 |
 |
 |
| |
Pharmacokinetics of once-daily regimens of the novel HCV NS3/4A-protease inhibitor TMC435350, with and without peg-IFN and ribavirin, in HCV-infected individuals
|
| |
| |
Reported by Jules Levin
AASLD Nov 3 2008 San Francisco, CA
Gerben van 't Klooster1, Iris Vanwelkenhuysen2, René Verloes1, Kris Marien1, Pieter Van Remoortere1 and Kenny Simmen1
1Tibotec BVBA, Mechelen, Belgium & Tibotec Pharmaceuticals Ltd, Ireland; 2Johnson & Johnson Pharmaceutical Research and Development, Beerse, Belgium
AUTHOR CONCLUSION
Once-daily regimens of 25 mg, 75 mg, and 200 mg TMC435 yield plasma concentrations well in excess of its EC50, and pharmaco-kinetic steady-state is attained by day 7 following these doses
BACKGROUND
TMC435 (Figure 1), formerly known as TMC435350, is a potent and selective HCV NS3/4A-protease inhibitor.
TMC435, preclinical profile:
EC50 = 6 ng/mL (8 nM; genotype 1b replicon).
About 2-fold shift of EC 50 value in the presence of plasma proteins.
Good oral bioavailability: 45-80%
High liver to plasma ratio: >30
TMC435 clinical pharmacokinetics (PK):
Supports once-daily (QD) dose regimens for efficacy · evaluation at doses as low as 25 mg.(1)
Lack of food effect with oral solution. (2)
Mean plasma concentrations increase in a dose-proportional fashion for doses up to 100 mg, and more than dose-proportional for higher doses, both for single and repeated dosing.(2)
Subject to prolonged absorption with a tmax of 6 hours.
Time to steady state is determined by characteristics of absorption rather than elimination.(1)
We currently present steady-state PK data of TMC435, both in healthy volunteers (200 mg QD), and in HCV-infected individuals (25 and 75 mg QD), from a completed phase I trial (TMC435-C101) and an interim analysis in an ongoing dose ranging trial (OPERA-1, TMC435-C201).
METHODS
Healthy volunteers:
Healthy volunteers received TMC435 at a dose of 200 mg QD for 7 days, as a capsule (trial TMC435-C104). Data were compared to those from a prior clinical trial(1), in which TMC435 was given as an oral solution at 200 mg QD for 5 days in healthy volunteers.
HCV-infected patients:
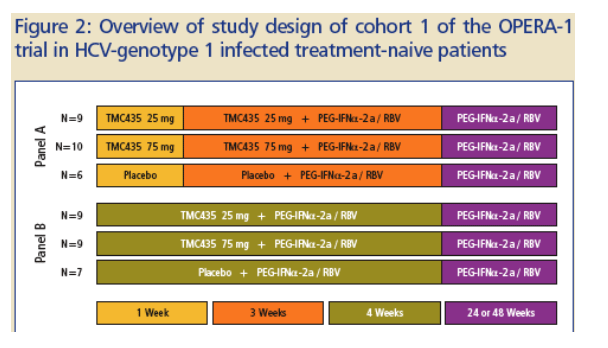
A dose escalating, Phase IIa or proof-of-concept trial is ongoing in Europe to assess antiviral activity, safety and pharmacokinetics (PK) of once-daily (QD) regimens of TMC435 in HCV genotype-1 treatment-naive and treatment-experienced (prior non-responders/relapsers to IFN based therapy) patients (OPERA-1; Figure 3).
In the first cohort, treatment-naive patients were rand-omized to receive either:
-- 7 days of mono therapy of TMC435, 25 or 75 mg QD, or placebo as capsules followed by 21 days of triple therapy with TMC435 QD or placebo, pegylated interferon-_-2a (PEG-IFN_-2a; 180 _g subcutaneous once-weekly) and ribavirin (RBV; 1000-1200 mg daily) (Figure 2; Panel A).
-- or 28 days of triple therapy with TMC435 QD or · placebo, PEG-IFN_-2a and RBV (Figure 2; Panel B).
After day 28, patients continued on PEG-IFN_-2a/RBV for a total of 24 or 48 weeks, at the discretion of the Investigator.
Plasma concentrations of TMC435 and of RBV were evaluated during the first 4 weeks.
Analytical methods:
TMC435 and RBV were quantified in (EDTA) plasma using a validated LC-MS/MS method, with a lower limit of quantification of 2.00 ng/mL.
PK analyses were performed using WinNonLin (Pharsight).
