 |
 |
 |
| |
Antiretroviral Therapy at 47th ICAAC 2007 (Sept 2007): RTV-boosted PIs in treatment-naives; CCR5 antagonists; integrase inhibitors
|
| |
| |
Eric S. Daar, M.D.
Chief, Division of HIV Medicine
Harbor-UCLA Medical Center
Professor of Medicine
David Geffen School of Medicine at UCLA
Ritonavir (RTV)-boosted protease inhibitors (PIs) in treatment-naive patients
There have been several large randomized-controlled trials comparing RTV-boosted PIs in treatment-naive individuals. One of the largest such studies was the KLEAN trial that compared lopinavir/RTV (LPV/r) with fosamprenavir (FPV) plus RTV, both given twice daily with abacavir/lamivudine. Results at 48 weeks were previously published (1), and at this meeting they reported data from 96 weeks of follow-up (2). The study population was limited to those in Europe or Canada with plasma HIV-1 RNA levels less than 400 copies/mL at 48 weeks. Consequently, of the original 878 treated subjects only 196 (22%) participated in the extension phase. During this period of follow-up there was confirmation of the earlier data with very similar rates of suppression and adverse events between groups with only one virologic failure in each study arm. It was largely based upon the original 48 week data that FPV/rtv was added to the list of preferred treatment options by the DHHS guidelines for adolescents and adults and remains a viable option for first line treatment.
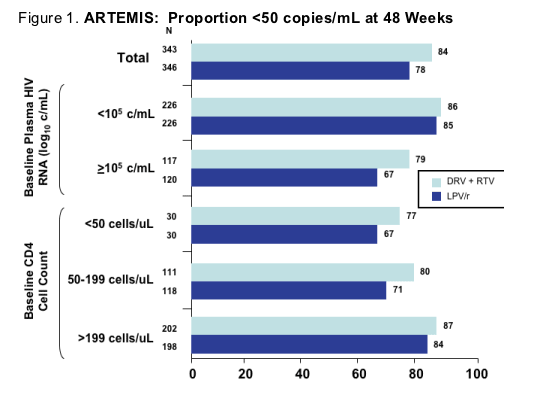
The much anticipated ARTEMIS trial was also presented at this meeting (3). This study included 689 treatment-naive individuals and was designed to compare TDF/FTC with either 800 mg of darunavir (DRV) with 100 mg RTV, both given once daily with LPV/r at either 400/100 mg twice daily or 800/200 mg once daily. LPV/r dosing varied across subjects with some taking it once daily, others twice daily and a subset starting out twice daily and then switching to once daily. There also was a mix with regards to whether patients used capsules, tablets or switched from capsules to tablets. The primary endpoint for the study was the proportion less than 50 copies/mL at 48 weeks with the objective being to show that once daily DRV plus RTV was noninferior to LPV/r. The median plasma HIV-1 RNA was between 62,000 and 70,000 copies/mL with the median CD4+ T-cells being 218 to 228 cells/uL with 36% of subjects in both arms having baseline plasma HIV-1 RNA levels of greater than 100,000 copies/mL. The overall discontinuation rate was modest for both groups, 12% for DRV and 16% for LPV/r with the difference mainly reflecting a somewhat higher rate of discontinuation for adverse events in the LPV/r arm compared to the DRV plus RTV arm (3 vs. 7%, respectively) and for virologic failure (<1 vs. 2%, respectively).
For the primary endpoint they did show DRV plus RTV to be non-inferior to LPV/r for the proportion less than 50 copies/mL at 48 weeks, 84 vs. 78%, respectively (p<0.001). Various analyses suggested that the differences were most prominent amongst those with higher baseline viral loads, lower baseline CD4+ T-cells and in those who received once daily LPV/r, although for the latter there were only 52 subjects included in the analysis (Figure 1). There was no difference in the change from baseline CD4+ T-cells at 48 weeks between the groups. Viral failure with a plasma HIV-1 RNA greater than 50 copies/mL was higher for those on LPV/r (14%) than DRV plus RTV (10%) arms with drug resistance mutations rare in both groups with one LPV/r patient having PI mutations and two with the M184V, and one DRV plus RTV patient having a M184V mutation. Finally, the main difference in the frequency of grade 2-4 adverse events was the presence of more diarrhea in the LPV/r group (10 vs. 4%, P<0.05) and a nonsignificantly higher frequency of rash in the DRV plus RTV group (3 vs. 1%). Grade 2-4 cholesterol and triglyceride increases were more common amongst those receiving LPV/r; however there was no apparent difference in Total cholesterol/HDL ratio.
