 |
 |
 |
| |
Resistance to the HIV-Integrase Inhibitor Raltegravir: Analysis of Protocol 005, a Phase 2 Study in Patients with Triple-Class Resistant HIV-1 Infection
|
| |
| |
Reported by Jules Levin
16th Intl HIV Drug Resistance Workshop, June 12-16, 2007, Barbados
D.J. Hazuda, M. D. Miller, B. Y. Nguyen and J. Zhao
for the P005 Study Team Merck Research Labs
Raltegravir (RAL; MK-0518) is an integrase strand transfer inhibitor (InSTI) from the hydroxypyrimidinone carboxamide class. Although structurally distinct from the diketo acids and naphthyridines, RAL is mechanistically similar and shares important chemical features with other InSTIs. RAL has demonstrated robust
efficacy in treatment experienced and treatment-naive patients (IAC 2006 Abstract #THLB0214; ICAAC 2006 Abstract # H-1670b; CROI 2007 Abstract # 105a/b LB).
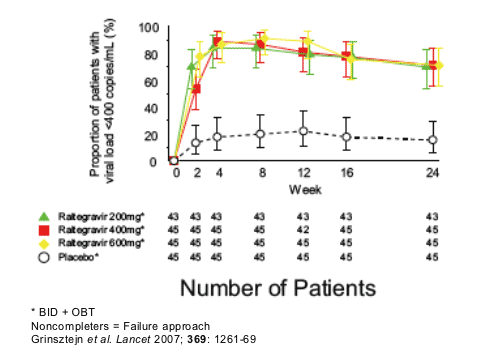
RAL resistance has been studied in vitro and is currently being evaluated in these clinical trials. Merck Protocol 005 is a phase 2 dose-ranging study in HIV-1 patients failing therapy with PI, NNNRTI and NRTI resistance. At week 24, 70% (95% CI: 54, 83) to 71% (56, 84) of patients on RAL and optimized background therapy (OBT) achieved HIV-1 RNA < 400 copies/mL compared to 16% (7, 30) of patients receiving placebo plus OBT (Non-completers = Failure).
In the 38 RAL patients with virologic failure (defined as not achieving > 1 log10 decrease in HIV RNA and HIV RNA < 400 copies/mL at Wk 16 or viral relapse), RAL resistance has been analyzed by sequencing integrase and evaluating all mutations identified relative to baseline as site directed mutants for their effects on susceptibility to RAL (and other InSTIs), and viral replication capacity in single cycle HIV-1 infectivity assays.
