 |
 |
 |
| |
In vivo Emergence of HIV-1 Resistance to the CCR5 Antagonist Vicriviroc: Findings from ACTG 5211
|
| |
| |
Reported by Jules Levin
16th Intl HIV Drug resistance Workshop, June 12-16, 2007, Barbados
Athe M.N. Tsibris1,2, Roy M. Gulick3, Zhaohui Su4, Michael D. Hughes4, Charles Flexner5, Timothy Wilkin3, Robert Gross6, Martin Hirsch1,2, Paul R. Skolnik7, Eoin Coakley8, Wayne L. Greaves9, and Daniel R. Kuritzkes2,10
1Massachusetts General Hospital, Boston, MA, USA; 2Harvard Medical School, Boston, MA, USA; 3Weill Medical College of Cornell University, New York, NY, USA; 4Harvard School of Public Health, Boston, MA, USA; 5Johns Hopkins University, Baltimore, MD, USA; 6The University of Pennsylvania, Philadelphia, PA, USA; 7Boston Medical Center, Boston, MA, USA; 8Monogram Biosciences, South San Francisco, CA, USA; 9Schering-Plough Research Institute, Kennilworth, NJ, USA; 10Brigham and Women's Hospital, Boston, MA, USA
AUTHOR CONCLUSIONS
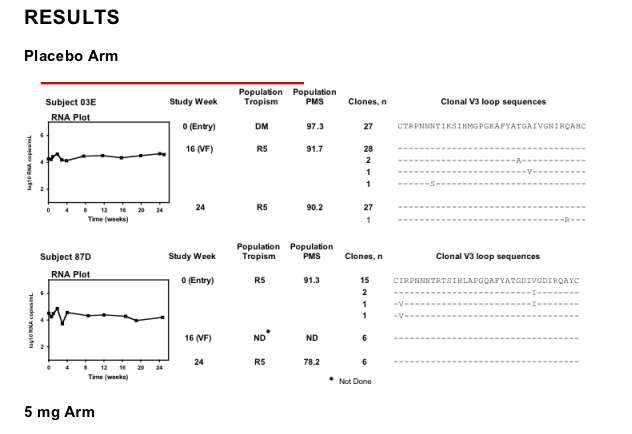
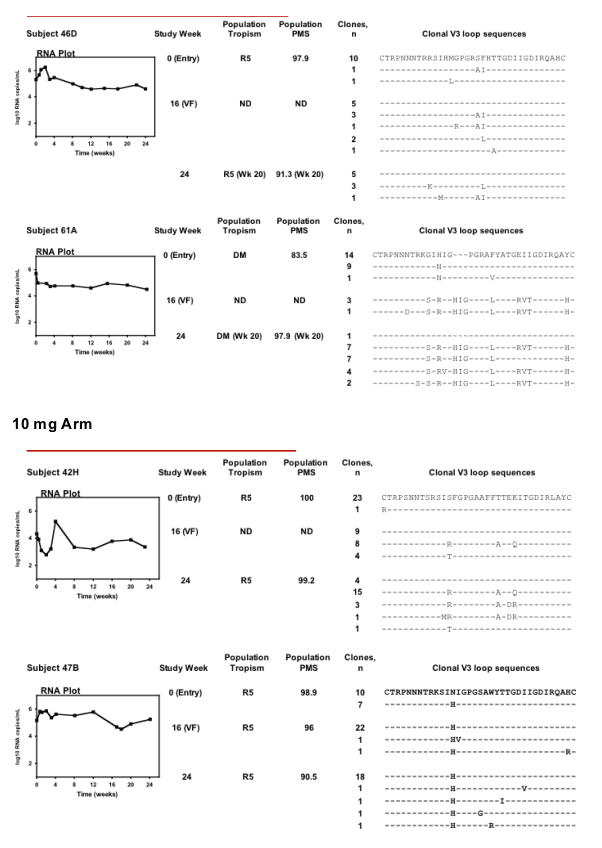
Protocol-defined virologic failure in six out of seven treatment-experienced subjects receiving a VCV-containing regimen was not associated with phenotypic evidence of VCV resistance. V3 loop sequence changes occurred in these subjects, but neither consistent nor stereotypic amino acid substitutions were observed.
