 |
 |
 |
| |
Raltegravir (RAL) Dose Proportionality and Effect of Food
|
| |
| |
Reported by Jules Levin
ICAAC Sept 2007, Chicago
L. A. Wenning1, M. S. Anderson1, A. S. Petry1, E. J. Friedman1, J. T. Kost1, S. James1, K. Strohmaier1, D. L. Miller1,
S. Merschman1, F. A. Bieberdorf2, S. D. Flach3, M. Iwamoto1, J. A. Stone1, J. A. Wagner1
1Merck & Co., Inc., Whitehouse Station, NJ, 2CEDRA, Austin TX, 3Covance, Madison, WI
Reported by Jules Levin
ICAAC Sept 2007, Chicago
AUTHOR CONCLUSIONS
Single doses of RAL up to 1600 mg are generally well tolerated.
RAL AUC0-∞, Cmax, and C12hr are dose proportional over the range 100 to 800 mg, indicating plasma clearance and bioavailability of RAL are independent of dose.
A high fat meal affected the rate, but not extent, of absorption of RAL. Data from Phase II trials suggests that the effect of food on C12hr is not clinically meaningful.
Twice-daily dosing without regard to food is appropriate for RAL.
INTRODUCTION
RAL is a novel HIV-1 integrase inhibitor with potent in vitro activity against HIV-1 (IC95 =33 nM in 50% human serum).
The studies presented here were designed to evaluate:
_ The safety, tolerability, and pharmacokinetic profile of RAL following administration of single doses of the final market image (FMI) formulation
_ The dose proportionality of RAL over the dose range 100 mg to 1600 mg
_ The effect of a high-fat meal on pharmacokinetics of RAL
ABSTRACT
Background RAL is a novel HIV-1 integrase strand transfer inhibitor with potent in vitro activity against HIV-1 (IC95 = 33 nM in 50% human serum). Two studies were performed to characterize biopharmaceutic properties of the final market image tablets of RAL.
Methods Two single-dose, randomized crossover studies were performed: a 5-period study to assess the dose proportionality of RAL within the range 100 to 1600 mg and a 2-period study to assess the effect of a standard highfat
meal on a 400 mg dose, each in 20 healthy adult subjects. Clinical safety evaluations were performed throughout the studies.
Results There were no serious adverse experiences (AEs) reported and no
discontinuations due to AEs. AUC0-∞ and Cmax of RAL were dose-proportional from 100 to 1600 mg. C12hr increased proportionally from 100 to 800 mg, and slightly less than proportionally from 100 to 1600 mg. The apparent terminal t1/2
of RAL was ∼9 hrs with a ∼1 hr initial phase t1/2 accounting for much of the AUC. From the food study, the geometric mean ratio (fed/fasted) and 90% confidence interval for AUC0-∞ was 1.19 (0.91, 1.54), for Cmax was 0.66 (0.44, 0.98),
and for C12hr was 8.5 (5.5, 13.1). The median difference (fed-fasted) in Tmax was 7.3 (5.8, 8.8) hr. The lower range of C12hr following fed and fasted doses was similar.
Conclusions 1) Single doses of RAL up to 1600 mg are generally well tolerated. 2) RAL AUC0-∞, Cmax, and C12hr are dose proportional over the range 100 to 800 mg, indicating plasma clearance and bioavailability of RAL are independent of dose. 3) A high fat meal affected the rate, but not extent, of absorption of RAL. Data from Phase II trials suggests that the effect of food on C12hr is not clinically meaningful. 4) Twice-daily dosing without regard to food is appropriate for RAL.
Study Design
Dose Proportionality Study
-- 5-period crossover study in 20 healthy adult subjects
--In each period, subjects received a single oral dose of RAL 100, 200, 400, 800, or 1600 mg, according to a randomization scheme appropriate for a 5-period crossover design.
-- All doses were given at same time of day in each period, after overnight 8-hour fast, and with 240 mL of water.
-- Washout interval of at least 4 days between periods
Food Effect Study
-- 2-period crossover study in 20 healthy adult subjects
-- In each period, subjects received a single oral dose of RAL 400 mg either in the fasted state or after a high-fat meal, according to a randomization scheme appropriate for a 2-period crossover design
-- Washout interval of at least 4 days between periods
Methods
Analytical and Pharmacokinetic
Plasma samples were analyzed for RAL concentrations using a validated HPLC - MS/MS assay with a lower limit of quantitation of 4.5 nM.