CCR5 antagonists
The FDA recently approved maraviroc, the first CCR5 antagonist for treatment-experienced patients. Previous reports of the pivotal phase III trials MOTIVATE 1 and 2 reported safety and efficacy at 24 weeks (4, 5). The 48 week data from MOTIVATE 1 was presented at this meeting demonstrating sustained viral suppression at a mean of 1.82 log10 copies/mL from baseline in those receiving twice daily maraviroc with approximately 47% maintaining plasma HIV-1 RNA levels of less than 50 copies/mL (6). Results were very similar across the groups receiving once and twice daily maraviroc, both having better virologic responses than those in the placebo arm where 16% of subjects maintained less than 50 copies/mL after 48 weeks. The drug continues to appear safe with little difference in the frequency of adverse or Category C events between the maraviroc and control groups. Notably there were fewer lymphoma or Kaposi's sarcoma cases in the maraviroc than placebo group. Data from this study was reassuring with regards to the durability of viral suppression with this class of drugs as well as its safety out to one year. Additional reassurance came from follow-up data on select individuals treated with another CCR5 antagonists in the latter stages of development, vicriviroc. Previous data has shown the efficacy of this drug along with optimized background regimen in treatment-experienced patients for up to 48 weeks (7, 8). At this meeting investigators described data from an open-label trial which followed select individuals in A5211 as well as some who did not enroll but were given vicriviroc. As in the case of the MOTIVATE studies all subjects were required to have no detectable CXCR4-utilizing virus at the time of screening and in this case optimized-background regimen had to include RTV along with vicriviroc given at 15 mg once per day. Additional adverse events in this group were rare with median follow-up being 504 days. Median change from baseline in plasma HIV-1 RNA in those on vicriviroc remained greater than 2 log10 copies/mL with more than 60% maintained plasma HIV-1 RNA levels below 50 copies/mL (9). Needless to say this represents a select group of individuals that remained on treatment but still shows that for these individuals viral suppression can be durable. Even more important in a study like this is the collection of additional safety data. While many experienced adverse events, as would be predicted in such a highly treatment-experienced populations, most were grade 1-2 with the only grade 3 event occurring in more than 1% being fatigue (3%). Furthermore, there were only two additional malignancies, both being skin cancers, neither of which were thought to be related to treatment.
Another potential concern in patients taking a CCR5 antagonist is the emergence of CXCR4-utilizing viruses. This is worrisome both as a mechanism by which treatment failure can occur as well as because of concerns that these viruses may be associated with increased risk of disease progression (10). Importantly, the latter is based upon an association between the presence of these viruses and disease progression in natural history cohorts and not direct evidence that it is actually the emergence of this virus population that drives disease progression. Nevertheless, the natural history data has continued to prove quite compelling. In fact, at this meeting there was a report of 314 treatment-naive patients with greater than 450 CD4+ T-cells/uL followed in the CPCRA Long Term Monitoring Protocol (11). A tropism assessment was made at baseline using the Monogram Biosciences TrofileTM assay, the test currently licensed for this purpose. They assessed the relationship between the presence of CXCR4-utilzing virus and progression to a CD4+ T-cell counts less than 350 cells/uL, the initiation of therapy or the development of clinical AIDS. The investigators found that 10.2% had dual/mixed virus at baseline and that even after controlling for baseline plasma HIV-1 RNA and CD4+ T-cells, compared to those with only R5 virus these subjects were at an increased risk (HR 2.14, p=0.003) for progression to this composite endpoint. This data is consistent with previous work with somewhat different populations but like all other studies in no way answers the question as to whether the presence of CXCR4-utilizing virus is the cause or effect of disease progression. Nevertheless, it continues to be a high priority in studies of CCR5 antagonists to track the frequency in which CXCR4-utlizing viruses emerge.
