 |
 |
 |
| |
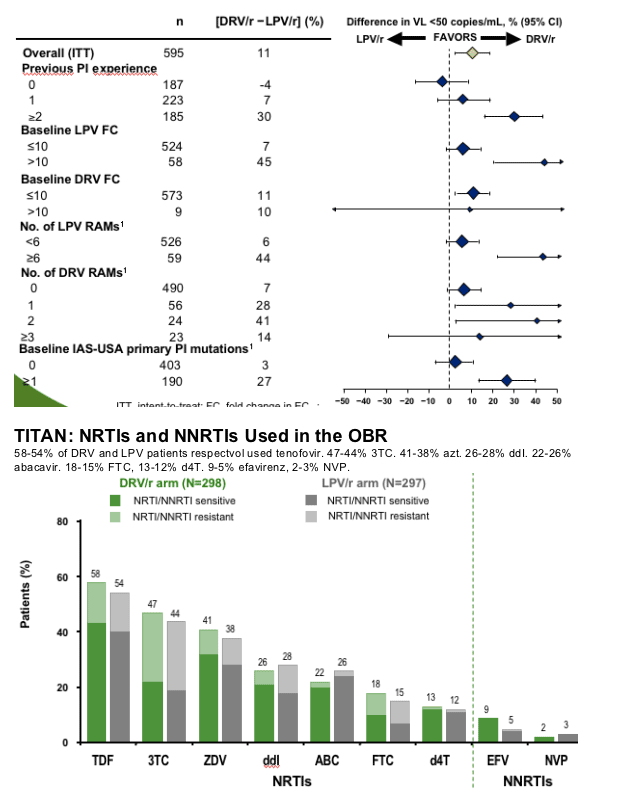
Influence of Baseline Factors on Virologic Response to Darunavir/Ritonavir (DRV/r) vs Lopinavir/r (LPV/r): Week 48 Outcome in TITAN
|
| |
| |
Reported by Jules Levin
IDSA, San Diego, CA, Oct 7, 2007
W. David Hardy, MD1; Daniel Berger, MD2; Els De Paepe, MSc3; Sandra De Meyer, PhD3; David Moriarty, PhD4; Joseph Mrus, MD5; Sabrina Spinosa-Guzman, MD3
1Cedars-Sinai Medical Center, Los Angeles, CA, USA; 2Northstar Medical Center, Chicago, IL, USA; 3Tibotec BVBA, Mechelen, Belgium; 4Tibotec Inc., Yardley, PA, USA; and 5Tibotec Therapeutics, Bridgewater, NJ, USA
TITAN = TMC114/r In Treatment-experienced pAtients Naive to lopinavir
AUTHOR CONCLUSIONS
In treatment-experienced, LPV-naive patients:
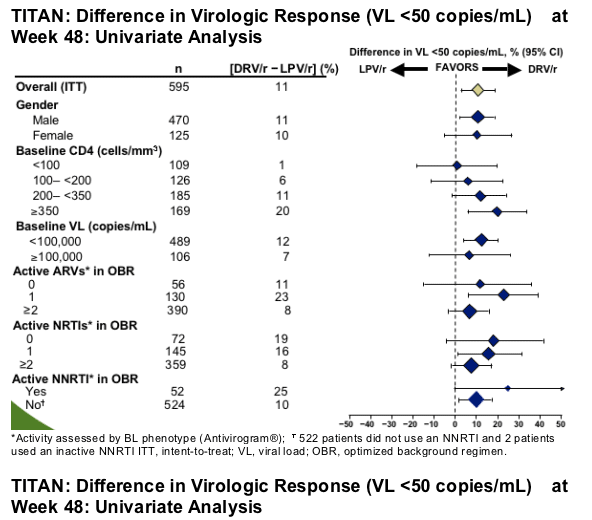
The difference in virologic response (VL <50 copies/mL) favoring DRV/r was generally consistent across subgroups
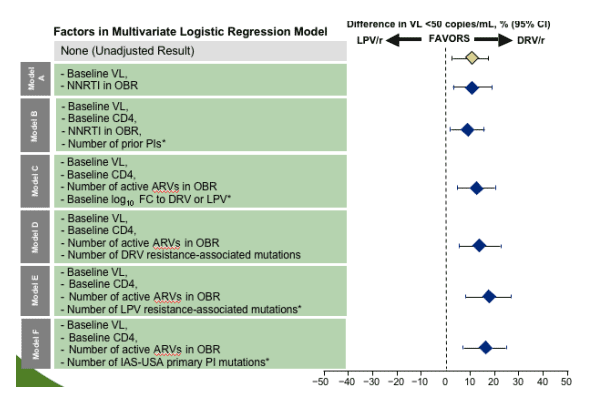
Multivariate analyses demonstrated that the difference in response favoring DRV/r was maintained after adjusting for baseline characteristics including VL and CD4, activity of OBR, PI resistance, and prior PI experience
Overall, and in patients phenotypically sensitive to both PIs at baseline (FC ≦10), the presence of 1 or more IAS-USA primary PI mutations affected the response to LPV/r but not DRV/r
--As the number of IAS-USA primary PI mutations increased, the difference in virologic response favoring DRV/r increased
TITAN is an ongoing, randomised, controlled trial. It is an international 96-week study, with primary analysis at Week 48.
Patients were screened for eligibility and had to be:
--treatment experienced but naive to lopinavir
--have documented HIV infection
--have a viral load of greater than one thousand copies per mL
