 |
 |
 |
| |
Nevirapine Increases High Density Lipoprotein-Cholesterol (HDL-C) by Stimulation of Apolipoprotein AI Synthesis
|
| |
| |
Reported by Jules Levin
9th Intl Workshop on Adverse Drug Reactions and Lipodystrophy in HIV
Sydney, Australia, July 19-21, 2007
"Nevirapine Intensive Lipid Evaluation trial"
Peter Reiss
Academic Medical Center, Amsterdam, the Netherlands
NNRTIs show relatively favorable lipid changes in regards to cardiovascular risk, notably increases in HDL-cholesterol. HDL-C levels are associated with CVD risk. This study investigated the mechanism by which the NNRTI nevirapine (NVP) increases HDL-c.
AUTHOR CONCLUSIONS & DISCUSSION
Nevirapine in patients with sustained HIV suppression indeed elevates HDLc and apoA1
Degree of HDL-c rise compatible with earlier reports in pretreated patients:
--21% increase HDL (Fisac, AIDS, 2005)
--20% increase HDL (Ruiz, AIDS 2002)
Rises in HDL-c and apoA-I lower than seen in naive patients
Suggests that part of the HDL rise in naive patients indeed results from "return-to-health" effect
Nevirapine increases apoA-I and HDLc by promoting apoA-I production without affecting HDL-c catabolism (no CETP inhibition!)
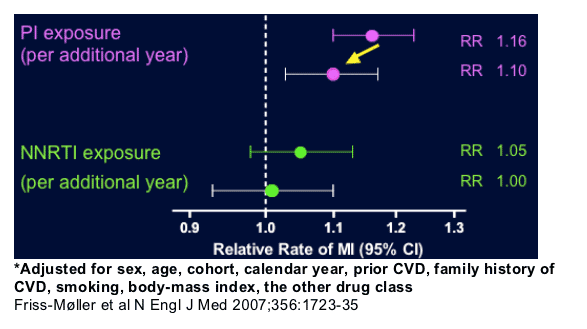
This may contribute to our understanding why in HIV-infected patients increased CVD risk has been reported with:
--PI-, but not with NNRTI-based therapy (D:A:D)
--Interruption of NNRTI-based therapy in particular (SMART)
In view of recent findings that CETP-inhibition may actually promote atherogenesis and CVD events*, our findings may hold promise for developing novel therapeutics aimed at increasing HDL-c in ways which hopefully would offer protection against CVD.
* Kastelein J et al. N Engl J Med 2007; ILLUMINATE trial
http/clinicaltrials.gov/show/NCT00134264
ABSTRACT
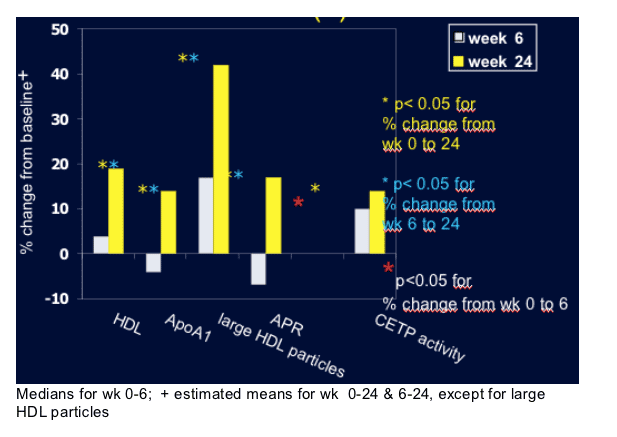
Methods: 13 HIV-1 patients with plasma HIV-1 RNA <50 copies/ml on AZT/3TC/abacavir for >6 months added NVP at usual dose for 24 weeks. Before (week 0), and 6 and 24 weeks later patients received a primed bolus infusion of stable isotope L-[1-13C]-valine for 12 h to study apolipoprotein A-1 (apoA1) kinetics. Using SAAM-II modeling, absolute production rates (APR) and fractional catabolic rates (FCR) of apoAI were calculated. All HDL-C modulating enzymes were assessed. Analysis by ITT. Wilcoxon signed rank test for % change in large HDL particles and % change from week 6 to week 24; mixed model repeated measures for other analysis.
Results: See Table 1 below. Other HDL-modulating enzymes, including hepatic lipase, lecithin:cholesterol acyl transferase and phospholipids transfer protein remained unchanged.
Conclusions: NVP increased apoAI and HDL-C by selectively promoting apoAI production without affecting HDL catabolism. This may contribute to why the increased CVD risk with some PI-therapy has not been found with treatment including NNRTI. Moreover, in view of the recent disappointing results of HDL increasing strategies with the CETP inhibitor torcetrapid targeting HDL degradation, our findings may lead to the identification of more promising novel targets for increasing HDL-C.
Background
Effects of Different ARV Classes on HDLc
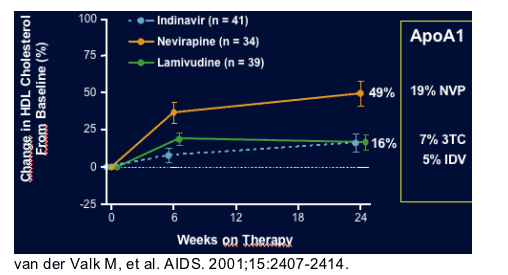
ATLANTIC Trial: Nevirapine use increased HDL cholesterol by 49% from
baseline.
