 |
 |
 |
| |
Impact of baseline NNRTI mutations on the virological response to TMC125 in the Phase III clinical trials DUET-1 and DUET-2
|
| |
| |
Reported by Jules Levin
16th Intl HIV Drug Resistance Workshop
June 12-16, 2007, Barbados
J Vingerhoets,1 A Buelens,1 M Peeters,1 G Picchio,2 L Tambuyzer,1 H Van Marck,1 G De Smedt,1 B Woodfall,1 MP de Béthune1
1Tibotec BVBA, Mechelen, Belgium; 2Tibotec Inc., Yardley, PA, USA
AUTHOR CONCLUSIONS
Using the pooled data from DUET-1 and DUET-2, 13 baseline RT mutations were associated with resistance to TMC125
-- V90I, A98G, L100I, K101E, K101P, V106I, V179D, V179F, Y181C, Y181I, Y181V, G190A, G190S
-- 70% of patients had no or only one TMC125 RAM
-- 15% of patients had 3 TMC125 RAMs
-- the TMC125 RAMs mainly occurred in the presence of other NNRTI RAMs.
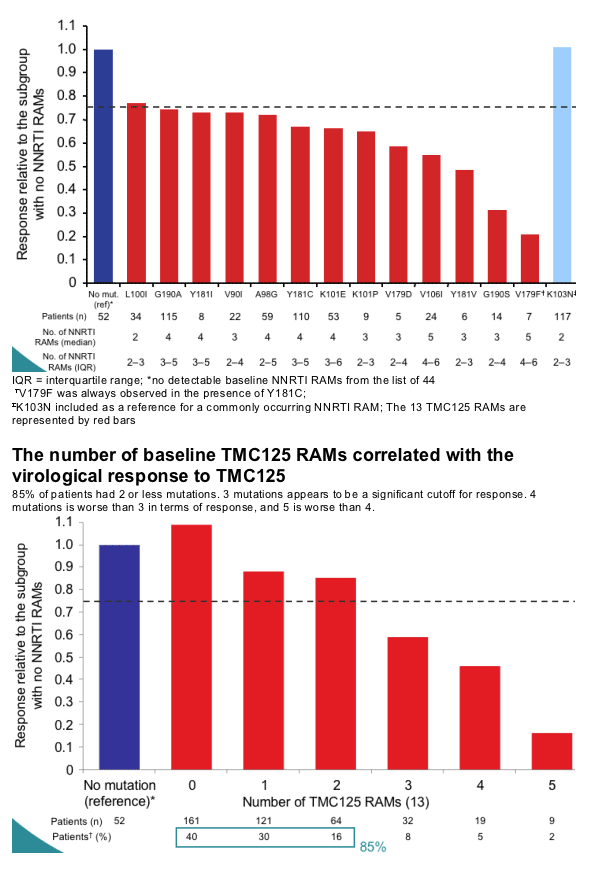
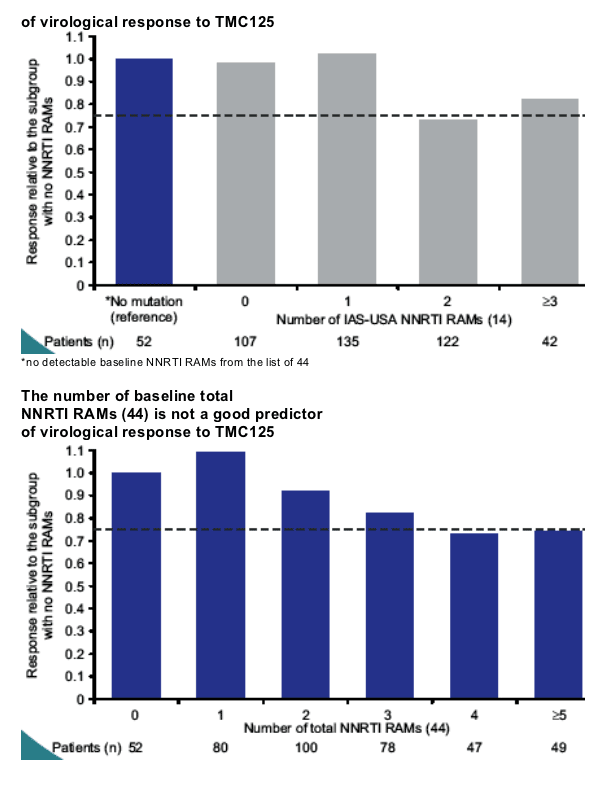
An increasing number of baseline TMC125 RAMs was associated with a decreasing virological response to TMC125
-- this effect was highly statistically significant in a multivariate analysis correcting for baseline parameters such as viral load, phenotype sensitivity score and DRV FC
-- the largest impact on virological response was observed in the subgroups of patients with three or more TMC125 RAMs.
