 |
 |
 |
| |
Intermittent Antiretroviral Prophylaxis with Tenofovir and Emtricitabine (FTC) Protects Macaques Against Repeated Rectal SHIV Exposures
|
| |
| |
Reported by Jules Levin
16th Intl HIV Drug Resistance Workshop, June 12-16, 2007, Barbados
J Gerardo Garcia-Lerma, Ron Otten, Mian-er Cong, Eddie Jackson, Robert Janssen, Thomas Folks, Walid Heneine
Laboratory branch of HIV/AIDS Prevention, NCHHSTP, centers for Disease Control and Prevention, Atlanta, GA
AUTHOR SUMMARY & CONCLUSIONS
Short and potent PrEP interventions can be as effective as daily PrEP.
Single-dose PrEP is highly effective but not sufficient to fully protect against rectal transmission.
Data indicates that the extended antiviral activity in the 2nd 24 hr dose also contributes to protection, possibly by blocking local expansiob/dissemination.
Findings illustrate the high vulnerability of SHIV to potent interventions during the initial stages of mucosal infection.
Our macaque model demonstrates the promise of intermittent PrEP with FTC/TDF as a strategy to prevent sexual HIV transmission.
ABSTRACT
Background: Antiretroviral pre-exposure prophylaxis (PrEP) is a promising new biomedical strategy to prevent the transmission of HIV. We recently showed that daily PrEP with tenofovir (TFV) and emtricitabine (FTC) fully protected macaques from rectal SHIV transmission. TFV and FTC have long intracellular half-lives and can potentially achieve extended prophylactic activity when given intermittently. We assessed if PreP given around the time of virus exposure can be as protective as daily PrEP.
Methods: Two different intermittent PrEP modalities with FTC (20 mg/kg) and (TFV (22 mg/kg) were given subcutaneously to rhesus monkeys. One group (n=6) received FYC/TFV 2 hours before and 24 hours after virus exposure and other group (n=6) received FTC/TFV only 2 hours before virus exposure. Protection was compared to that previously seen with daily FTC/TFV PrEP. An additional 21 macaques (9 real time and 12 historical controls) did not reveice any drug treatment. All animals were exposed rectally once weekly for up to 14 weeks with a low dose of SHIV-SF162p3 (10 TCID50 or 7.6x10-5th vRNA copies) that expresses an R5 tropic HIV-1 envelope. Infection was monitored by serology and PCR amplification of SHIV sequences from plasma and PBMCs. Animals were considered protected if they were seronegative and PCR negative during PreP and up to 70 days thereafter.
Results:
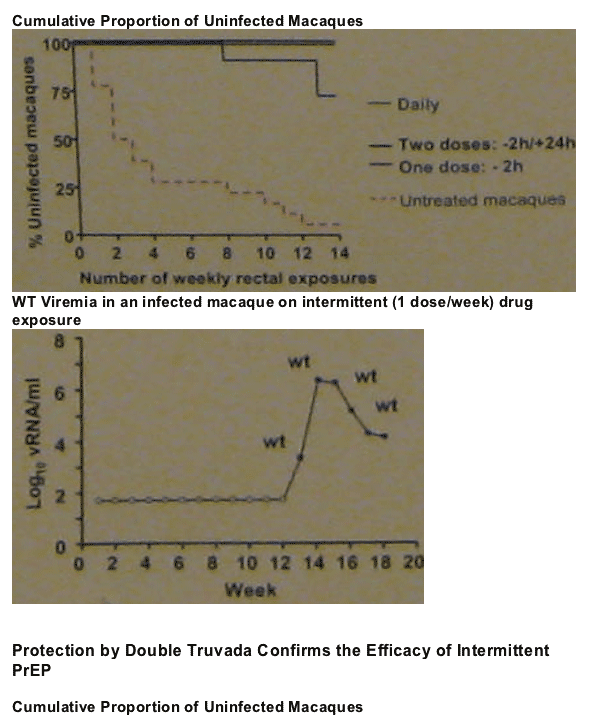
20 of the 21 untreated macaques became infected after a median of 2.5 rectal exposures. The majority of the animals (16/21 or 76%) were infected during the first 4 challenges (median=2.1), 4 (19%) are infected between exposures 8 and 12 (median=10.5), and only 1 (4.8%) remained uninfected after 14 exposures.