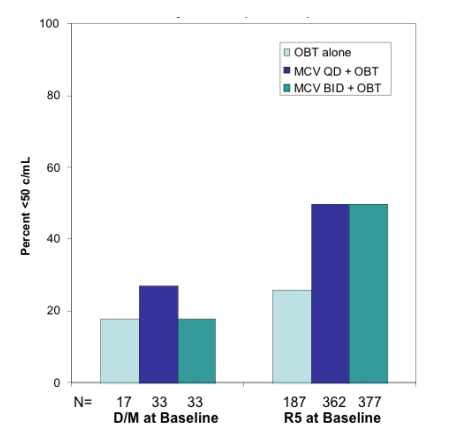
The frequency and significance of emerging dual/mixed or X4 virus was reported from those followed for up to three years after completion of the 48 weeks A5211 study (8). They found that 6 of 79 had experienced the emergence of CXCR4-utilizing virus with little evidence that this was associated with CD4+ T-cell decline. Similarly, further analyses were reported on the frequency and significance of emerging CXCR4-utlizing viruses during MOTIVATE 1 and 2 (12). As previously reported they found that 8% of patients that screened in based upon having undetectable dual/mixed or X4 virus were found to have these viruses present at entry, prior to starting treatment. It is felt that this is a result of low levels of these viruses present at the time of screening but not detected by the current phenotypic assay. In fact, these observations have prompted attempts to improve the current assay (as discussed below) (13, 14). Further analyses from this study demonstrated that those that had dual/mixed or X4 virus at entry tended to have lower CD4+ T-cells and higher plasma HIV-1 RNA levels at the time of screening and to have an attenuated response to treatment with maraviroc (Figure 2). Additional studies focused upon the frequency of dual/mixed and X4 virus being present at the time of virologic failure. The investigators showed that amongst the patients experiencing virologic failure that also had tropism data available, 64% had CXCR4-utlizing virus detected compared to only 5% of those failing in the placebo group. When analyzed by time to failure it was found that those failing within the first 70 days were more likely to fail with dual/mixed or X4 virus than those failing after this period of time. These results along with others showing that the CXCR4-utlizing viral population identified at failure appears to be from a pre-existing population suggests that many failures result from selection for minority dual/mixed or X4 population missed with standard tropism testing. In fact, it appears that this viral population is likely to be less fit in vivo since upon stopping maraviroc many of these viruses returned to undetectable levels in the blood. While these studies support the need for more sensitive tropism assays it is clear that the majority of patients that started a good regimen achieved full suppression and did not fail with CXCR4-utlizing virus. Moreover, when virologic failure occurred with CXCR4-using virus it did not appear to be associated with adverse consequences, at least in the relative short-term. Further follow-up will be needed to define the long-term effects of this phenomenon.
In attempts to improve outcomes with CCR5 antagonists the group at Monogram Biosciences has worked to improve the performance of the current tropism assay. They report that the current test is 100% sensitive to detect minor variants present at 10% of the viral population and 83% sensitive for those present at 5%. Although specifics regarding the refinements made in the assay were not fully described they did present data on the ability of the modified assay to detect minority species (13). These studies used mixtures of cloned viruses as well as longitudinal plasma samples from patients who experienced the emergence of CXCR4-utilizing virus. Using mixtures of cloned viruses they showed the sensitivity to detect minority species increased 3-10-fold and ranged from 0.1 to 0.3%. Consistent with this enhanced sensitivity they were able to detect the presence of this viral population earlier in the course of follow-up using the longitudinally collected plasma samples. Investigators from this group also attempted to use sequencing methods to identify CXCR4-using virus (14). These experiments demonstrated the complexity of doing this in light of the variability in the envelop gene, as well as the relative insensitivity of the method. This is consistent with recently reported data from other independent groups (15).
A novel agent that blocks the use of the CCR5 chemokine receptor is a humanized CCR5 monoclonal antibody PRO 140 that targets this receptor and prevents R5 virus triggering. It is stated that it binds an extracellular portion of the receptor, competitively inhibits HIV infection and does not interfere with normal activity of CCR5. Preliminary data at this meeting showed the pharmacokinetics of this drug as well as its profound anti-HIV activity in vivo (16). The monoclonal antibody administered as an intravenous infusion was given to HIV-infected individuals that were found to only have detectable R5 virus. A total of 39 subjects were randomized to receive 0, 0.5, 2.0 or 5.0 mg/kg of this agent as a single infusion with 59 days of follow-up. Mean viral load reduction was 1.7 log10 copies/mL at 10 days with a clear dose response which was consistent with the pharmacokinetics. Furthermore, viral load did not return to baseline until approximately 30 days after the infusion, which is likely related to the prolonged half life (4 days) and perhaps persistent binding of the monoclonal antibody to CCR5. Further work demonstrated that this agent also has activity against HIV-2 (17). Based upon this study and others it is planned that this novel therapeutic will advance into phase II trials with attempts to administer it subcutaneously.