These pooled data from the Phase III DUET trials in treatment-experienced
patients with HIV-1:
-- identify the baseline genotypic determinants of decreased virological response to TMC125
-- provide guidance in the interpretation of genotypic resistance data related to TMC125.
Background
TMC125 (etravirine, ETR) is a next-generation NNRTI, with activity against NNRTI-resistant HIV-1 and a high genetic barrier to the development of resistance. This analysis was aimed at identifying baseline genotypic determinants of decreased virological response to TMC125 (200mg bid) in
the Phase III, double-blind, placebo-controlled trials, DUET-1 and DUET-2. To provide guidance in the interpretation of genotypic resistance information related to TMC125.
Methods
Effect of baseline phenotype (Antivirogram®) and genotype (virco®TYPE HIV-1) on the virological response to TMC125 at Week 24 was studied in the subgroup of patients not using enfuvirtide as a new drug in analyses and excluded
patients who discontinued for other reasons than virological failure (n=406). Virological response at Week 24 is presented relative to the response achieved by the subgroup of patients without detectable NNRTI RAMs at baseline* (n=52; relative response = 1.0)
(*patients had NNRTI mutations from previous genotype)
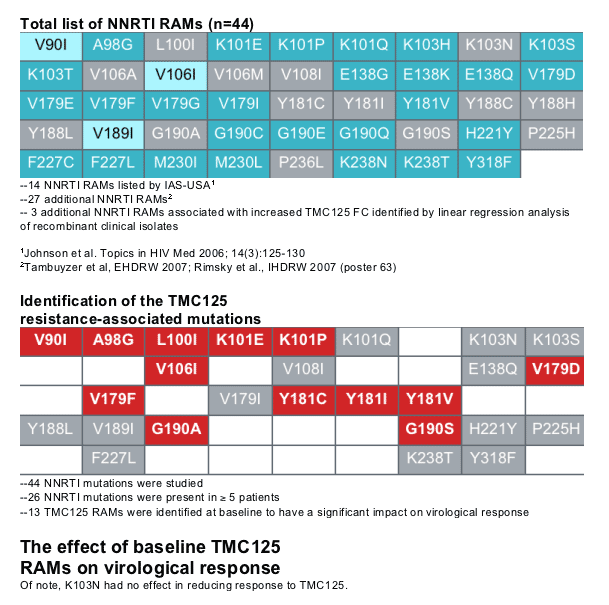
Decreased virological response was defined as a response that was at least 25% lower than that of the subgroup of patients without detectable NNRTI RAMs at baseline, using the primary efficacy endpoint of confirmed viral load <50 HIV-1 RNA copies/mL; (primary efficacy endpoint: <50 HIV-1 RNA copies/mL). Forty-four reverse transcriptase (RT) mutations were studied, but only those present in 35 patients at baseline were included in the final analyses. RT mutations were included in the analysis if they were present at baseline in 5 or more patients
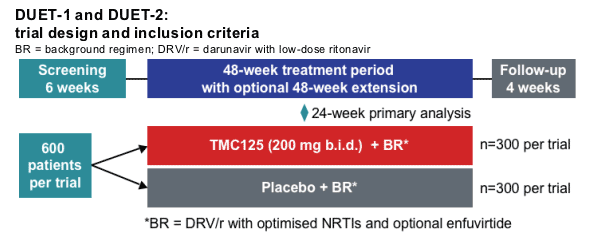
--Plasma viral load at screening: >5,000 HIV-1 RNA copies/mL
--Stable antiretroviral therapy for at least 8 weeks at screening, and remain on that treatment until baseline
--At least three primary protease inhibitor mutations at screening
--At least one NNRTI resistance-associated mutation (either at screening or in documented historical genotype)
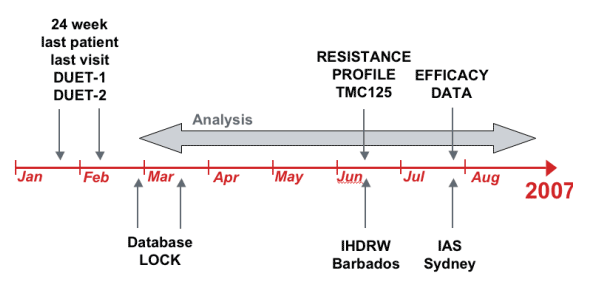
DUET-1 and DUET-2: timeline; Efficacy Data Will Be Presented at Sydney IAS, July 2007
No efficacy data will be shown. Virologic response data will be shown relative to the response in the subgroup of TMC125 treated subjects without detectable baseline NNRTI mutations.
