg-negative patients with HBV DNA levels of 10 000 to 100 000 copies/mL (2000 to 20 000 IU/mL), particularly if liver histologic examination shows significant inflammation or fibrosis (5). Here, we put other aspects of their proposal in the context of existing evidence.
People with ALT values within the normal range are traditionally considered to have healthy livers. However, recent evidence suggests that the upper limits of normal in most diagnostic laboratories are erroneously high, owing to inclusion of patients with asymptomatic liver disease (6). Two studies reported that persons with ALT levels that are 0.5 to 1 times greater than the upper limit of normal had higher mortality from liver disease or cirrhosis complications than did persons with ALT levels less than 0.5 times the upper limit of normal (7, 8). Small studies have also found that up to one third of HBV carriers with normal ALT levels have moderate inflammation or advanced fibrosis (9, 10). However, the natural course of chronic HBV infection varies and may be punctuated by recurrent fluctuations in serum HBV DNA and ALT levels (11), and a study from Taiwan found that disease progressed slowly among HBeAg-positive patients with normal ALT levels: The cumulative probability of cirrhosis after 17 years was 12.6%, and none of the 240 patients developed hepatocellular carcinoma (12). These latter data suggest that treatment can be deferred in most patients with persistently normal ALT levels and that the presence of risk factors for progressive liver disease, such as older age, HBeAg positivity, high serum HBV DNA levels, and co-infection with hepatitis C or D virus, can be used to identify patients with normal ALT levels who warrant further evaluation and treatment.
Hepatitis B e antigen seroconversion was long thought to be associated with cessation of HBV replication and remission of liver disease. However, evidence from the past 2 decades shows that HBV DNA remains detectable in serum after HBeAg seroconversion, albeit at low levels (13). In some patients, high serum HBV DNA levels and active liver disease persist after HBeAg seroconversion (HBeAg-negative chronic hepatitis), a state frequently associated with precore HBV variant (14) and HBV genotype D (and less commonly genotype B and C) infection.
The recognition that HBV persists after HBeAg seroconversion has led to the term inactive carrier state, with the understanding that the virus is not eradicated and liver disease can become active again. Nevertheless, a study from Taiwan found that two thirds of patients with perinatal HBV infection had sustained remission and very low risk for cirrhosis and hepatocellular carcinoma up to 9 years after spontaneous HBeAg seroconversion (15). On the basis of these data, HBeAg seroconversion seems an appropriate treatment end point in most patients, provided that they continue to be monitored and treatment is reinitiated when hepatitis is reactivated. This is particularly true when a strict definition of HBeAg seroconversion\namely, HBeAg loss, HBe antibody detection, a nondetectable (or very low) serum HBV DNA level, and normalization of ALT values\is used as the treatment end point.
The major difference between perinatal and adult-acquired HBV infection is the presence of a long immune tolerance phase (10 to 40 years) in patients with perinatal HBV infection, during which patients are HBeAg-positive with a high serum HBV DNA level but a normal ALT level (11). Patients in this phase have a very low rate of spontaneous or treatment-related HBeAg seroconversion. Available data suggest that the next phases of immune clearance (HBeAg positivity, high HBV DNA levels, and high ALT levels) and inactive carrier state (HBeAg negativity, low or undetectable HBV DNA levels, and persistently normal ALT levels) are similar for patients with perinatal or adult-acquired HBV infection. Some patients will progress from the inactive carrier state to HBeAg-negative chronic hepatitis. This transition is not unique to patients with perinatal HBV infection; in fact, it is most commonly associated with HBV genotype D, which is usually acquired during childhood or during adulthood. The basis for existing recommendations to defer treatment in HBeAg-positive patients with normal ALT levels is the low rate of HBeAg seroconversion with treatment and the generally slow rate of disease progression during the immune tolerance phase in patients with perinatal infection. Although HBV replication after HBeAg seroconversion can be reactivated in all patients independent of age at infection, an occurrence of 30% over 10 years (15) does not justify continuing treatment for all patients.