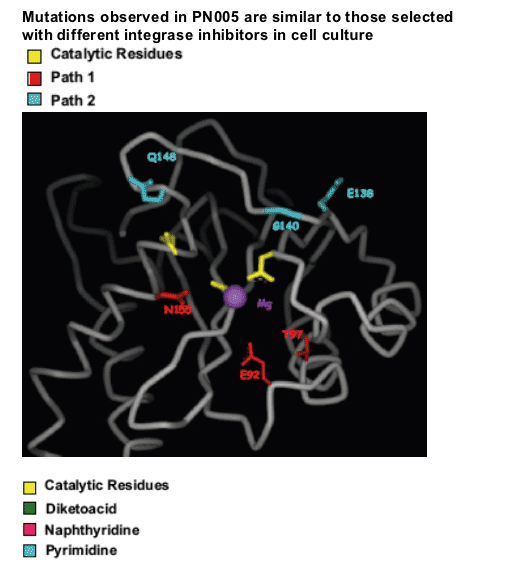
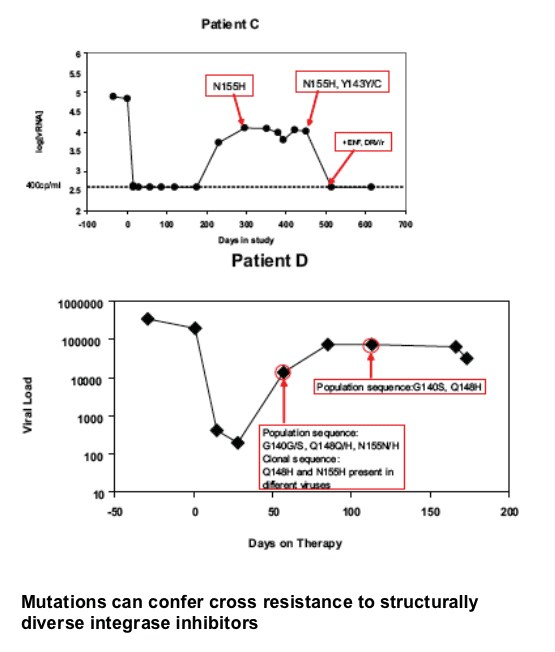
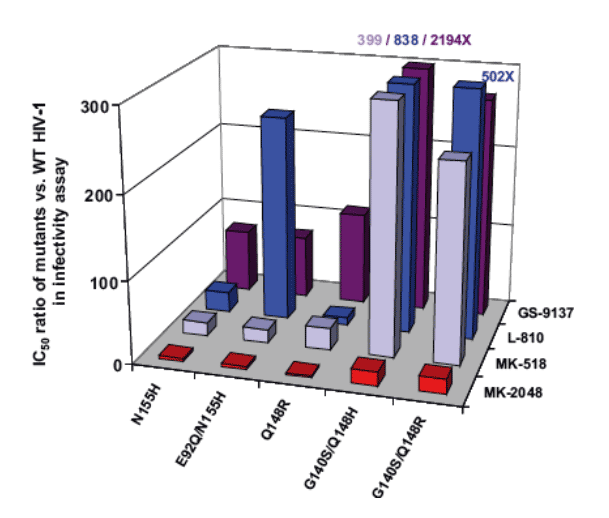
Mutations in the integrase were identified in 35 of the 38 RAL failures. RAL resistance was associated with two genetic pathways defined by mutations at either N155(H) or Q148 (H, R or K) which reduce susceptibility by 10 to 25 fold, respectively. Both in vitro and in vivo, the evolution of additional mutations resulted in high level resistance (N155 plus L74M, E92Q, G163R or Q148H/R/K plus E138K, G140S/A). While certain secondary mutations appear to moderate replication defects associated with specific N155 or Q148 mutations, all secondary changes studied resulted in increased resistance. These results suggest a single mutation may not have been sufficient to confer complete resistance in patients with these viruses. HIV-1 variants with N155 or Q148 mutations exhibited cross resistance to diverse InSTIs consistent with a mechanism that affects binding of the common pharmacophore within the integrase active site.
AUTHOR CONCLUSIONS
Raltegravir failure <25% in triple class resistant patients in PN005
- GSS=0 in >68% of virologic failures
Raltegravir failure was associated with integrase mutation in two distinct genetic pathways defi ned by two or more mutations including
-- A signature mutation at either Q148(H/R or K) or N155(H) and
-- one or more secondary mutations unique to each pathway
Signature mutations at Q148 and N155 conferred >10X reduced susceptibility to raltegravir and resulted in decreased viral replication capacity
Secondary mutations appear to serve primarily to enhance the level of resistance to raltegravir