--to have been on stable HAART therapy or off treatment for 12 weeks or more.
A total of 595 patients were randomised to treatment of either, darunavir 600mg with 100mg ritonavir or lopinavir 400mg, again with the same low dose of ritonavir.
Both treatments were administered twice daily, and all patients also received an optimised background regimen. This consisted of at least 2 to 3 antiretrovirals from the NRTI and NNRTI classes.
LPV/r patients were allowed to switch to new formulation upon its approval by the regulatory authorities. At the time of this analysis 18% had switched top the new formulation.
Background
Patients co-infected with chronic hepatitis B or C were allowed to enter the trial if their condition was clinically stable and they were not expected to require hepatitis treatment during the study period.
Randomisation was performed using a centralised system to ensure balance across treatments groups and across two stratification factors (the use of an NNRTI in the OBR and screening plasma viral load of <50,000 or ≥50,000 copies/mL).
Darunavir was administered as 300-mg tablets, ritonavir as 100-mg capsules and lopinavir-ritonavir as 133.3/33.3-mg capsules. During the study, a new formulation of lopinavir-ritonavir (200/50-mg tablet) became available. The study protocol was amended to allow patients randomised to lopinavir-ritonavir to switch to the new formulation as soon as it was approved by local regulatory authorities; subsequently, all patients on this arm would be switched to the new formulation.
Patients who experienced virological failure (defined as plasma viral load >400 copies by week 16 or beyond) or a treatment-related grade 4 AE or a confirmed grade 4 laboratory abnormality were eligible to participate in the planned rollover phase. Results of the 48-week primary analysis are reported here.