Beyond questions about when to start and stop treatment, Lai and Yuen's proposal reflects the conviction that the ideal treatment end point, permanent suppression of HBV DNA to undetectable levels on polymerase chain reaction, can be achieved with current therapies. Available data suggest that after a 1-year course of pegylated interferon therapy, sustained HBeAg seroconversion is achieved in only approximately 30% of HBeAg-positive patients and sustained suppression of serum HBV DNA to undetectable levels is achieved in approximately 20% of HBeAg-negative patients (5). A 1-year course of oral nucleoside therapy (lamivudine, adefovir, entecavir, or telbivudine) results in undetectable serum HBV DNA in 20% to 70% of HBeAg-positive patients and 50% to 90% of HBeAg-negative patients (5). However, viral relapse occurs in more than 90% of patients after treatment withdrawal, except in patients who have sustained HBeAg seroconversion or the rare patient who has cleared HBsAg (16). Extending treatment with lamivudine or telbivudine beyond 1 year is associated with a decreasing proportion of patients with undetectable serum HBV DNA because of selection of drug-resistant mutants. Continued treatment for HBeAg-negative patients with adefovir for up to 5 years has been reported to increase the proportion of patients with undetectable serum HBV DNA from 72% at the end of 1 year to 80% at the end of 2 years, with a subsequent decrease to 67% at the end of 5 years (17). Preliminary data indicate that treating HBeAg-positive patients with entecavir for up to 3 years resulted in undetectable serum HBV DNA in 87% of patients (18).
These long-term efficacy data are based on few patients: 70 for adefovir and 122 for entecavir. More important, viral suppression will not be permanent in all patients, given that the rate of drug resistance will increase with duration of treatment even with new drugs (such as entecavir) and nonadherence to therapy is more likely over time. Finally, most clinical trials enroll only patients with elevated pretreatment ALT levels; response rates may be lower in patients with normal ALT levels. A high pretreatment ALT level is the best predictor of interferon- and nucleoside treatment-related HBeAg seroconversion (19). Thus, although currently approved treatments can suppress serum HBV DNA to undetectable levels on polymerase chain reaction, no data show yet that any of these treatments can result in permanently undetectable virus in all patients.
Because HBV cannot be eradicated, many physicians have argued that patients with hepatitis B should receive lifelong treatment, as is given for other chronic medical illnesses. However, rates of drug resistance increase with duration of treatment (even with new therapies), long-term safety data are lacking, and treatment is very expensive (approximately $18 000 per year for pegylated interferon and $2500 to $9000 per year for nucleosides). Until treatments that are safe and affordable for long-term use can achieve permanent suppression of HBV replication in all or most patients, we recommend treatment only for patients who have high serum HBV DNA levels and active or advanced liver disease and deferral of treatment for patients with normal ALT levels who have a low likelihood of progressive liver disease (those who are young, are HBeAg- positive, and have persistently normal ALT levels) until it is indicated. Likewise, discontinuation of treatment in HBeAg-positive patients who have confirmed HBeAg seroconversion and have completed an additional 6 months of consolidation treatment is appropriate as long as they continue to be monitored and treatment is reinitiated if HBV is reactivated. We believe that these recommendations apply to all patients with hepatitis B regardless of their age at infection.
Potential Financial Conflicts of Interest: Consultancies: A.S.F. Lok (Bristol-Myers Squibb, Gilead Sciences, GlaxoSmithKline, Idenix, Innogenetics, Pharmasset, Roche, Schering-Plough); Grants received: A.S.F. Lok (Bristol-Myers Squibb, Gilead Sciences, GlaxoSmithKline, Idenix, Innogenetics, Roche, Schering-Plough).