Cmax, Tmax, and C12hr determined by inspection; half-lives by fitting a biexponential equation to each profile; AUC0-∞ calculated using linear up/log down trapezoidal method.
Statistical Analysis
--For the analysis of the effect of a high-fat meal, confidence intervals for RAL C12hr, AUC0-∞, and Cmax were constructed using a linear mixed-effects model appropriate for a 2-period crossover design.
--The assessment of Dose Proportionality (DP) was carried out using the power model, log(RAL PK)=_ + _ log (dose), with DP to be declared if the 90% CI for _ was contained within equivalence bounds.
--Equivalence bounds for _ determined such that ratio (dose-adjusted high dose / dose-adjusted low dose) was contained within (0.50, 2.00).
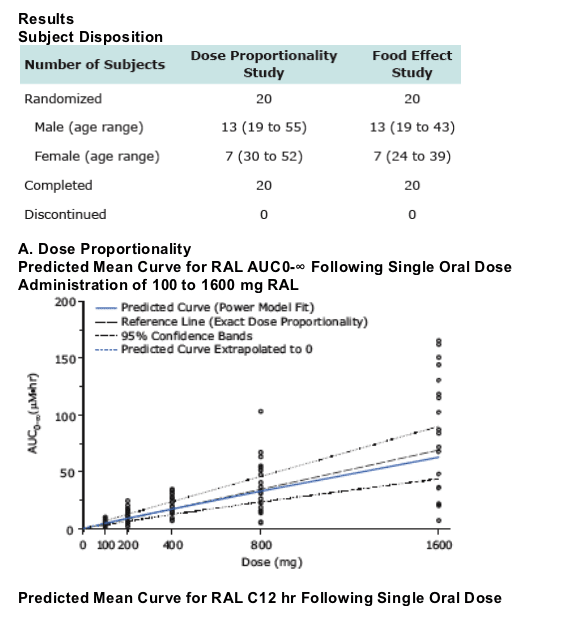
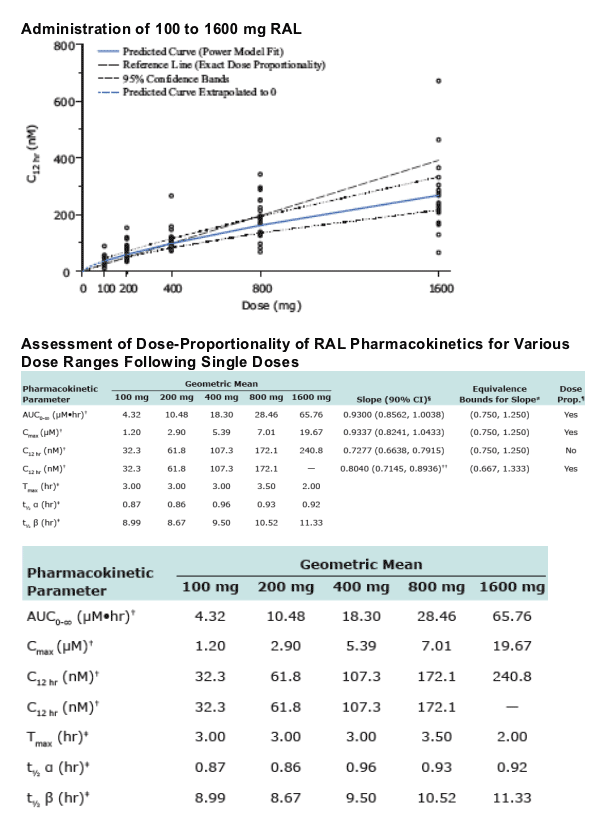
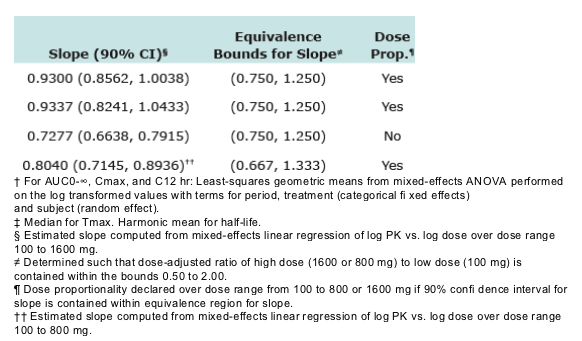
-- AUC0-∞ and Cmax of RAL were dose-proportional from 100 to 1600 mg.
