 |
 |
 |
| |
Evaluation of Long-term Entecavir Treatment in Stable Chronic Hepatitis B Patients Switched from Lamivudine Therapy
|
| |
| |
Reported by Jules Levin
AASLD Nov 4 2008, San Francisco, CA
Tatsuya Ide1, Michio Sata1, Michiko Shindo2, Kazuaki Chayama3, Joji Toyota4, Satoshi Mochida5, Eiichi Tomita6, Hiroshi Yatsuhashi7, Norio Hayashi8, Hiroki Ishikawa9, Taku Seriu10, Masao Omata11
1Division of Gastroenterology, Department of Medicine, Kurume University School of Medicine, Fukuoka, Japan; 2Division of Liver Disease, Department of Internal Medicine, Akashi Municipal Hospital, Hyogo, Japan; 3Department of Medicine and Molecular Science, Graduate School of Biomedical Sciences, Hiroshima University, Hiroshima, Japan; 4Department of Gastroenterology, Sapporo Kosei General Hospital, Hokkaido, Japan; 5Department of Gastroenterology and Hepatology, Saitama Medical University, Saitama, Japan; 6Department of Gastroenterology, Gifu Municipal Hospital, Gifu, Japan; 7Clinical Research Center, National Hospital Organization Nagasaki Medical Center, Nagasaki, Japan; 8Department of Gastroenterology and Hepatology, Osaka University Graduate School of Medicine,Osaka, Japan; 9Research and Development, Bristol-Myers K.K., Tokyo, Japan; 10Research and Development, Bristol-Myers Squibb, Princeton, NJ, USA; 11Department of Gastroenterology, Graduate School of Medicine, University of Tokyo, Tokyo, Japan
Author Summary of Results
Switching CHB patients from LVD to long-term ETV therapy
resulted in the following:
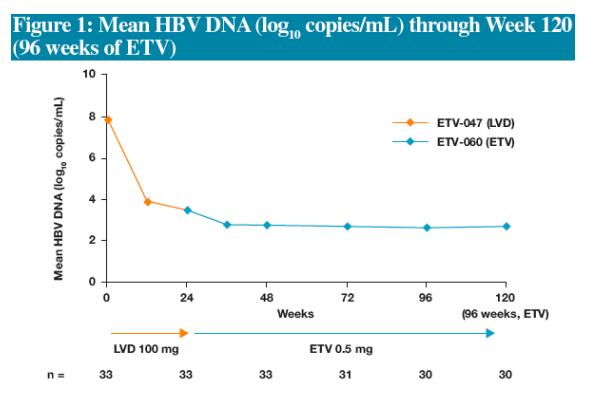
Additional suppression of HBV DNA replication
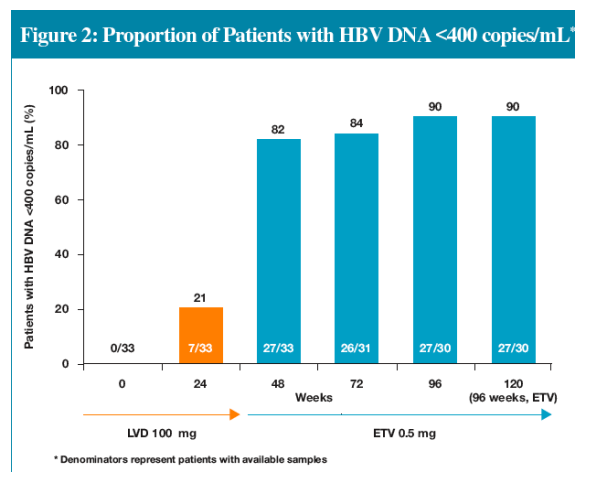
-- Proportion of patients with HBV DNA <400 copies/mL increased from 21% to 90% after 72 weeks of ETV and was maintained through 96 weeks of ETV
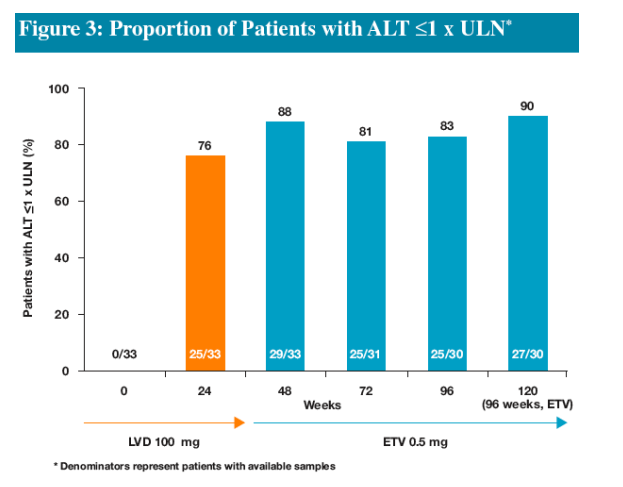
-- Increasing proportions of patients achieving ALT ≦1 x ULN
No evidence of resistance emergence during 96 weeks of ETV treatment
ETV was well tolerated during long-term treatment
AUTHOR CONCLUSION
CHB patients switched from LVD to long-term ETV achieve increased rates of virologic suppression, with no evidence of resistance through 2 years of ETV treatment
INTRODUCTION
The goal of chronic hepatitis B (CHB) treatment is to achieve sustained suppression of HBV DNA and remission ofliver disease1
Long-term treatment of CHB patients with lamivudine (LVD) is associated with the development of resistance and loss ofclinical benefit2