Integrase Inhibitors
The 48 week data was presented from a phase II study of raltegravir in treatment-experienced patients (18). This was a dose finding study that complements the limited follow-up available from the BENCHMRK 1 and 2 studies (19, 20). The current study randomized 178 patients to an optimized background regimen with either 200, 400 or 600 mg of raltegravir given twice daily. The activity of the optimized background in these regimens was limited due to high level of resistance and the lack of availability of tipranavir or darunavir at the time the study began. In fact, it was reported that approximately 50% of subjects had no phenotypically active drugs included in the regimen. Despite this 64-71% of patients had less than 400 copies/mL and 46 to 64% less than 50 copies/mL of plasma HIV-1 RNA at week 48 of follow-up. Moreover, there was no apparent difference in adverse events between the raltegravir and placebo arms of the study. Of the 38 patients failing raltegravir 35 (92%) had integrase mutations, including the signature changes at position N155 or Q148, with most having one of these mutations along with others. This does represent the longest reported follow-up experience with this exciting agent and supports the sustained efficacy and safety of this drug in treatment-experienced patients.
The other integrase inhibitor in the later stages of clinical development is elvitegravir (21). Unlike raltegravir this drug is a substrate for cytochrome P450 and is given once daily with ritonavir. This was a follow-up analysis of data from the Phase II study that randomized treatment-experienced patients to optimized background NRTIs with or without enfuvirtide and either elvitegravir or a RTV-boosted PI of the investigators choice. During the course of the study drug-drug interaction data with PIs became available and if felt appropriate patients were allowed to add darunavir or tipranavir to their elvitegravir-containing regimen, which occurred in two subjects prior to week 16. The objective of the study was to show that elvitegravir was noninferior to the PI-containing regimen for the primary endpoint of time-weighted average change from baseline in HIV-1 RNA at 24 weeks (DAVG24). The study enrolled 278 patient with at least one primary PI mutation and randomized them to comparator PI, which included darunavir in approximately 50% and tipranavir in 25% of cases, versus elvitegravir with RTV at either 20, 50 or 125 mg per day. The 20 mg arm was prematurely discontinued due to high rate of virologic failure. This analysis focused an the 125 mg elvitegravir group and showed that the study population was similar to that in the PI arm except for slightly higher first time use of enfuvirtide, 26 vs. 19%, respectively. Overall virologic responses were significantly better for this elvitegravir group compared to the control arm, an observation that was also true for those receiving enfuvirtide for the first time. Consistent with other studies, both the controls and elvitegravir groups had better viral suppression if enfuvirtide was included for the first time in their optimized background regimen. In fact, although the numbers were small, 14 of 19 (74%) of subjects who received elvitegravir at 125 mg per day along with enfuvirtide for the first time achieved plasma HIV-1 RNA levels of less than 50 copies/mL. Similarly, analyses demonstrated incremental increases in viral load reduction as increasing numbers of active drugs were included in the optimized background with the apparent rapid loss of virologic response seen in those without active drugs included with elvitegravir.
These studies demonstrate the clear activity of this new class of drugs in treatment-experienced patients. They further emphasize that it will be critical for clinicians to use them with caution to assure that the background regimen is sufficient to minimize the risk of viral rebound and emergence of resistant virus, which has already been shown to often be associated with cross resistance between raltegravir and elvitegravir.
REFERENCES
1. Eron J Jr, Yeni P, Gathe J Jr, et al. The KLEAN study of fosamprenavir-ritonavir versus lopinavir-ritonavir, each in combination with abacavir-lamivudine, for initial treatment of HIV infection over 48 weeks: a randomised non-inferiority trial. Lancet 2006;368:476-82.
2. Pulido F, Baril JG, Staszewski S, et al. Long-term efficacy and safety of fosamprenavir + ritonavir (FPV/r) vs. lopinavir/ritonavir (LPV/r) over 96 weeks. 47th ICAAC 2007, Chicago, IL, Abstract H-361.
3. DeJesus E, Ortiz R, Khanlou H, et al. Efficacy and safety of darunavir/ritonavir versus lopinavir/ritonavir in ARV treatment-naive HIV-1-infected patients at week 48: ARTEMIS. 47th ICAAC 2007, Chicago, IL, Abstract H-718b.
4. Lalezari J, Goodrich J, DeJesus E, et al. Efficacy and safety of maraviroc plus optimized background therapy in viremic ART-experienced patients infected with CCR5-tropic HIV-1: 24-week results of a phase 2b/3 study in the US and Canada. 14th Conference on Retroviruses and Opportunistic Infections 2007, Los Angeles, California, Abstract #104bLB.
5. Nelson M, Fatkenheuer G, Konourina I, et al. Efficacy and safety of maraviroc plus optimized background therapy in viremic, ART-experienced patients infected with CCR5-tropic HIV-1 in Europe, Australia, and North America: 24-week results. 14th Conference on Retroviruses and Opportunistic Infections 2007, Los Angeles, California, Abstract #104aLB.
