 |
 |
 |
| |
Histologic Assessment of Long-term Entecavir Treatmentin Chronic
Hepatitis B Patients
|
| |
| |
Reported by Jules Levin
AASLD Liver Meeting Nov 4 2008 San Francisco, CA
Yoshiaki Katano1, Hiromitsu Kumada2, Haruhiko Kobashi3, Joji Toyota4, Osamu Yokosuka5, Koichi Takaguchi6, Masayoshi Kage7, Mitsuhiko Moriyama8, Fumio Imazeki5, Hiroki Ishikawa9, Taku Seriu10, Masao Omata11
1Department of Gastroenterology, Graduate School of Medicine, Nagoya University, Aichi, Japan; 2Department of Gastroenterology and Hepatology, Toranomon Hospital, Tokyo, Japan; 3Department of Gastroenterology and Hepatology, Okayama University Graduate School of Medicine, Dentistry, and Pharmaceutical Sciences, Okayama, Japan; 4Department of Gastroenterology, Sapporo Kosei General Hospital, Hokkaido, Japan; 5Department of Medicine and Clinical Oncology, Graduate School of Medicine, Chiba University, Chiba, Japan; 6Department of Internal Medicine, Kagawa Prefectural Central Hospital, Kagawa, Japan; 7Department of Pathology, School of Medicine, Kurume University, Fukuoka, Japan; 8Division of Gastroenterology and Hepatology,Department of Medicine, Nihon University School of Medicine, Tokyo, Japan; 9Research and Development, Bristol-Myers K.K., Tokyo, Japan; 10Research and Development, Bristol-Myers Squibb, Princeton, NJ, USA; 11Department of Gastroenterology, Graduate School of Medicine, University of Tokyo, Tokyo, Japan
Author Summary of Results
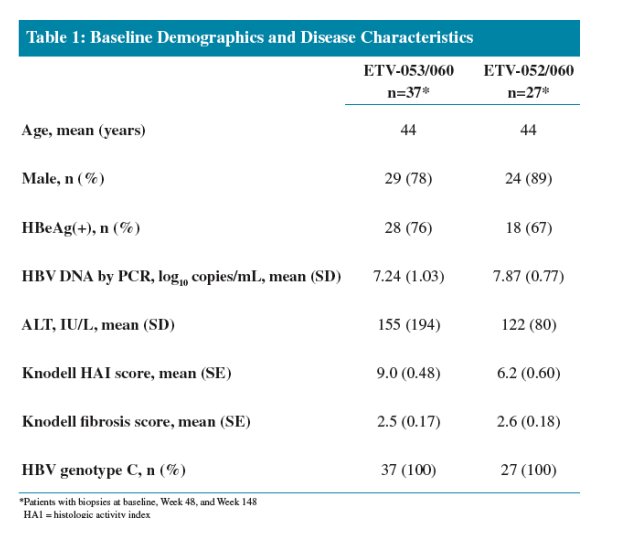
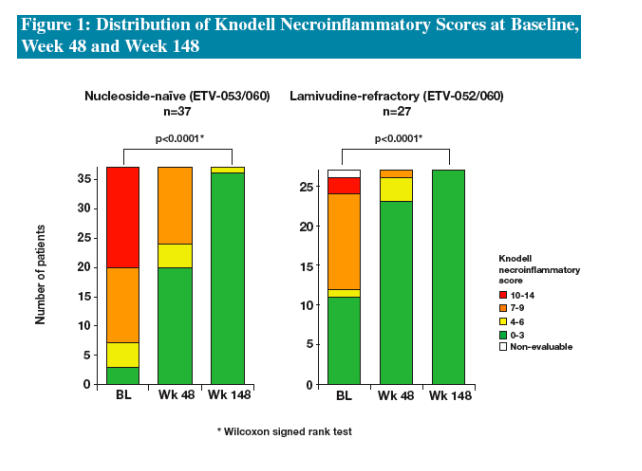
Three years of ETV treatment resulted in histologic improvement in 100% of nucleoside-naïve and 89% of LVDr patients
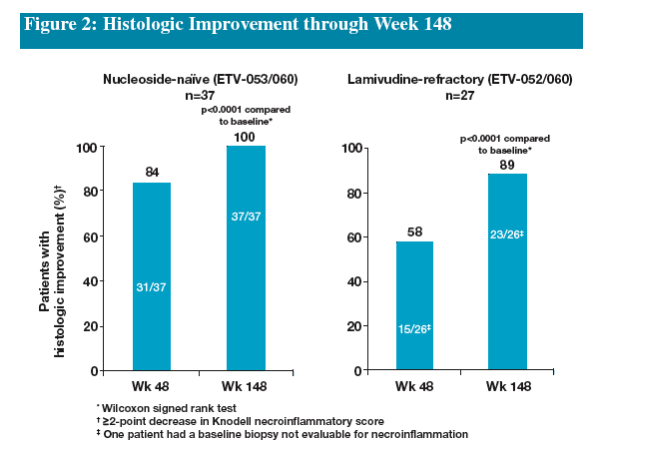
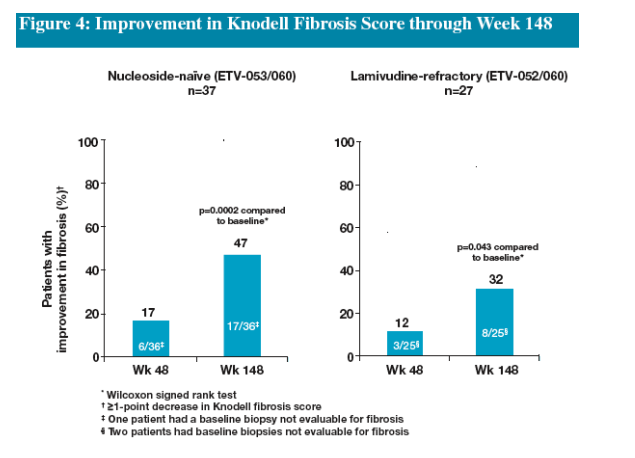
