 |
 |
 |
| |
Telaprevir in Combination with Peginterferon-a-2a With or Without Ribavirin in the Treatment of Chronic Hepatitis C Treatment-Naïve Genotype 1:
Final Results of the PROVE2 Study
(conducted in Europe)
|
| |
| |
Reported by Jules Levin
59th EASL Nov 3 2008 San Francisco, CA

PROVE2 SUMMARY
Telaprevir (TVR)-based regimen demonstrated a significantly higher SVR rate with a shortened treatment duration compared with the control arm in treatment-naïve, HCV genotype-1 infected patients.
-- T12/PR24, 69% SVR PR48 48% SVR
(T12/pr24 = 12 weeks telaprevir, 24 weeks pegIFN/RBV PR48 = 48 weeks pegIFN/RBV)
RBV is still a necessary component of the treatment regimen
The most common adverse events reported more frequently in the T/PR arm versus control were pruritis, rash and anemia
-- rash led to discontinuation of all study drugs in 7% of patients
-- Anemia led to discontinuation of all study drugs in 1% of patients
AUTHOR CONCLUSIONS
Telaprevir based regimens have the potential for a new treatment approach, including a 24-week treatment duration for most treatment-naïve HCV genotype-1 infected patients
Currently being confirmed in the Phase 3 ADVANCE Study
PROVE2: PROtease Inhibition for Viral Evaluation
Randomized partially blinded, controlled, Phase 2b clinical trial
-- 323 patients
Key eligibility criteria:
-- HCV genotype-1 infection, treatmnt-naïve, age 18 to 65 years, METAVIR stage F0 to F3
Molecular diagnostics
-- HCV RNA assessment by Roche Cobas Taqman HCV test for use with the HighPure System Vital Nucleic Acid Kit version 1.0 (limit of detection 10 IU/mL: lower limit of quantification 30 IU/mL)
-- Genotyping by sequence analysis
Statistical analysis
-- Intention-To-Treat (ITT) analysis included all randomized patients who received at least one dose of study drug
-- Fisher's exact test was used for statistical comparison
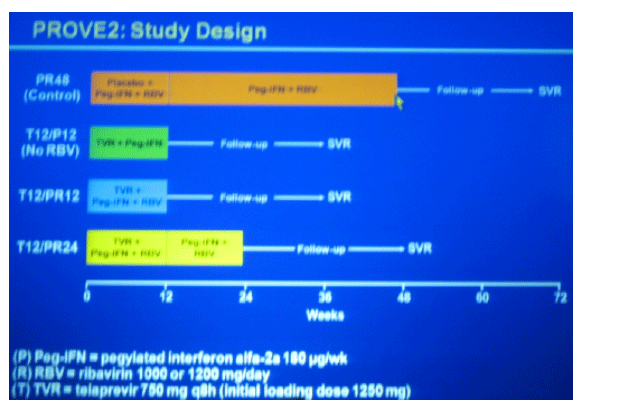
4 study groups:
PR48 (Control): placebo+peg?RBV
T12/P12 (No RBV): TVR+pegIFN 12 weeks
T12/PR12: TVR + pegIFN + RBV
T12/PR24: TVR + pegIFN + RBV
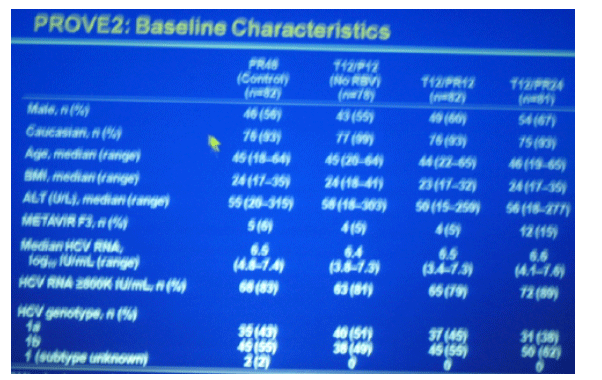
56-67% men.