All the animals receiving daily PrEP or prep before and after virus exposure were protected.
Available data from 10 challenges in macaques receiving a single-dose PreP showed 1 of 6 animals infected at exposure 8.
Conclusions:
Two-dose PrEP given before and 24 hours after virus exposure can be as effective as daily PrEP. Single-dose PreP is highly effective but not sufficient to achieve full protection, suggesting that the extended antiviral activity in the second 24 hour dose also contributes to protection.
These data highlight the importance of effectively blocking the early stages of virus replication and show the promise of intermittent PreP as a strategy for HIV prevention.
BACKGROUND
There is a need to identify highly effective biomedical interventions to complement behavioral interventions.
Pre-exposure prophylaxis (PrEP) with ART drugs may have considerable potential to prevent sexual HIV transmission. The most effective is not known.
There is encouraging data with daily PrEP in macaques model:
--daily PrEP with TFV, FTC, or FTC/TFV delayed or prevented infection.
--potent drug combinations may be more effective than single drugs in preventing mucosal HIV transmission (note from Jules: previous data I recall found 2 drug combination TDF/FTC was more effective than single drug alone, I think both either TDF or FTC alone).
Ongoing clinical trials in high risk populations (Botswana, West Africa, Thailand, Peru, Ecuador, US).
Can intermittent PrEP with FTC/Tenofovir be effective as daily PrEP?
RATIONALE
FTC and tenofovir have long intracellular half-life and can potentially achieve extended prophylactic activity.
Potential cost-effectiveness and lower risks for drug toxicities.
The initial founder virus population is small (SIV models). Window of maximum virus vulnerability before local expansion and dissemination.
METHODS
REPEATED EXPOSURE MACAQUE MODEL OF SHIV TRANSMISSION
-- SHIV162p3 virus challenge containing an R5 tropic HIV-1 envelope.
-- More physiologic virus inoculum (10 TCID50 or 7.6 x 10-5th RNA copies). Virus inoculations repeated weekly to better assess protection over several exposures.
-- Protection measured by the degree infection is delayed or prevented.
--Infection monitored by serology and PCR amplification of SHIV RNA and SHIV DNA sequences.
--Resistance testing done by sequence analysis and more sensitive allele-specific real-time PCR assays.
DESIGN
SUBCUTANEOUS FTC/TFV
Group 1 (n=6): 2-dose PrEP with TFV (22 mg/kg) and FTC (20 mg/kg) administered 2 hrs before and 24 hrs after virus exposure, repeated weekly for a total of 14 exposures.

Group 2 (n=6): One-dose PrEP with TFV (22 mg/kg) and FTC (20 mg/kg) administered only 2 h before virus exposure.

Comparison Group (n=6): Daily PrEP with TFV (22 mg/kg) and FTC (20 mg/kg).