NVP arm 49% (p<0.001)
IDV arm 16% ( p= 0.010)
3tc arm 16% (p= 0.010)
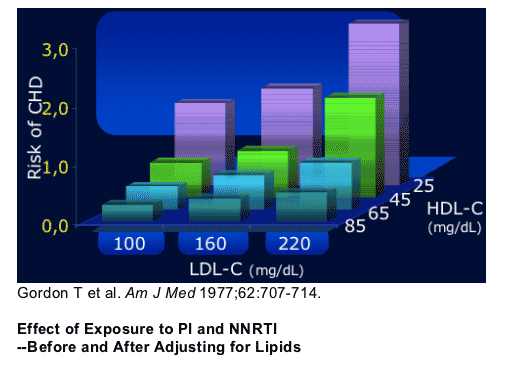
Low HDL-C Independent Predictor of
CVD Risk even when LDL-C is low
Low HDL-C is an independent predictor of CHD risk even when LDL-C is low
Data from the Framingham Heart Study indicate that high-density lipoprotein cholesterol (HDL-C) is an independent risk factor. Even in individuals whose low-density lipoprotein cholesterol (LDL-C) levels were approximately 100 mg/dL, HDL remained a very strong risk factor, and individuals with low HDL-C were still at considerably elevated risk.
Aim of the Nevirapine Intensive Lipid Evaluation study
To explore the possible mechanism by which NVP induces increases in HDL cholesterol
HDL Metabolism Involves a Complex Sequence
of Synthetic and Degradative Steps
Enzymes involved in HDL metabolism
LCAT = Lecithin:cholesterol acyltransferase
LPL = Lipoprotein Lipase
CETP = Cholesteryl Ester Transfer Protein
HL = Hepatic Lipase
EL = Endothelial Lipase
PLTP = Phospholipid Transfer Protein
CETP deficiency was shown to play a role in the development of high HDL-c in the 1980s in the Japanese population. Recent research confirmed that 64% of high HDL-cases in Japan could be attributed to CETP-deficiency (defined as having CETP activity <75% of control subjects). The remaining 35% results therefore from other factors than CETP deficiency (secondary, other genetic factors).
In the present patient group we evaluated LCAT (higher activity = higher HDL-c), CETP (higher activity is lower HDL-c), HL (higher activity is lower HDL), EL (higher activity is lower HDL), LPL (higher activity is higher HDL) and PLTP (high activity is low HDL)
Causes for HDL increase can thus be: stimulation of LCAT (very rare!!) or LPL
Or inhibition of CETP, HL, EL or PLTP.
In the present study LCAT, CETP, HL, EL, LPL and PLTP activity were, however, unchanged.
HIV Infection By Itself Affects Lipids
--reduces HDL-C
--reduces LDL-C
--predominance of small dense LDL (LDL-B phenotype)
--increased triglycerides
Part of the early lipid changes, including a rise in HDL-C observed with any ART combination may represent, in part, a 'return to health' rather than a direct 'toxic' drug effect.
Grunfeld C, et al. J Clin Endocrinol Metab. 1992;74:1045-1052.
Riddler et al. JAMA 2003; 289: 2978-2982.
Study Design & Patient Population
Open label, non-randomized, single-arm study
Patients with HIV-RNA ≦ 50 c/ml while on ZDV/3TC/ABC (Trizivir) for >6 mos
Addition of NVP at baseline: 200 mg first 2 wks, followed by 200 mg bid
24 week follow up
Exclusion criteria:
-Prior NNRTI exposure
-Diabetes Mellitus
-Hypertension
-History of cardiovascular disease
-Use of lipid modifying agents
Methods
Main outcome measures as % change from baseline:
_--Fractional synthetic rate (FSR)
and absolute production rate (APR) of apoA-I
--TC, HDLc, calc LDLc, TG
--Various lipoprotein particles: size & concentration
by NMR (nuclear magnetic resonance spectroscopy)
--Activity of HDL-metabolism-related enzymes
ApoA-1 kinetic study
Prior to, and 6 and 24 weeks after initiation of NVP
12 hr continuous i.v. infusion of stable isotope L-13C valine which is incorporated into newly synthesized apoA1
Valine enrichment determined by GC/C/IRMS:
--Tracer / tracee ratio
--SAAM-II modeling:
**apoA1 fractional synthetic rate (FSR) and absolute production rate (APR)
**At steady state: FSR equals fractional catabolic rate (FCR)
Statistical Analysis
All analyses: percent change from baseline (i.e. adjusted for baseline values) by intention to treat:
--For each outcome measure from week 0 to 6:
Wilcoxon signed-rank test
--For FSR, APR, lipid enzymes, and TC, HDLc, calc LDLc,TG
from week 0 and 24 (also taking week 6 data into account), and week 6 to 24
:
Repeated measures analyses
--For lipoprotein particle size & conc, by NMR (wk 0-6, wk 0-24, wk 6-24)
Wilcoxon signed-rank test
RESULTS
15 patients screened: 1 never started, 1 pt viral load >50 c/ml.
13 started NILE: 2 pts prematurely stopped NVP for hepatotoxicity after 3 weeks (I think Reiss said study was started before the CD4 guidelines were instituted) (BL CD4 705 & 610).
Week 6 (n=11) to week 24 (n=11):
Baseline Characteristics
13 males
Mean Age (SD): 44 (10)
Median CD4 count (range): 595 (200-840)
(9/13 men BL CD4 >400; none after implementation of CD4 thresolds for NVP).

|
| |
|
 |
 |
|
|