-- Although mitigation of effects on replication capacity were observed for a subset of combinations (e.g., G140S/Q148H the most common variant), all combinations evaluated to date displayed increased resistance
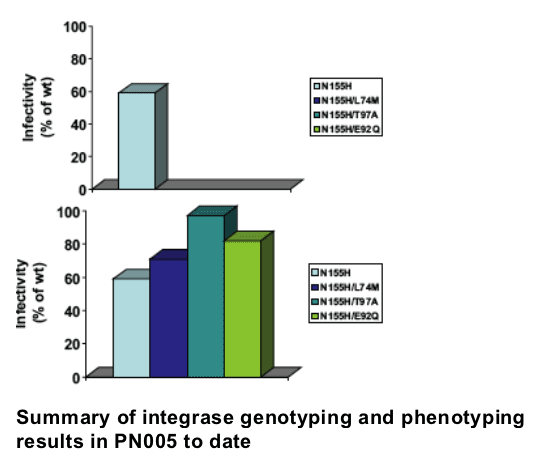
-- Despite >10X resistance, viruses with a single mutation at N155 evolved to acquire increased resistance suggesting a pharmacologic threshold
Mutations at Q148 and N155 can confer cross resistance to structurally diverse integrase inhibitors
MK-0518: Raltegravir
· Integrase strand transfer inhibitor (IC50 about 10 nM)
-- hydroxypyrimidinone carboxamide
· Antiviral IC95 ~ 31 ± 20 nM (50% NHS)
· Active across diverse HIV-1 clinical isolates and HIV-2
· Potent antiviral effect in treatment naive patients
- Phase II 24 week data (Markowitz et. al. IAS 2006)
· Robust activity in HIV-1 patients with triple class resistance
-- Phase II 24 week data (Grinsztejn et al ICAAC 2006)
-- Phase III data (Cooper et al & Steigbigel et al CROI 2007)
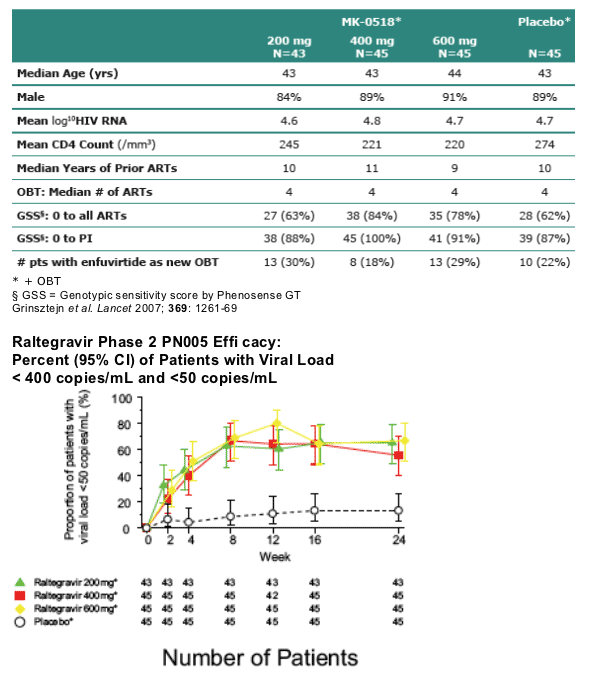
PN005 Phase 2 Study in Treatment Experienced
Patients: Baseline Characteristics
You can see in this table that 18-30% of patients received Fuzeon as new OBT. GSS of 0 to PI: 88% in 200mg arm, 100% in 400mg arm, 91% in 600mg arm. GSS 0 to all ARTs: 63% in 200mg arm, 84% in 400mg arm, 78% in 600mg arm.
Methods: PN005 Phase 2 Integrase Genotyping/
Phenotyping Analysis
· Genotyping
- population sequencing
- first point identifi ed as failure (>400 copies/mL)
- cross sectional analysis
- limited longitudinal studies (ongoing)
· Phenotyping
- site directed mutants
- limited data on patient derived sequences
- single cycle infection assays with LTR- responsive reporter cell lines
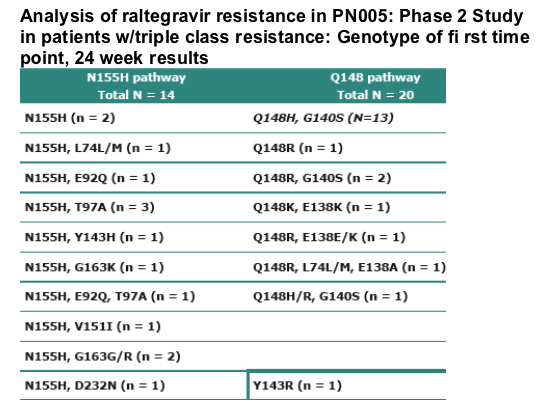
-- Virologic failure was observed in 38/133 (24.8%) patients on RAL
-- Integrase mutations were observed in 35 of 38 patients failing RAL