Current Japanese CHB treatment guidelines recommend that patients should be switched to entecavir (ETV) 0.5 mg daily if they have received less than 3 years of LVD therapy, have HBV DNA <400 copies/mL and no breakthrough hepatitis orYMDD mutations
ETV 0.5 mg daily for 24 weeks demonstrated superior HBV DNA reduction compared to LVD 100 mg daily in phase 2 studyETV-047 in Japan3
After completing ETV-047, all patients could enroll inopen-label ETV rollover study ETV-060
We report long-term efficacy, safety and resistance for patients who were switched from LVD to ETV therapy
METHODS
Study Population
Thirty-four patients in ETV-047 received LVD 100 mg daily
Thirty-three LVD-treated patients from ETV-047 enteredETV-060 and received ETV 0.5 mg daily
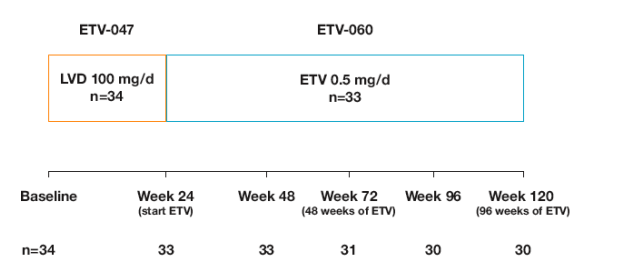
Eligibility criteria (study ETV-047):
- CHB infection with compensated liver disease
- HBV DNA ≥7.6 log10 copies/mL by PCR assay
- ≦12 weeks of prior treatment with anti-HBVnucleoside analogues
- Alanine transaminase (ALT) 1.25-10 x ULN
- HBeAg(+) or HBeAg(-)
Analyses through Week 120 (96 weeks of ETV)
Efficacy
Efficacy assessments evaluated proportions of patients who had available samples (Non-completer=Missing) at Weeks 24, 48, 72, 96 and 120 for the following parameters:
-HBV DNA by PCR assay
-ALT normalization (ALT ≦1.0 x ULN)
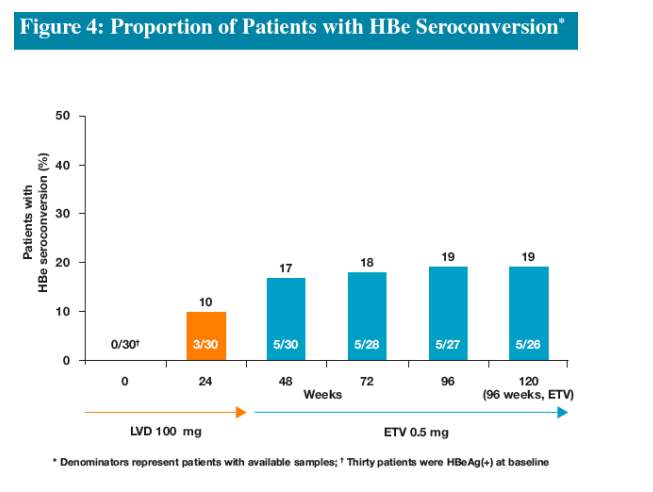
-HBe seroconversion among HBeAg(+) patients
Resistance
Paired samples from all patients with HBV DNA ≥400 copies/mL at Week 96 (72 weeks of ETV), Week 120 (96 weeks of ETV) or last on-treatment measurement (for patients discontinuing prior to Week 120) were analyzed for substitutions associated with ETV or LVD resistance
All patients with virologic breakthrough (≥1 log10 increase from nadir on two consecutive measurements) werealso genotyped
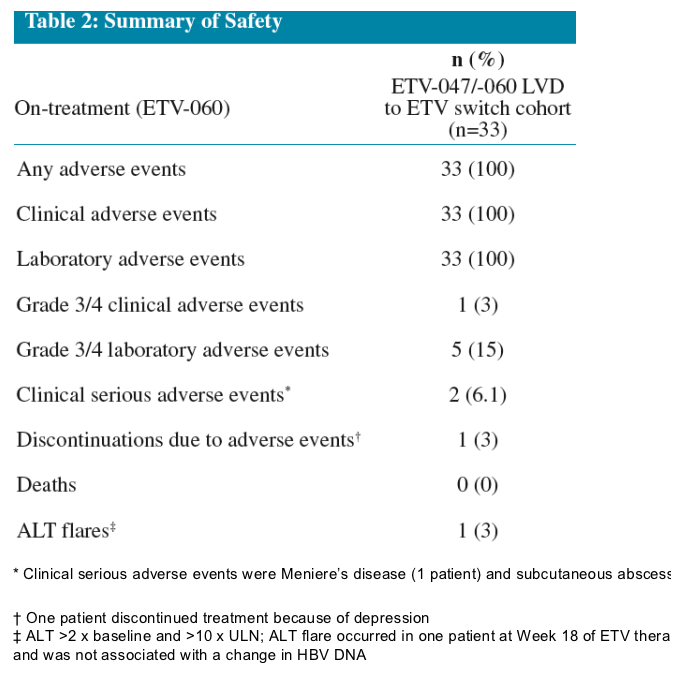
Safety
Safety was assessed throughout the treatment period
Results

Resistance
Of 33 treated patients, 4 had HBV DNA ≥400 copies/mLduring ETV-060:
One patient discontinued at Week 68 (44 weeks of ETV)
-- Testing of isolates revealed no substitutions associated with ETV resistance
Three patients had HBV DNA ≥400 copies/mL at Week 120 (96 weeks of ETV)
-- Two of three had samples available for resistance testing
-- Neither (0/2) demonstrated substitutions associated with ETV resistance
Safety

REFERENCES
1. Lok AS and McMahon BJ. Hepatology 2007;45:507-39
2. Lai CL, Dienstag J, Schiff E, et al. Clin Infect Dis 2003;36:687-96
3. Shindo M, Chayama K, Toyota J, et al. J Clin Virol 2006;363. (Suppl 2):S94.
|
| |
|
 |
 |
|
|