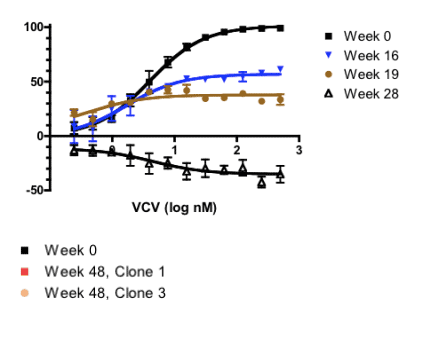
In one of these subjects, however, virologic failure was associated with phenotypic evidence of VCV resistance and the sequential accumulation of mutations in the V3 loop stem. Viruses with subject-derived env clones demonstrated a progressive plateau in VCV susceptibility, suggesting improved efficiency of viral entry using VCV-bound CCR5. By week 28, enhanced viral growth in the presence of VCV further suggests that HIV-1 had adapted preferentially to enter target cells more efficiently using VCV-bound CCR5 than native CCR5. Discontinuation of VCV was associated with a return toward genotypically wild-type, VCV-susceptible virus at week 48, suggesting a fitness advantage of wild-type over VCV-resistant virus.
ABSTRACT
Background: Few data are available regarding in vivo emergence of resistance to the CCR5 antagonist vicriviroc (VCV). We determined co-receptor usage, VCV susceptibility, and env sequences of HIV-1 from subjects with virologic failure (VF) in ACTG A5211, a phase IIb trial of VCV.
Methods: Samples from eight subjects with VF at week 16 were randomly selected for clonal sequence analysis (two subjects each from the placebo, 5-, 10-, and 15-mg VCV arms, respectively). VCV susceptibility (Entry Susceptibility assay), co-receptor usage (Trofile), and env cloning and sequencing were performed on samples obtained at entry, confirmation of virologic failure and week 24. Sequencing was performed on full-length molecular clones of env obtained from plasma virus by RT-PCR. Env sequencing was also performed on samples from one additional subject from the 10-mg arm after susceptibility testing showed progressive loss of inhibition by VCV.
Results: No consistent increase in VCV IC50 was observed in samples obtained from the 8 randomly selected subjects through week 24 (maximum increase compared to control = 2.83-fold). HIV-1 variants from four of four subjects enrolled in the VCV 5- and 10-mg arms had changes in the V3 loop stem that became fixed in the population following VF; such changes were not found in samples from subjects in the placebo or 15-mg arms. Observed sequence changes involved different amino acid positions and substitutions in samples from each subject. Serial samples from the subject with phenotypic evidence of resistance showed emergence of multiple changes in the V3 loop sequence. Tropism assays on samples from these same time points showed a modest capacity to infect CXCR4-expressing cells, although the virus remained predominantly R5. A sample obtained 5 months after stopping VCV showed R5 virus with return towards phenotypic susceptibility and baseline V3 loop sequences.
Conclusions: VF in subjects receiving a VCV-containing regimen was associated with changes in the V3 loop in samples from 5 of 9 subjects tested. In the subject with phenotypic evidence of VCV resistance, discontinuation of VCV was associated with return toward genotypically wild-type, VCV-susceptible, R5-only virus, suggesting a fitness advantage of wild-type over VCV-resistant virus.
BACKGROUND
ACTG 5211 was a phase IIb study assessing the safety and efficacy of the experimental CCR5 antagonist vicriviroc (1). The study population consisted of 118 treatment-experienced subjects with screening HIV-1 RNA isolates of R5 phenotype only who were experiencing virologic failure (HIV-1 RNA ≥5,000 copies/mL) on a ritonavir-containing antiretroviral regimen. Following the screening phase, subjects added one of four randomized treatments to their existing regimen: vicriviroc placebo, vicriviroc 5mg daily, vicriviroc 10mg daily or vicriviroc 15mg daily; background antiretroviral therapy was optimized at day 14 on the basis of treatment history and resistance testing. At 14 days and 24 weeks, mean changes in HIV-1 RNA levels were greater in subjects receiving an optimized background regimen (OBT)+VCV than OBT alone.
METHODS
Subject Selection
Eight subjects experiencing protocol-defined virologic failure at week 16 (confirmed <1 log10 decrease in HIV-1 RNA levels) were selected at random for analysis - two from each of the four treatment arms. A ninth subject (07J) was included on the basis of our post hoc observation of phenotypic evidence of VCV resistance (Entry Susceptibility Assay).