-- RAL failure was generally associated with one of two genetic pathways (N155 or Q148)
-- Additional mutations were frequently observed with both pathways
-- Q148 containing mutants were more common than N155
-- Most common pattern Q148H/G140S

--Similar number of patients with mutations at amino acids 148 or 155 across 200, 400, and 600 mg treatment groups
-- Factors decreasing likelihood of developing mutations at a.a. 148 alone, 155 alone, either 148 or 155 (hazard ratio < 1):
-- Lower viral load (≦ 100,000 copies/mL vs. > 100,000 copies/mL)
-- Use of enfuvirtide in OBT
-- PSS > 0
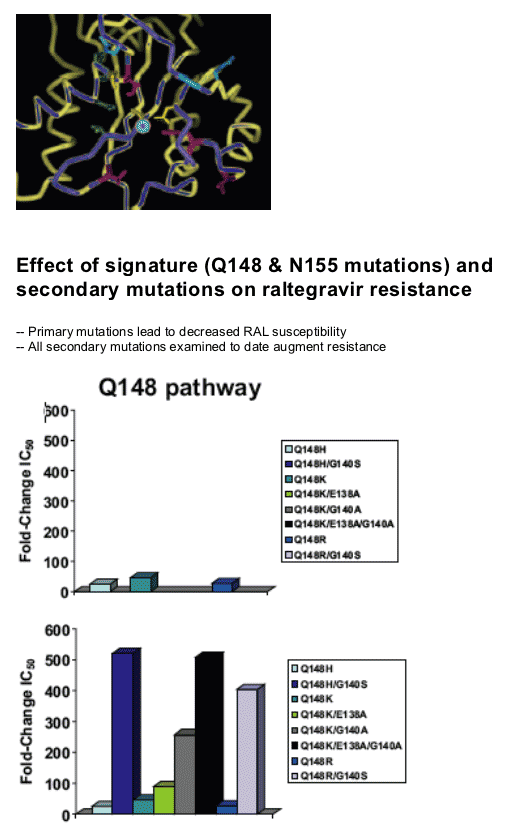
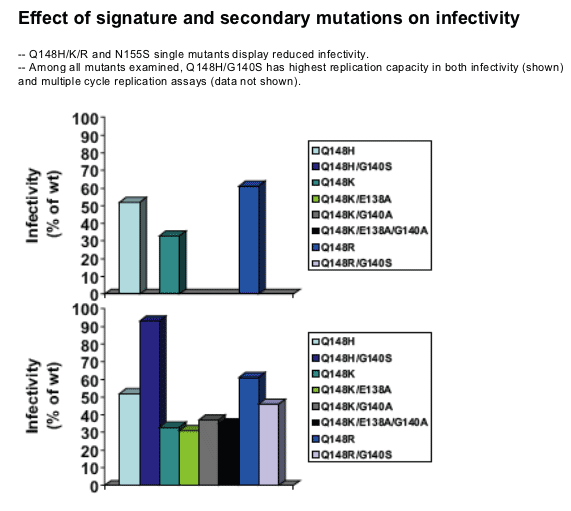
The signature mutations Q148H/R/K and N155S engendered reduced susceptibility to raltegravir and decreased viral replication capacity as individual mutations
Secondary mutations consistently enhanced the level of resistance to raltegravir
-- And also improved viral replication capacity in a subset of variants
Q148 combinations displayed higher level resistance than combinations containing N155H
-- Q148 mutations were more prevalent is PN005
The G140S/Q148H variant was the most resistant and most fit variant
-- Resistance >100x; replication capacity near WT
-- Most common combination observed in PN005
Q148 pathway may be preferred
-- Resistance to raltegravir is higher with 148 mutations (especially doubles)
-- Patients who fail with these Q148 combinations return to baseline vRNA
-- Some patients with N155H viruses appear to retain lower vRNA set point, partial activity or fitness?
Resistance is the primary driver for evolution, is there a pharmacologic threshold?
More longitudinal analysis is required
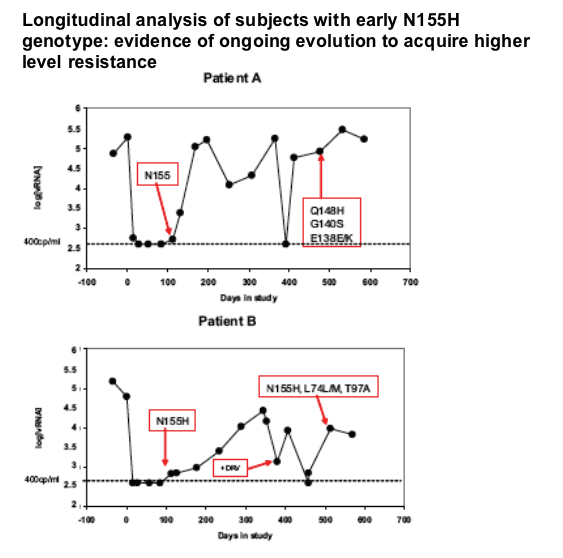
|
| |
|
 |
 |
|
|