 |
 |
 |
| |
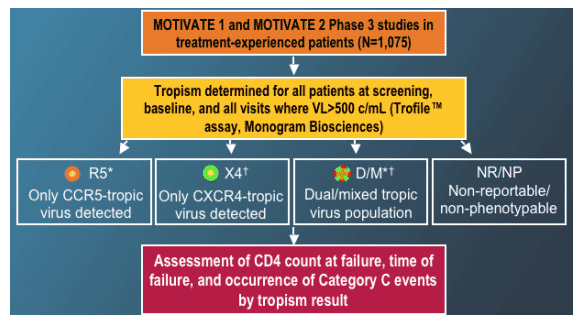
Changes in HIV-1 Co-receptor Tropism for Patients Participating in the Maraviroc MOTIVATE 1 and 2 Clinical Trials
|
| |
| |
Reported by Jules Levin
47th ICAAC
Chicago, USA, September 17-20, 2007
E van der Ryst and M Westby
Pfizer Global Research and Development, Sandwich, UK
AUTHOR CONCLUSIONS
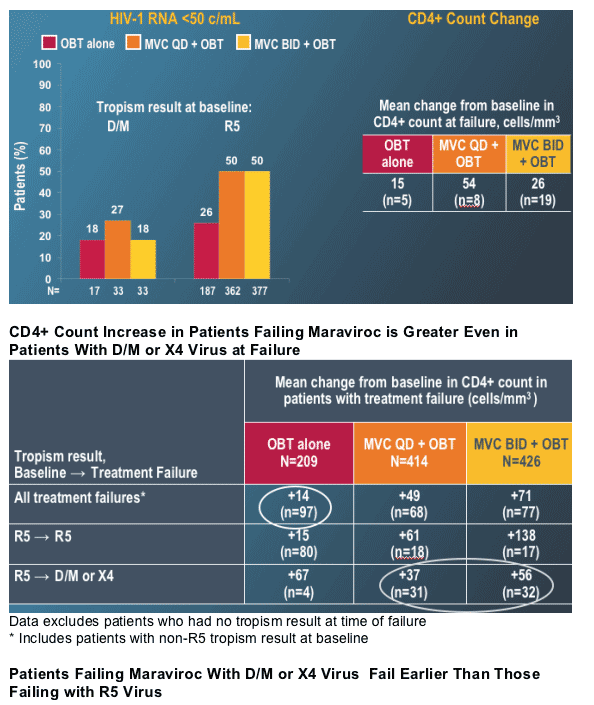
--Tropism changes are associated with MVC treatment failure
--Patients failing MVC therapy had higher mean CD4+ count increases even in the context of emergence of D/M or X4 virus
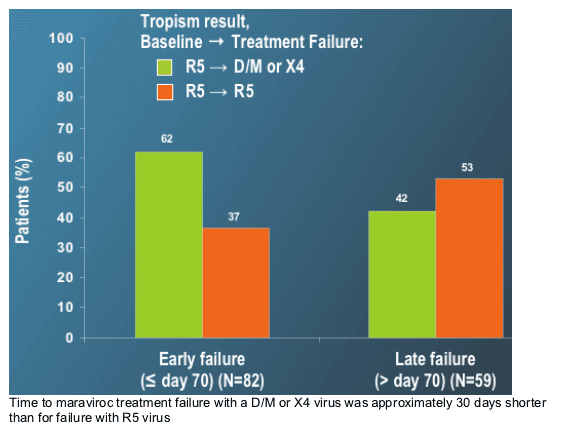
--Time to failure was shorter for patients failing with D/M or X4 virus vs R5 virus
--Patients who failed MVC therapy with D/M or X4 virus reverted to an R5 virus tropism result after cessation of MVC therapy
--There was no association between Category C events and treatment-emergent D/M or X4 virus
--These data are consistent with the selective and reversible suppression of R5 virus during MVC therapy, resulting in detection of D/M or X4 virus at time of failure in two-thirds of failing patients
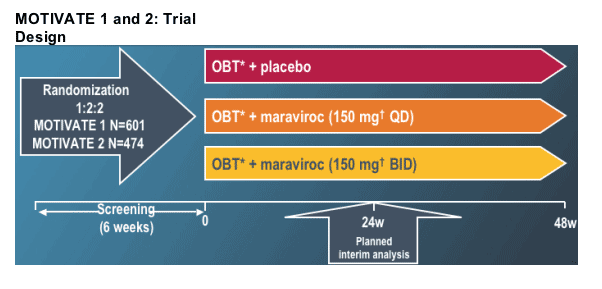
Patient eligibility criteria:
--R5 HIV-1 infection
--HIV-1 RNA ≥5,000 copies/mL
--Stable pre-study ARV regimen, or no ARVs for ≥ 4 weeks
-- Resistance to and/or ≥ 6 months' experience with ≥ one ARV
from three classes (≥ two for PIs)
Patients stratified by:
-- Enfuvirtide use in OBT
-- HIV-1 RNA < and ≥100,000 copies/mL at screening
Patients With a Change in Tropism Result from R5 at Screening to D/M at Baseline had a Lower Median CD4+ Count
--Of the 1042 patients with R5 virus at screening, approximately 8% had a change in tropism result between screening and baseline to non-R5 virus, prior to a change in ARV regimen or administration of study drug
--This subgroup had a lower median CD4+ count and higher mean HIV-1 RNA at screening

No Association Between Category C Events and Treatment-emergent D/M or X4 Virus
--The number of patients experiencing CDC Category C events in the study was low: 14 (6.7%) OBT alone, 26 (6.3%) MVC QD and 18 (4.2%) MVC BID
--There was no evidence of an increased rate of Category C events in patients receiving maraviroc + OBT vs those receiving OBT alone despite the extended treatment duration on maraviroc1,2