TITAN (TMC114-C214): Study Design
Phase III randomized, controlled trial with primary analysis at Week 48
--785 patients screened, 595 randomized and treated
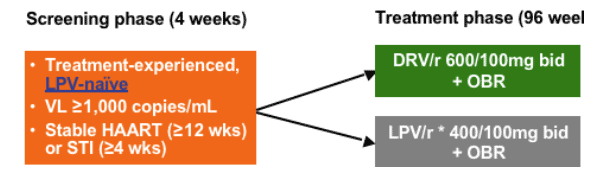
All patients received optimized background therapy (OBR)
--Two to three ARVs from approved NRTI and/or NNRTI classes
--Enfuvirtide disallowed
Stratification factors: Baseline VL and use of NNRTI in OBR
*LPV/r patients were allowed to switch to new formulation upon its approval by the regulatory authorities
Summary of Previous Findings from TITAN
Overall, in treatment-experienced, LPV-naive patients:
DRV/r was virologically non-inferior and superior to LPV/r (VL <400 and VL<50 copies/mL)
Twice as many patients receiving LPV/r experienced virologic failure (VF) compared with patients receiving DRV/r
Following VF and compared with LPV/r, DRV/r-based therapy was associated with lower rates of development of
--Primary PI mutations or NRTI RAMs
--Phenotypic resistance to the PI or NRTI(s) in the study regimen
DRV/r was safe and well tolerated, with a lower rate of diarrhea and lower increases in triglycerides than LPV/r, and a higher rate of rash
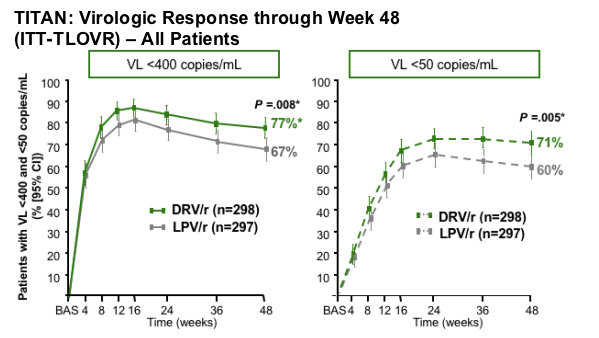
DRV/r was non-inferior and superior to LPV/r
*P value for superiority derived from logistic regression model including treatment and stratification factors: baseline log10 VL and use of NNRTI in the optimized background regimen
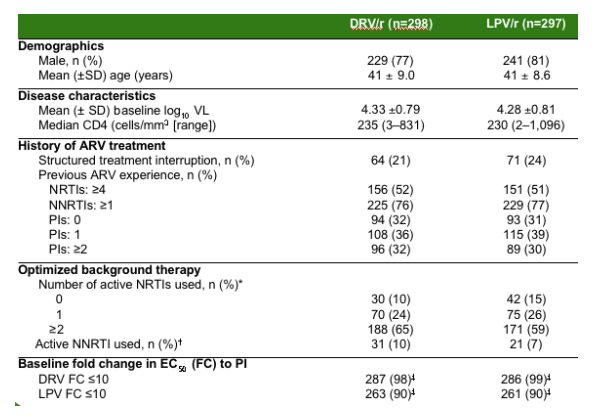
TITAN: Baseline Characteristics
--The demographic, disease characteristics and treatment history were well balanced across both treatment arms: in the table below.
Previous ART experience: NRTIs 4 or more used; 52 & 51%. NNRTIs 1 or more used; 76 & 77%. 0 PIs; 32 & 31%. 1 PI; 36% DRV, 39% LPV. 2 OR MORE PIs; 32% DRV, 30% LPV.
Optimized background therapy:
Number of active NRTIs used:
0, 10% DRV, 15% LPV;
1, 24% DRV, 26% LPV;
2 or more: 65% DRV, 59% LPV;
active NNRTI used: 10% DRV, 7% LPV.
Baseline fold change in EC50 to PI:
DRV FC 10 or less: 98% in DRV group, 99% in LPV group;
LPV/r fold-change 10 or less: 90% in DRV, 90% in LPV.
--Patients had an average age of 41 years. Mean plasma viral load was 4.3 log10 copies/mL and median CD4 cell count was 232 cells/mm3
--The population of patients in this trial were representative of those we see failing in clinical practice
--Baseline resistance characteristics were also well balanced between treatment arms.
Background
The mean duration of study treatment was similar in both groups (53•5 weeks for darunavir-ritonavir and 51•5 weeks for lopinavir-ritonavir).