93-99% Caucasian
age median 44-46
BMI median 23-24
ALT 50-58
METAVIR F3 15% in T12/PR24; 5-6% in other 3 arms
Median HCV RNA 6.4-6.9 log
HCV RNA >800K 79-89%
HCV genotype:
1a 38-51%
1b 49-66%
N= 78-82 in each group
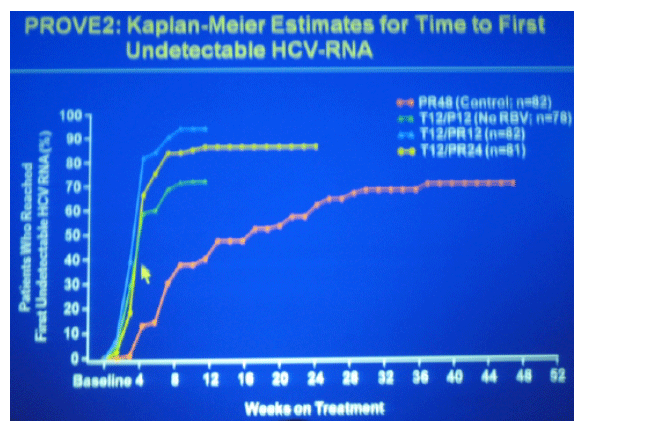
blue T12/PR12 n=82
yellow T12/PR24 n=81
green T12/P12 (no RBV n=78)
red PR48 (control n=82)
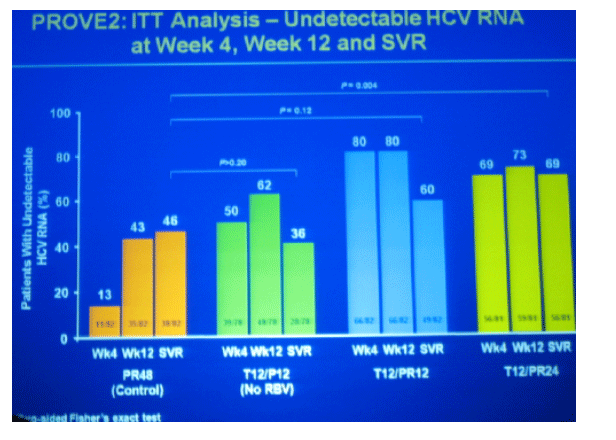
69% SVR in T12/PR24
60% SVR in T12/PR12
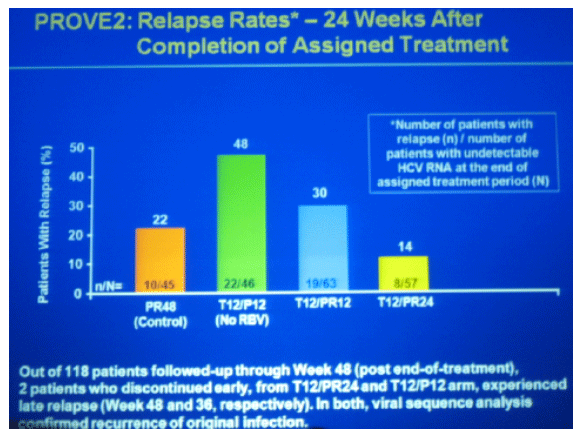
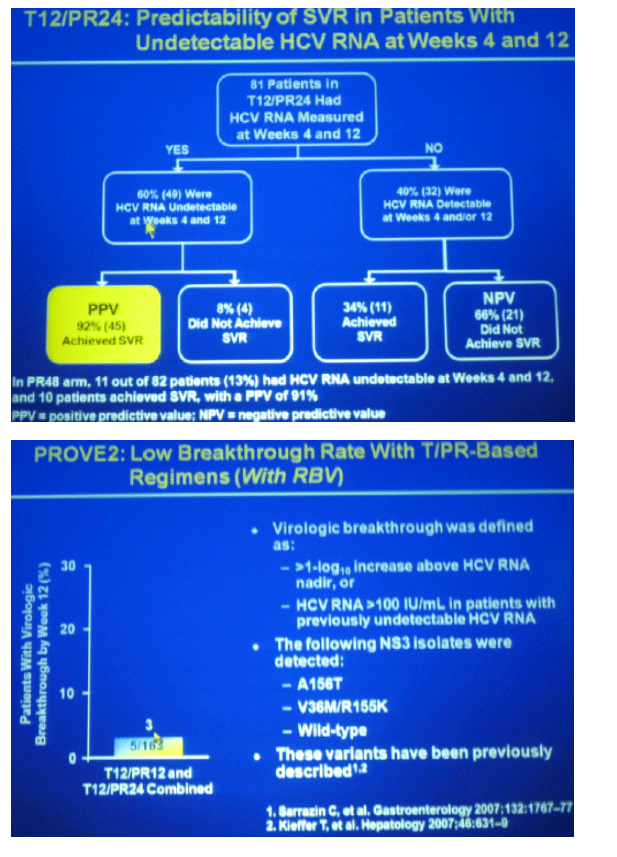
pruritis 51-63% in TVR groups, 35% in peg/RBV control