Tropism Assay and Calculation of Percent Maximal Suppression
Subjects enrolled in A5211 had HIV coreceptor testing (Trofile Assay) and VCV susceptibility testing (Entry Susceptibility assay) performed by Monogram Biosciences.
Env Cloning and Sequencing
Viral RNA was extracted from available subject plasma samples and full-length env amplicons (gp160) generated by nested PCR. The second round gp160 PCR amplicon was purified and ligated into the pCR4 TOPO vector (Invitrogen) at a DNA:vector ratio of 2-3:1. After ligation, bacteria were transformed by electroporation. Putative positive colonies were selected for subcloning. Plasmid DNA was isolated and positive clones were sequenced using the MGH DNA Core Facility. Sequences were edited, aligned and assembled using Sequencher 4.7 DNA sequence analysis software (Gene Codes Corporation, Ann Arbor, Michigan).
Generation of Replication-Competent Recombinant Virus
We modified a yeast gap repair homologous recombination system to create full-length HIV-1 recombinant clones incorporating the env genes (gp160) from subject 07J at different time points (2). Briefly, full-length HIV-1 clone (Q23-17) was isolated and ligated into the multiple cloning site of pRS315 (New England Biolabs). pRS315 sequences permit plasmid replication in both bacteria and yeast, and also contains the beta-isopropylmalate dehydrogenase gene for the leucine synthetic pathway (LEU2). Thus, yeast with the recombined plasmid can be selected in leucine dropout media. A yeast auxotrophic selection marker (HIS3) was then introduced into the nef gene using yeast gap-repair homologous recombination. Yeast auxotrophic marker (URA3) was subsequently introduced into the recombinant plasmid in the place of the HIV-1 full-length envelope segment. Yeast with the recombined plasmid (HIV-pRS315-nef-marker-DEnv) were selected in leucine, histidine, and uracil dropout media. PCR amplified HIV-1 full-length envelopes of interest were introduced in the place of URA3. The recombinant plasmid was selected in dropout media enriched with fluoroorotic acid (FOA). URA3 converts FOA to a toxic product which inhibits yeast with URA3 expression. After plasmid recovery, supernatants with recombinant virus were collected from transfected 293T cells. Virus supernatants are titered on TZM-bl cells using previously described methods (3). TZM-bl is a HeLa based cell line that has been genetically engineered to stably express CD4, CCR5 and CXCR4. In addition, they express Escherichia coli b-galactosidase (β-gal) under the transcriptional control of the HIV-1 LTR (4).
CCR5 inhibitor assay
Sensitivity to VCV has been estimated using a modified version of a previously described assay (5). Briefly, 1X104 TZM-bl cells were incubated with two-fold serial dilutions of VCV in a total of 100 ml at 37∞C for 1 hour. Then, 500 infectious viruses were added to each well. In 48 hours, β-gal expression in the cells was assessed with relative chemi-luminescence units (RLU) using Galacto-Light Plus (Applied Biosystems). All infections were performed in triplicate. Data from Week 16 and 19 env clones represent 2 independent experiments; 3 independent experiments were averaged to generate the data for the weeks 0, 28 and 48 clones. Percent inhibition is expressed as RLUs generated under VCV concentrations relative to a positive viral control in the absence of drug.