Treatment with ETV beyond 48 weeks resulted in further improvement in fibrosis scores in both naïve and LVDr patients
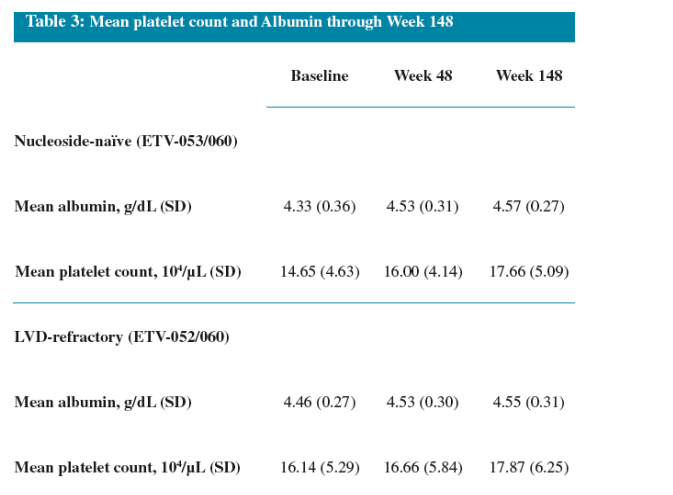
High proportions of both naïve and LVDr patients achieved HBV DNA suppression and ALT normalization during 3 years of ETV
Author Conclusion
The results from these cohorts demonstrate that long-term continuous treatment with ETV results in durable suppression of HBV replication and significant histologic improvement in nucleoside-naïve and LVDr patients
INTRODUCTION
In large observational studies, elevated baseline HBV DNA has been demonstrated to be a significant risk factor for cirrhosis and hepatocellular carcinoma1-3
HBV DNA suppression with antiviral therapy can significantly improve liver histology in HBeAg(+) patients4
Entecavir (ETV) demonstrated potent suppression of HBV DNA replication and improvement in liver histology in nucleoside-naïve and lamivudine-refractory (LVDr) Japanese patients with chronic hepatitis B (CHB) (studies ETV-053 and ETV-052)5,6
All patients who completed studies ETV-053 and ETV-052 could enroll in rollover study ETV-060
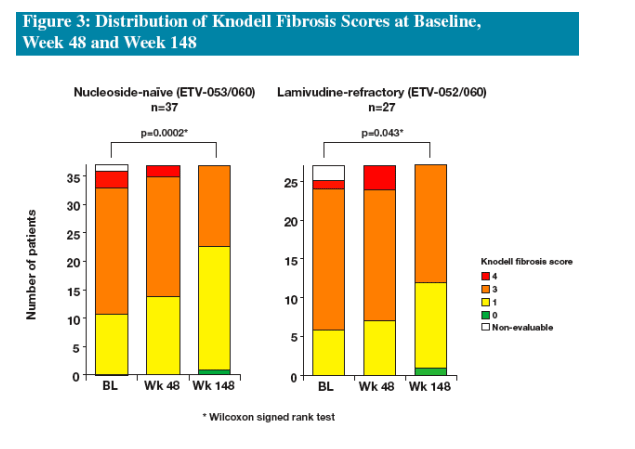
We present histologic results from nucleoside-naïve and LVDr patients who received at least 3 years of ETV therapy in studies ETV-052, ETV-053 and ETV-060
METHODS
Study population
The Long-term Histology Cohorts from Japan consist of patients who:
--were initially treated with ETV in studies ETV-053 or ETV-052
--subsequently enrolled in ETV-060
--had biopsies from three time points: baseline, Week 48 and Week 148

Eligibility criteria (ETV-053 and ETV-052)
-- CHB infection with compensated liver disease
-- HBV DNA ≥5 log10 copies/mL by PCR assay
-- ETV-053
· ≦12 weeks prior treatment with anti-HBV nucleoside analogues