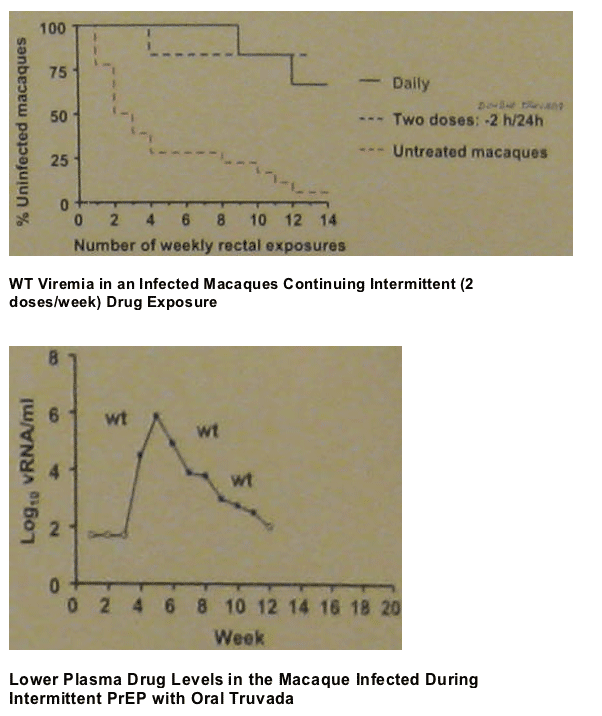
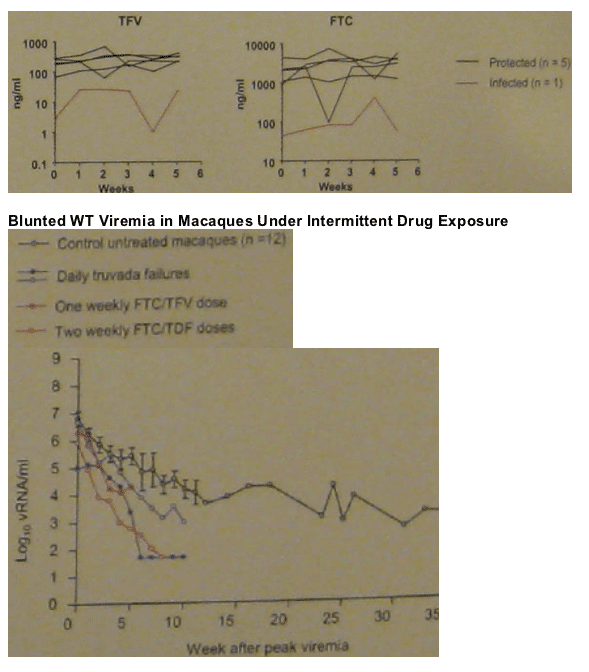
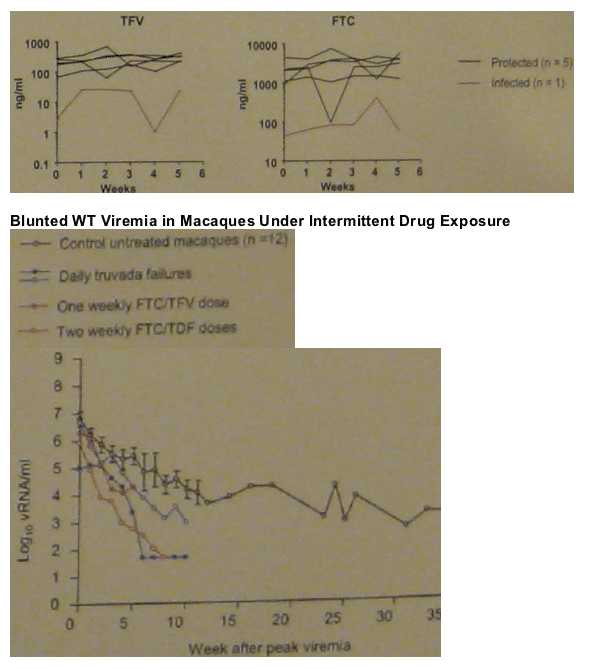
DOUBLE TRUVADA
Group 4 (n=6): 2-dose PrEP with twice the human equivalent dose of TDF (44 mg/kg) and FTC (40 mg/kg) administered 2 h before and 24 h after virus exposure.

Comparison group (n=6): daily PrEP with oral TDF (22 mg/kg) and oral FTC (20 mg/kg).
UNTREATED
Group 6 (n=21; 12 real time and 9 historical controls).
RESULTS
2-dose PrEP is fully protective; 1-dose PrEP is not sufficient.
|
| |
|
 |
 |
|
|