Results
Of the 44 mutations studied, 26 were present in 35 patients at baseline. Univariate analyses identified the following mutations to be associated with decreased virological response to TMC125: V90I, A98G, L100I, K101E, K101P,
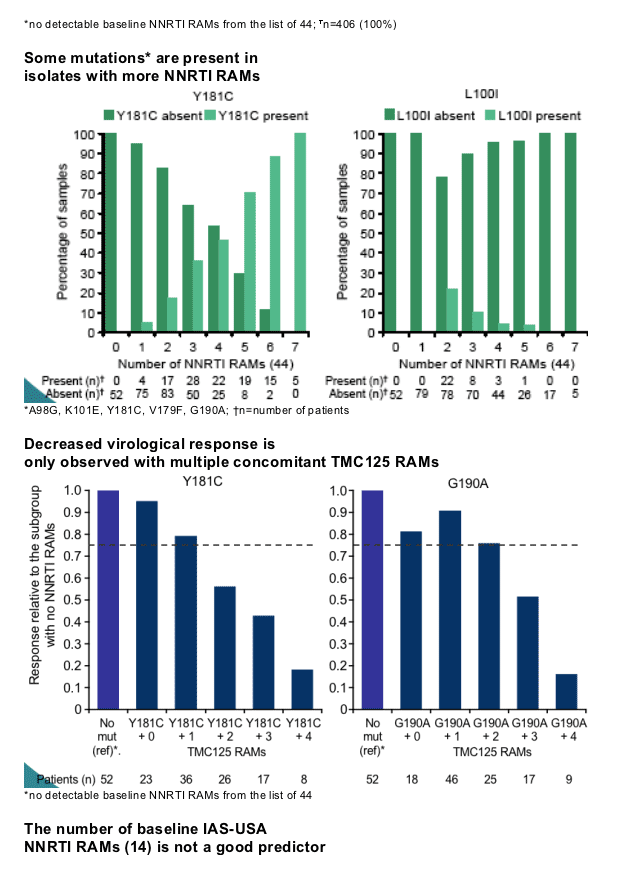
V106I, V179D, V179F, Y181C, Y181I, Y181V, G190A, and G190S (TMC125-RAMs). Isolates with A98G, K101E, Y181C, V179F, or G190A harboured more NNRTI mutations than isolates without these mutations. V179F was never present without Y181C. Y181V, G190S and V179F, which had the most pronounced effect on response, were present in <5% of patients.
TMC125 fold change (FC) in EC50 increased with increasing numbers of NNRTI RAMs or TMC125 RAMs and was a strong predictor of virological response. Accordingly, virological response decreased progressively among subgroups with increasing numbers of TMC125 RAMs. The largest impact was observed in the subgroup of patients with >3 TMC125 RAMs at baseline.
Conclusions
Thirteen mutations, mainly occurring in the presence of other NNRTI RAMs were associated with a decreased response to TMC125. The decrease was a function of the number of baseline TMC125 RAMs, with the largest impact in the subgroup of patients with >3 of those. Additional analyses will provide more insight into the role of these mutations, individually and combined, in resistance to TMC125.
|
| |
|
 |
 |
|
|