--ETV-052
· ≥24 weeks prior lamividine therapy, ongoing at the time of randomization; or
· documented evidence of infection with HBV-carrying LVDr substitutions
-- ALT 1.3-10 x ULN
-- HBeAg(+) or HBeAg(-)
ETV-060
--Enrollment immediately after completion of ETV-053 or ETV-052 with no gap in dosing
--Patients enrolling from ETV-053: ETV 0.5 mg/day for a total of up to 148 weeks (3 years) of ETV treatment
--Patients enrolling from ETV-052: ETV 1.0 mg/day for a total of up to 148 weeks (3 years) of ETV treatment
Efficacy and resistance analyses of the Long-term Histology Cohorts from Japan
Efficacy assessments at Week 48 (1 year) and Week 148 (3 years) included proportions of patients with:
--histologic improvement (≥2-point decrease in Knodell necroinflam-matory score)
--improvement in fibrosis (≥1-point decrease in Knodell fibrosis score)
- detectable HBV DNA by PCR
--ALT normalization (ALT ≦1 x ULN)
Resistance
--Paired samples from baseline and all patients with HBV DNA ≥400 copies/mL at Week 148 (or last on-treatment measurement for patients discontinuing prior to Week 148) were analyzed for substitutions associated with ETV resistance
--All patients with virologic breakthrough (≥1 log10 increase from nadir on two consecutive measurements) were also genotyped
RESULTS


REFERENCES
1. Chen CJ, Yang HI, Su J, et al. JAMA 2006;295:65-73.
2. Iloeje UH, Yang HI, Su J, et al. Gastroenterology 2006;130:678-686.
3. Chen G, Lin W, Shen F, et al. Am J Gastroenterol 2006;101:1797-1803.
4. Mommeja-Marin H, Mondou E, Blum MR, et al. Hepatology 2003;37:1309-1319.
5. Kobashi H, Takaguchi K, Ikeda H, et al. J Gastroenterol Hepatol 2008. In press
6. Suzuki F, Toyoda J, Katano Y, et al. J Gastroenterol Hepatol 2008;23:1320-26.
|
| |
|
 |
 |
|
|