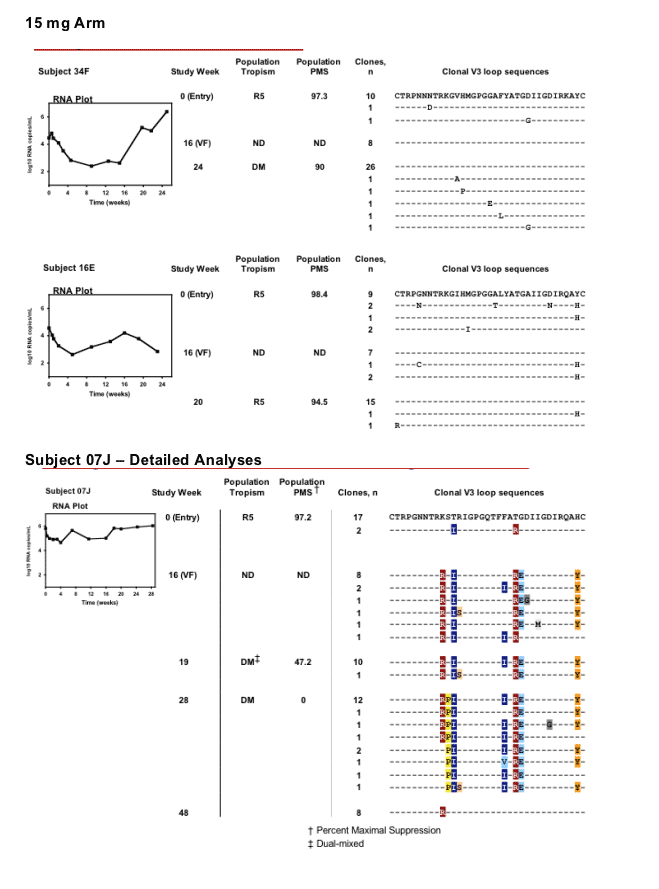
Subject 07J - Detailed Analyses
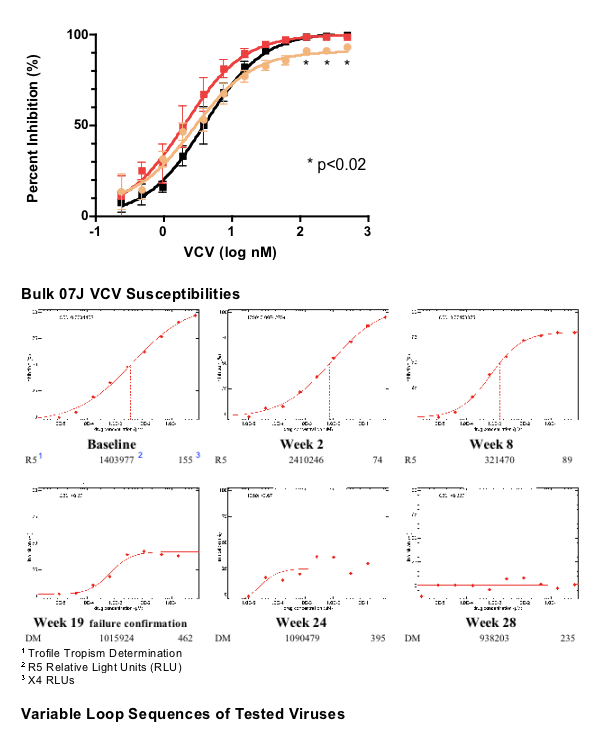
Clonal 07J VCV Susceptibilities
(Percent inhibition)
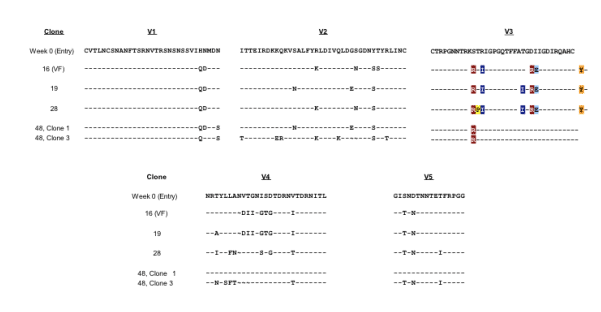
Legend. Using a yeast recombination system (see Methods), infectious recombinant HIV clones with a clonally isolated env gene (gp160) from subject 07J taken from 5 time points - Weeks 0, 16, 19, 28 and 48 - were generated. Subject 07J discontinued VCV in Week 28. Sequence differences in the variable loops between clones is shown for comparison. VCV susceptibility testing from week 0 to 48 demonstrated a progressive plateau effect. Susceptibility testing of a week 28 clone revealed greater viral growth in the presence of VCV than in its absence. Following drug discontinuation and a reversion of V3 loop sequence changes by week 48, VCV susceptibility returned to baseline in one clone (clone 1) but had a 90% plateau of inhibition in the other (clone 3). As shown above, these two week 48 clones have identical V3 loop sequences, suggesting that regions outside of the V3 loop can influence the plateau effect.
|
| |
|
 |
 |
|
|