Activity of ARVs assessed by baseline phenotype (Antivirogram); 576 of 582 patients with phenotype data used ≥1 NRTI in the OBR; 54 patients used an NNRTI, irrespective of activity; 582 (DRV/r=292; LPV/r=290) patients had baseline phenotype available.
RESULTS
NRTI use was generally well-balanced between treatment groups and consistent with current practice
-- NRTI/NNRTI activity assessed by baseline phenotype (Antivirogram)
TITAN: Impact of IAS-USA Primary PI Mutations* at Baseline on VL <50 Copies/mL at Week 48
The table below contains the following data and these tables will be posted along with this full report on NATAP website tomorrow. For 'all patients' with 0 mutations on drv 70% had and 67% on lpv had <50 c/ml.
For 'patients with prior PI experience 69% on drv and 62% on lpv had < 50 c/ml.
For 'all patients' with 1, 2, or 3 or more PI mutations the differences in % <50 c/ml:
- 79% DRv, 58% LPV - patients with 1 PI mutation.
- 68% DRV, 44% LPV - patients with 2 PI mutations
- 69& DRV, 33% LPV - patients with 3 or more PI mutations
For 'patients with Prior PI Experience:
-- 78% vs 53% for patients with 1 PI mutation
-- 68% vs 42% for patients with 2 PI mutations
-- 68% vs 31% for patients with 3 or more mutations
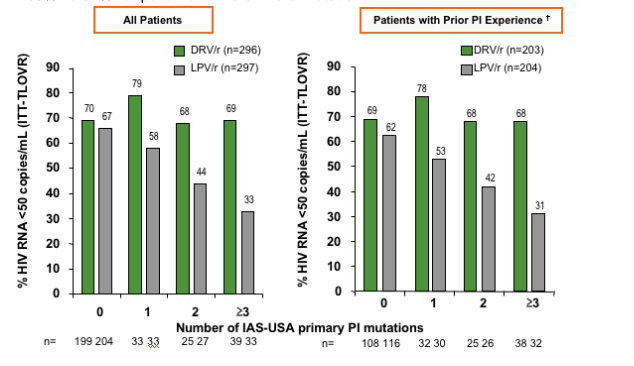
Patients with non-missing genotype data; Excludes patients with missing LPV FC at baseline.
*D30N, V32I, L33F, M46I/L, I47A/V, G48V, I50V/L, I54L/M, L76V, V82A/F/L/S/T, I84V, N88S, or L90M1
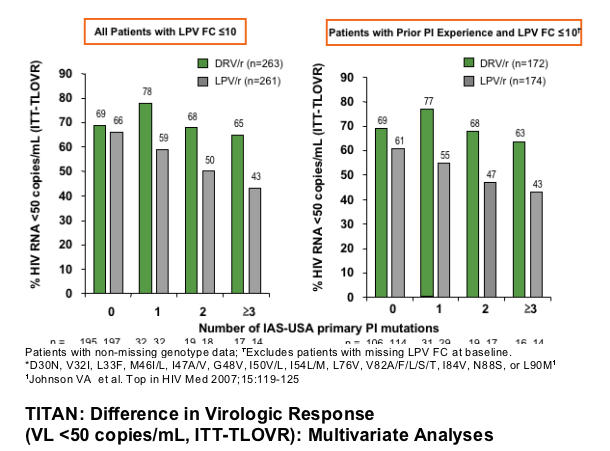
TITAN: Impact of IAS-USA Primary PI Mutations* at Baseline on VL <50 Copies/mL at Week 48-
Patients with LPV FC ≦10 at BL
For all patients with LPV FC 10 or less 69% taking DRV and 66% taking LPV achieved <50 c/ml. The results reported for patients with 1 or more PI mutations are similar to that above. For patients with prior PI experience and LPVFC 10 or less 69% taking DRV and 61% taking LPV achieved <50 c/ml and for patients with 1 or more PI mutations again results are similar to that in above table.
|
| |
|
 |
 |
|
|