References
1. Liaw YF, Leung N, Guan R, Lau GK, Merican I, McCaughan G, et al. Asian-Pacific Consensus Update Working Party on Chronic Hepatitis B. Asian-Pacific Consensus Statement on the management of chronic hepatitis B: a 2005 update. Liver Int. 2005;25:472-89. [PMID: 15910483].[Medline]
2. de Franchis R, Hadengue A, Lau G, Lavanchy D, Lok A, McIntyre N, et al. EASL Jury. EASL International Consensus Conference on Hepatitis B. 13-14 September, 2002 Geneva, Switzerland. Consensus statement (long version). J Hepatol. 2003;39(Suppl 1):S3-25. [PMID: 14708673].[Medline]
3. Lok AS, McMahon BJ, Practice Guidelines Committee American Association for the Study of Liver Diseases (AASLD). Chronic hepatitis B: update of recommendations. Hepatology. 2004;39:857-61. [PMID: 14999707].[Medline]
4. Lai CL, Yuen MF. The natural history and treatment of chronic hepatitis B: a critical evaluation of standard treatment criteria and end points. Ann Intern Med. 2007;147:58-61.[Abstract/Free Full Text]
5. Lok AS, McMahon BJ. Chronic hepatitis B. Hepatology. 2007;45:507-39. [PMID: 17256718].[Medline]
6. Prati D, Taioli E, Zanella A, Della Torre E, Butelli S, Del Vecchio E, et al. Updated definitions of healthy ranges for serum alanine aminotransferase levels. Ann Intern Med. 2002;137:1-10. [PMID: 12093239].[Abstract/Free Full Text]
7. Kim HC, Nam CM, Jee SH, Han KH, Oh DK, Suh I. Normal serum aminotransferase concentration and risk of mortality from liver diseases: prospective cohort study. BMJ. 2004;328:983 [PMID: 15028636].[Abstract/Free Full Text]
8. Yuen MF, Yuan HJ, Wong DK, Yuen JC, Wong WM, Chan AO, et al. Prognostic determinants for chronic hepatitis B in Asians: therapeutic implications. Gut. 2005;54:1610-4. [PMID: 15871997].[Abstract/Free Full Text]
9. Nguyen M, Trinh H, Garcia T, Ahmed A, Keeffe E. Significant histologic disease in HBV-infected patients with normal to minimally elevated ALT levels at initial evaluation [Abstract]. Hepatology. 2005;42:593A. Abstract no. 1008.
10. Lai M, Hyatt B, Afdahl N. Role of liver biopsy in patients with normal ALT and high HBV DNA [Abstract]. Hepatology. 2005;42:720A. Abstract no. 1322.
11. Yim HJ, Lok AS. Natural history of chronic hepatitis B virus infection: what we knew in 1981 and what we know in 2005. Hepatology. 2006;43:S173-81. [PMID: 16447285].[Medline]
12. Chu CM, Hung SJ, Lin J, Tai DI, Liaw YF. Natural history of hepatitis B e antigen to antibody seroconversion in patients with normal serum aminotransferase levels. Am J Med. 2004;116:829-34.[Medline]
13. Chu CJ, Hussain M, Lok AS. Quantitative serum HBV DNA levels during different stages of chronic hepatitis B infection. Hepatology. 2002;36:1408-15. [PMID: 12447866].[Medline]
14. Carman WF, Jacyna MR, Hadziyannis S, Karayiannis P, McGarvey MJ, Makris A, et al. Mutation preventing formation of hepatitis B e antigen in patients with chronic hepatitis B infection. Lancet. 1989;2:588-91. [PMID: 2570285].[Medline]
15. Hsu YS, Chien RN, Yeh CT, Sheen IS, Chiou HY, Chu CM, et al. Long-term outcome after spontaneous HBeAg seroconversion in patients with chronic hepatitis B. Hepatology. 2002;35:1522-7. [PMID: 12029639].[Medline]
16. Hadziyannis SJ, Tassopoulos NC, Heathcote EJ, Chang TT, Kitis G, Rizzetto M, et al. Adefovir Dipivoxil 438 Study Group. Long-term therapy with adefovir dipivoxil for HBeAg-negative chronic hepatitis B. N Engl J Med. 2005;352:2673-81. [PMID: 15987916].[Abstract/Free Full Text]
17. Hadziyannis SJ, Tassopoulos NC, Heathcote EJ, Chang TT, Kitis G, Rizzetto M, et al. Adefovir Dipivoxil 438 Study Group. Long-term therapy with adefovir dipivoxil for HBeAg-negative chronic hepatitis B for up to 5 years. Gastroenterology. 2006;131:1743-51. [PMID: 17087951].[Medline]
18. Chang T, Chao YC, Kaymakoglu S, Cheinquer H, Pesoa M, Gish R, et al. Entecavir maintained virologic suppression through 3 years of treatment in antiviral naive HBeAg(+) patients (ETV 022/901) [Abstract]. Hepatology. 2006;44:229A. Abstract no. 109.
19. Perrillo RP, Lai CL, Liaw YF, Dienstag JL, Schiff ER, Schalm SW, et al. Predictors of HBeAg loss after lamivudine treatment for chronic hepatitis B. Hepatology. 2002;36:186-94. [PMID: 12085364].[Medline]
|
|
| |
| |
|
|
|