6. Lalezari J, Goodrich J, DeJesus E, et al. Efficacy and safety of maraviroc in antiretroviral-experienced patients infected with CCR5-tropic HIV-1: 48-week results of MOTIVATE 1. 47th ICAAC 2007, Chicago, IL, Abstract ??.
7. Gulick RM, Zu S, Flexner C, et al. Phase 2 Study of the safety and efficacy of vicriviroc, a CCR5 inhibitor, in HIV-1-infected, treatment-experienced patients: AIDS Clinical Trials Group 5211. Journal of Infectious Diseases. 2007;196:304-12.
8. Gulick R, Zu S, Flexner C, et al. ACTG 5211: Phase 2 Study of the safety and efficacy of vicriviroc (VCV) in HIV+ treatment-experienced subjects: 48-week results. 4th International AIDS Society Conference on HIV Pathogenesis, Treatment and Prevention 2007; Sydney, Australia. Abstract TUAB102.
9. Gulick R, Haas D, Collier AC, et al. Two-year follow-up of treatment-experienced patients on vicriviroc (VCV). 47th ICAAC 2007, Chicago, IL, Abstract H-1030.
10. Daar ES, Kesler KL, Petropoulos CJ, et al. Baseline HIV-1 co-receptor tropism predicts disease progression. Clin Infect Dis 2007;45:643-9.
11. Goetz MB, Leduc R, Kostman JR, et al. Prediction of disease progression by HIV co-receptor tropism (CRT) in persons (P) with untreated chronic HIV infection. 47th ICAAC 2007, Chicago, IL, Abstract H-1027.
12. Van der Ryst E, Westby M. Changes in HIV-1 co-receptor tropism for patients participating in the maraviroc Motivate 1 and 2 clinical trials. 47th ICAAC 2007, Chicago, IL, Abstract H-715.
13. Reeves JD, Han D, Liu Y, et al. Enhancements to the Trofile HIV coreceptor tropism assay enable reliable detection of CXCR4-using subpopulations at less than 1%. 47th ICAAC 2007, Chicago, IL, Abstract H-1026.
14. Stawiski E, Whitcomb J, Coakley E, et al. Co-receptor tropism predictions based on V3 loop sequence in antiretroviral-experienced patients are specific but insensitive for the detection of CXCR4-using variants. 47th ICAAC 2007, Chicago, IL, Abstract H-1028.
15. Low AJ, Dong W, Chan D, et al. Current V3 genotyping algorithms are inadequate for predicting X4 co-receptor usage in clinical isolates. AIDS 2007;21:F17-F24
16. Jacobson JM, Thompson M, Saag MS, et al. Antiretroviral activity and pharmacodynamics of PRO 140, a CCR5 monoclonal antibody, in HIV-infected individuals. 47th ICAAC 2007, Chicago, IL, Abstract H-716.
17. Ketas TJ, Maddon PJ, Olson WC. Comparative susceptibility of HIV-1 and HIV-2 to the humanized CCR5 monoclonal antibody PRO 140. 47th ICAAC 2007, Chicago, IL, Abstract H-1029.
18. Grinsztejn B, Nguyen B, Katlama C, et al. 48 week efficacy and safety of MK-0518, a novel HIV-1 integrase inhibitor, in patients with triple-class resistant virus. 47th ICAAC 2007, Chicago, IL, Abstract H-713.
19. Cooper D, Gatell J, Rockstroh J, et al. Results of BENCHMRK-1, a phase III study evaluating the efficacy and safety of MK-0518, a novel HIV-1 integrase inhibitor, in patients with triple-class resistant virus 14th Conference on Retroviruses and Opportunistic Infections 2007, Los Angeles, CA, Abstract #105aLB.
20. Steigbigel R, Kumar P, Eron J, et al. Results of BENCHMRK-2, a phase III study evaluating the efficacy and safety of MK-0518, a novel HIV-1 integrase inhibitor, in patients with triple-class resistant virus 14th Conference on Retroviruses and Opportunistic Infections 2007, Los Angeles, CA, Abstract #105bLB.
21. Zolopa AR, Lampiris H, Blick G, et al. The HIV integrase inhibitor elvitegravir (EVG/r) has potent and durable activity in treatment-experienced patients with active optimized background therapy (OBT). 47th ICAAC 2007, Chicago, IL, Abstract H-714.
|
| |
|
 |
 |
|
|