 |
 |
 |
| |
Pharmacokinetics of TMC125 in HIV-negative volunteers with mild or moderate hepatic impairment
|
| |
| |
Reported by Jules Levin
ICAAC Sept 17-20, 2007, Chicago
M Scholler-Gyure,1 TN Kakuda,2 G De Smedt,1 B Woodfall,1 C Berckmans,1 M Peeters,1 RM Hoetelmans1
1Tibotec BVBA, Mechelen, Belgium; 2Tibotec Inc., Yardley, USA
AUTHOR CONCLUSIONS
No clinically relevant difference was observed between the pharmacokinetics of TMC125 when administered in HIV-negative volunteers with and without hepatic impairment (mild or moderate).
Short-term administration of TMC125 in HIV-negative volunteers with and without hepatic impairment was generally safe and well tolerated.
TMC125 can be administered in patients with mild or moderate hepatic impairment without dose adjustment.
Abstract
Background: TMC125 (etravirine; ETR) is a next-generation nonnucleoside reverse transcriptase inhibitor (NNRTI) with potent activity against both wildtype HIV and viruses resistant to currently approved NNRTIs. TMC125 is mainly eliminated via the hepatobiliary route. This study aimed to assess the pharmacokinetics of TMC125 in HIV-negative volunteers with and without hepatic impairment.
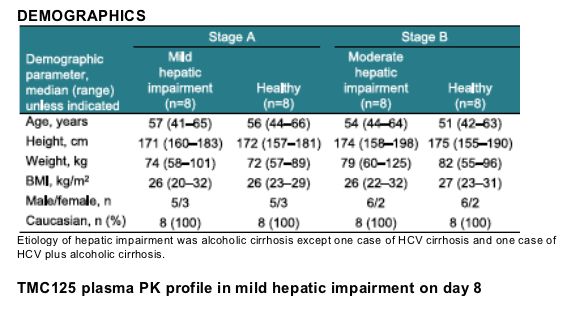
Methods: This was an open-label trial including volunteers with mild or moderate hepatic impairment (Child-Pugh1 A or B, respectively) and healthy volunteers matched for age, gender, race, and body mass index (BMI). All volunteers received TMC125 200mg twice-daily (bid) following a meal for 7 days with a morning dose on Day 8. TMC125 pharmacokinetics over 12 hours on Days 1 and 8 were determined using non-compartmental methods and analyzed by a linear mixed effect model. Safety and tolerability were assessed.
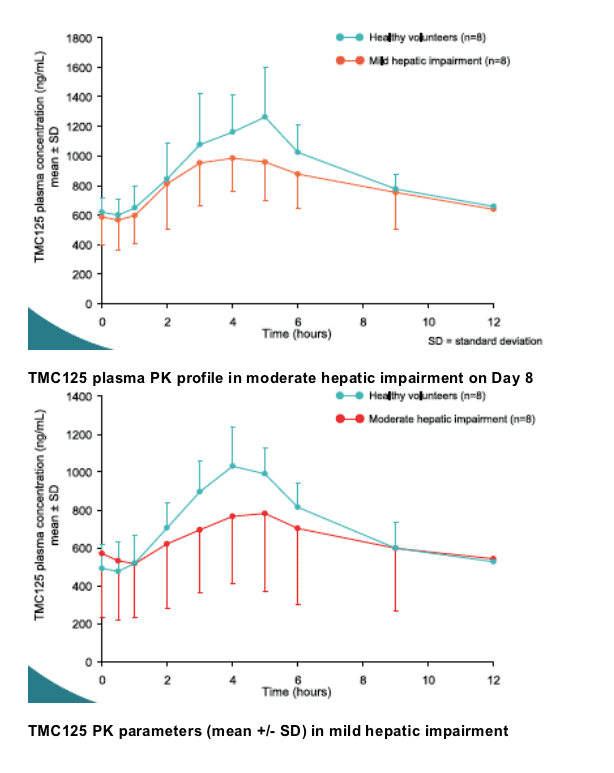
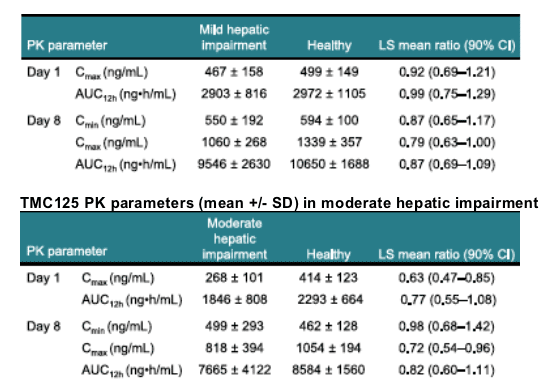
Results: Eight volunteers with mild hepatic impairment (five males; median age 57 years), eight with moderate hepatic impairment (six males; median age 54 years) and 16 matched healthy volunteers participated. Pharmacokinetic (PK) results are shown below.
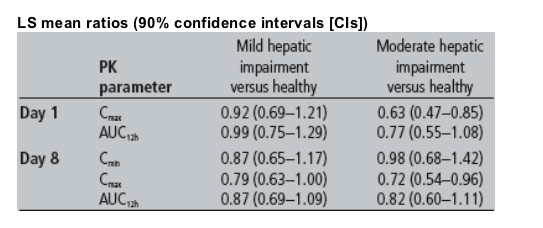
LS = least square; Cmax = maximum plasma concentration; AUC12h = area under the plasma concentration-time curve from time of administration to 12 hours after dosing; Cmin = minimum plasma concentration
All treatment emergent adverse events (AEs) were mild or moderate. The most common AEs were headache, fatigue and nausea. No clinically significant changes in laboratory parameters were observed.
Conclusions: TMC125 was generally safe and well tolerated. The systemic
exposure to TMC125 in volunteers with hepatic impairment was comparable
to that in healthy volunteers. Dose adjustment of TMC125 in patients with
mild or moderate hepatic impairment is not required.
SAFETY SUMMARY
All volunteers completed the trial.
The most frequently reported AEs for volunteers with mild hepatic impairment were nausea and fatigue (both in two volunteers with mild hepatic impairment and one healthy volunteer).
Dizziness and muscle spasms were the most common AEs for volunteers with moderate hepatic impairment (both in two volunteers compared to none in the healthy volunteers).
Headache was reported for four healthy volunteers (none of the volunteers with hepatic impairment).
No rash was reported.
All AEs reported were mild or moderate (grade 1 or 2) in severity.
No serious AEs were reported, except one case of tachyarrhythmia with cardiac failure in a volunteer with moderate hepatic impairment and pre-existing cardiomyopathy, which occurred 36 days after the last intake of TMC125.
There were no consistent or relevant changes in lab or cardiovascular safety parameters or the results of physical exams.
Grade 3 or 4 lab abnormalities were reported in one volunteers with mild hepatic impairment (hypercholesterolemia) and two volunteers with moderate hepatic impairment (low hemoglobin and hypophosphatemia in one, and increased lipase in another volunteer).
References
1. Pugh RNH, et al. Br J Surg 1973;60:646-9.
2. Vingerhoets J, et al. J Virol 2005;79:12773-82.
3. Madruga JV, et al. Lancet 2007;370:29-38.
4. Lazzarin A, et al. Lancet 2007;370:39-48.
5. Sulkowski M, et al. JAMA 2000;283:74-80.
6. Barreiro P, et al. J Inf Dis 2007;195:973-9.
|
| |
|
 |
 |
|
|