-- C12hr increased proportionally from 100 to 800 mg, and slightly less than proportionally from 100 to 1600 mg.
-- Apparent terminal t1/2 of RAL was ∼9 hrs with a ∼1 hr initial phase t1/2 accounting for much of the AUC.
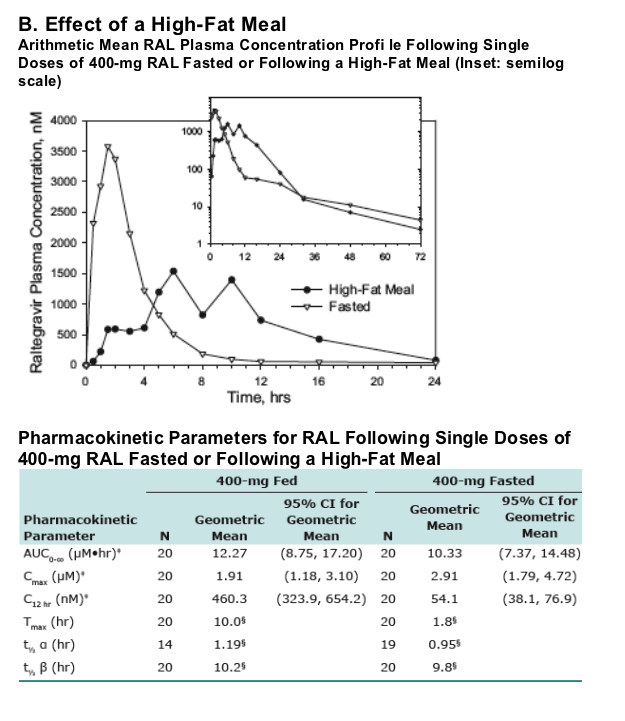

Compared to fasted dosing, dosing with a high-fat meal resulted in
-- No significant change in AUC0-∞
-- Small decrease (about 35%) in Cmax
-- Increase (about 8.5-fold) in C12hr
-- Later Tmax (about 7.3 hrs)
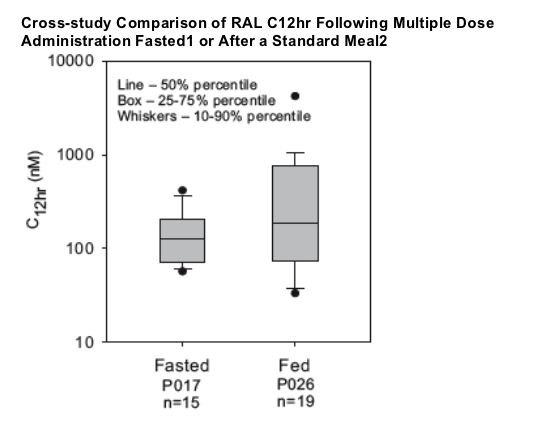
-- Effect of food may be less with multiple dosing and more moderate meal types
-- Lower range of C12hr values similar for fasted versus fed
-- Dosing with food increases variability in C12hr
-- No relationship in PK/PD analyses of Phase II studies between C12hr and effi cacy (all C12hr values were equally effi cacious)3,4
Safety
Dose Proportionality Study
-- No serious adverse events
-- No discontinuations due to adverse events
-- No laboratory adverse events
-- Most common drug-related clinical adverse events were headache and diarrhea
Food Effect Study
-- No serious adverse events
-- No discontinuations due to adverse events
-- No laboratory adverse events
-- Most common drug-related clinical adverse event was headache
Conclusions
-- Single doses of RAL up to 1600 mg are generally well tolerated.
-- RAL AUC0-∞, Cmax, and C12hr are dose proportional over the range 100 to 800 mg, indicating plasma clearance and bioavailability of RAL are independent of dose.
-- A high fat meal affected the rate, but not extent, of absorption of RAL. Data from Phase II trials suggests that the effect of food on C12hr is not clinically meaningful.
-- Twice-daily dosing without regard to food is appropriate for RAL.
|
| |
|
 |
 |
|
|