RESULTS
Healthy volunteers:
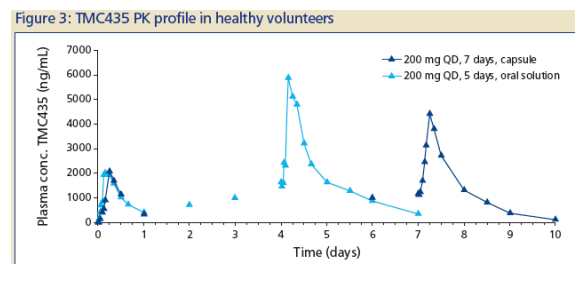
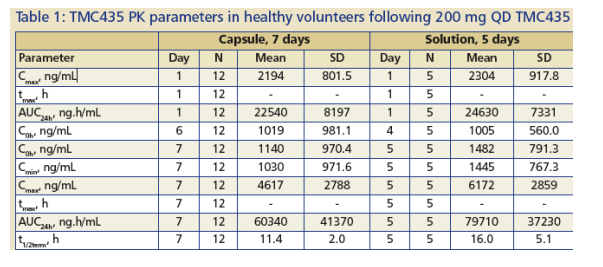
In healthy volunteers receiving 200 mg TMC435 QD for 7 days as · a capsule formulation, steady state PK was attained within 7 days (Figure 3; Table 1).
The overall PK profile in healthy volunteers for the capsule was similar to that for the oral solution, which was used in earlier 5-day trials with TMC435 in HCV-infected- and healthy volunteers(1,2), with a slightly lower overall bioavailability for TMC435 given as the capsule.
HCV-infected patients:
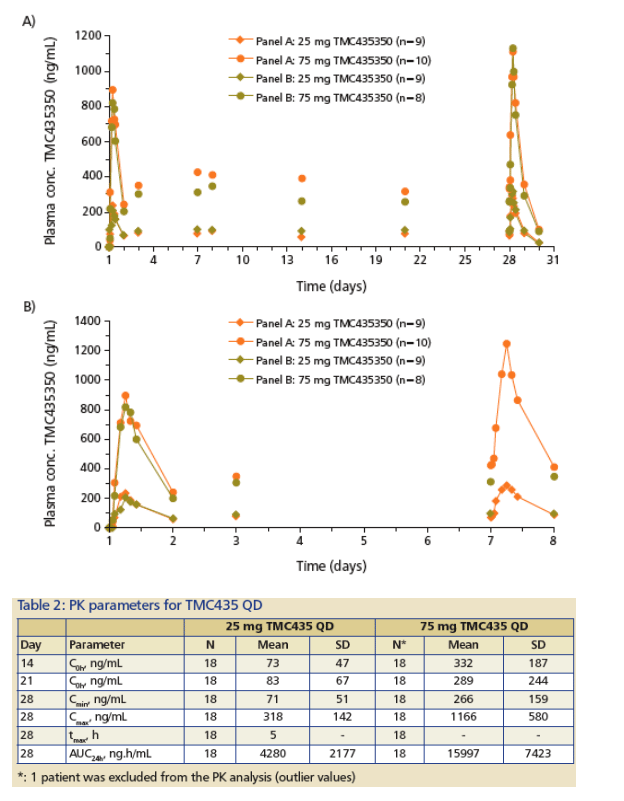
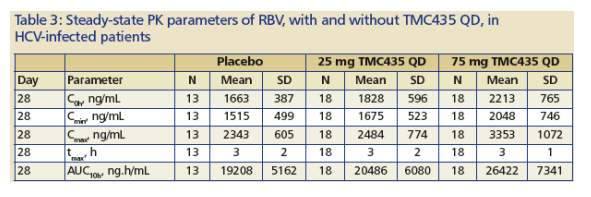
Results of 48 patients receiving QD regimens of 25 mg or 75 mg TMC435 (or placebo) as capsules (Figure 4; Table 2 and 3):
-- Details on antiviral activity and safety are presented in a separate poster by Manns et al.(3)
-- For both regimens, steady state conditions were attained within 3 days (Fig 4A), with plasma concentrations essentially proportional to the dose.
-- The mean steady-state Cmin (±
standard deviation) values were 71 ±
51 ng/mL for the 25 mg QD regimen, and 266 ±
159 ng/mL for the 75 mg QD regimen, 12 to 44-fold in excess of the EC50 based on plasma concentrations, and over 300-fold when taking into account the anticipated liver distribution extrapolated from animal studies.
-- No relevant differences were observed in TMC435 plasma levels with or without PEG-IFN_-2a and RBV (Fig 4B, Table 3), and no major differences were observed in RBV plasma concentrations with or without TMC435 (Table 3).
Figure 4: PK profile in HCV-infected patients, 7 days TMC435 QD mono therapy + 21 days combination with PEG-IFN_-2a/RBV (Panel A) vs. 28 days combination with PEG-IFN_-2a/RBV (Panel B). (A) 28-days; (B) 7 days.
REFERENCES
(1) G. Van 't Klooster et al., 43rd EASL, Milan, Italy, April 23-27, 2008, Poster 2590.
(2) R. Verloes et al., 58th AASLD, Boston, MA, November 2-6, 2007, Poster 1318.
(3) M. Manns et al., 59th AASLD, San Francisco, CA, October 31 - November 4, 2008, Poster LB08.
|
| |
|
 |
 |
|
|