rash, all types 35% peg/RBV control, 44-49% in TVR groups
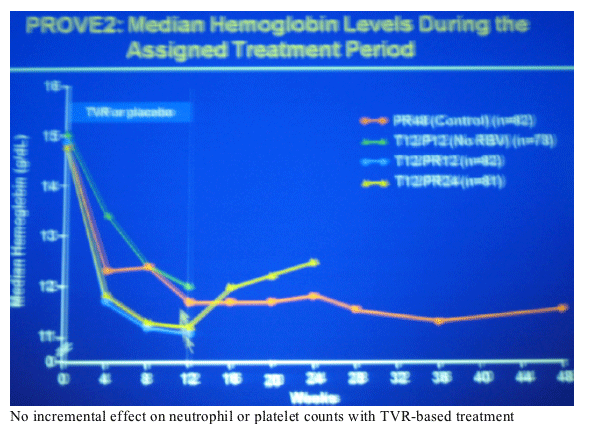
anemia 17% peg/RBV control, 9% T12/P12, 13% T12/PR12, and 27% in T12/PR24

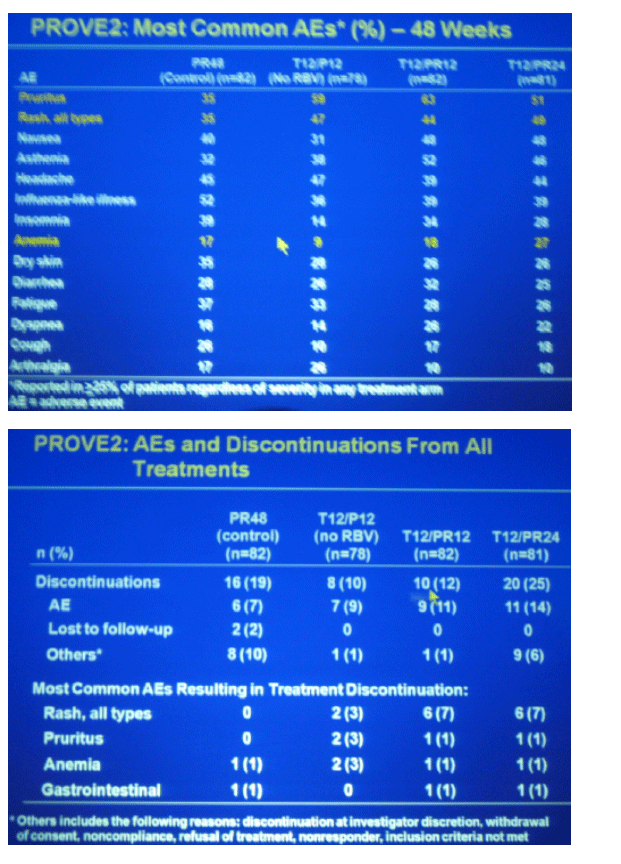
RASH
7% (12 patients) discontinued all study drugs due to rash
Typical, maculopapular exanthema
Median time from start of antiviral treatment to first onset of severe rash was 40 days (range: 1 to 74 days)
Rash and pruritis regressed after TVR withdrawal and appropriate therapeutic measures
Median time from onset to resolution of all rash was 32 days
|
| |
|
 |
 |
|
|