--Only 5 patients with R5 virus at baseline who experienced a Category C event had D/M or X4 virus at the time of the event (3 on MVC QD, 1 on MVC BID, and 1 on OBT alone)
--All 5 patients had a baseline CD4 count <20 cells/mm3 and were therefore at high risk of developing a Category C event
--Category C events were therefore not associated with treatment-emergent CXCR4-using virus
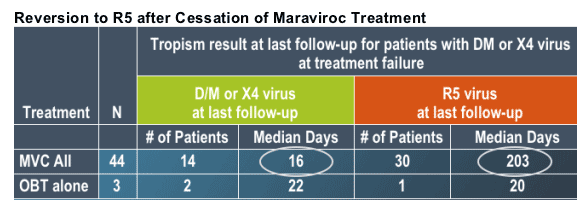
--For maraviroc patients with D/M or X4 virus at treatment failure and with in-study off-drug (ISOD) follow-up data, virus in 68% of patients was R5 at last follow-up
--Time of follow-up was significantly shorter for patients with D/M or X4 virus at their last study visit
--Where follow-up >1 month, virus in 30 out of 31 patients reverted to R5 during ISOD follow-up
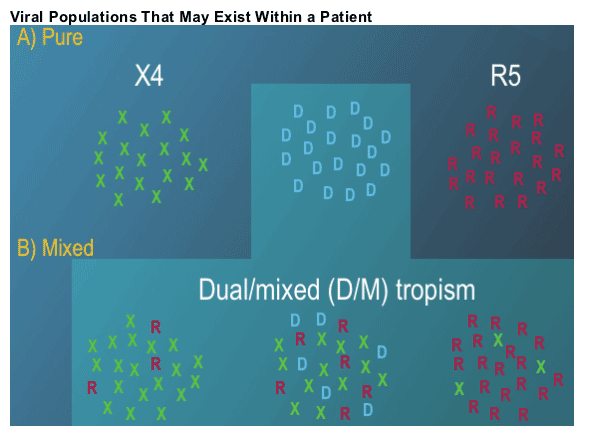
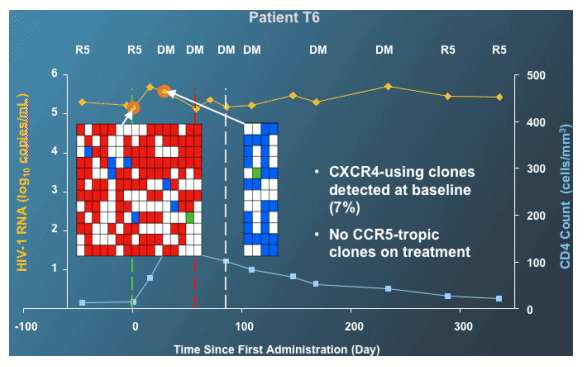
Selective Inhibition of R5 Viruses can Lead to a Change in Tropism Result to D/M or X4
--Trofile (like all resistance tests) measures relative proportions (not absolute amounts) of different viruses (Panel A)
--Selective inhibition of a majority virus type, increases the sensitivity to detect the minor variant (Panel B)
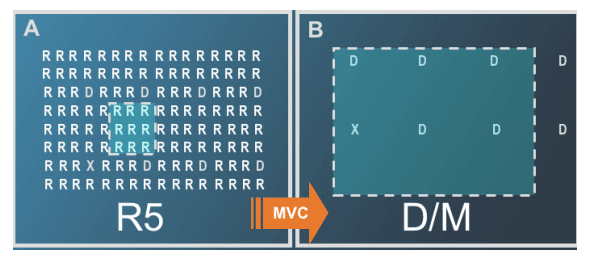
Selective Inhibition of R5 Viruses Can Lead to a Change in Tropism Result to D/M or X4
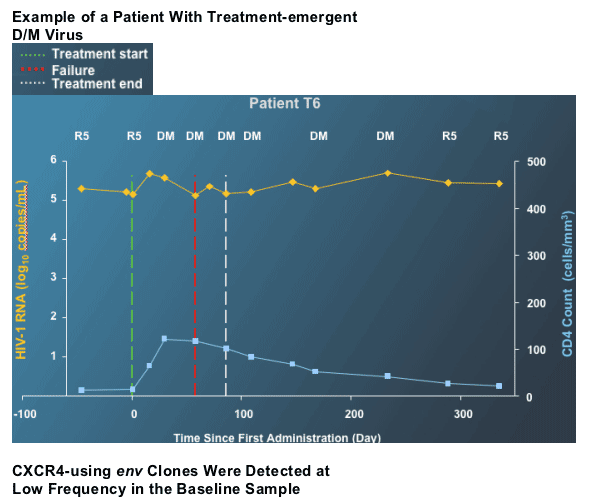
--Maraviroc selectively inhibits R5 virus
--If maraviroc is administered as part of a sub-optimal regimen, pre-existing low (undetected) levels of D/M or X4 virus will emerge as the dominant viral population
--Since the D/M or X4 virus is pre-existing, time to failure is shorter than with R5 virus (where maraviroc resistance must be selected de novo)
--Similar to the rapid outgrowth of pre-existing (archived) drug-resistant virus when failed ARV therapy is reinitiated after treatment interruption
--After withdrawal of maraviroc, selective pressure on R5 virus is removed, allowing R5 virus to re-emerge as the dominant population
--Reversion to R5 takes approximately 16 weeks, consistent with loss of 3TC1 or enfuvirtide2 resistance after withdrawal of these ARVs
|
| |
|
 |
 |
|
|