 |
 |
 |
| |
SVR in African American Patients:
Results of the IDEAL Study
(Individualized Dosing Efficacy vs
Flat Dosing to Assess OptimaL Pegylated Interferon Therapy)
|
| |
| |
Reported by Jules Levin
American Association for the Study of Liver Diseases
The Liver Meeting 2008
San Francisco, California
October 31 - November 4, 2008
J McCone, K-Q Hu, JG McHutchison, ML Shiffman, J King, SK Herrine, GW Galler, MS Sulkowski, J Bloomer, FA Nunes, KA Brown, SC Gordon, KD Mullen,
N Ravendhran, W Cassidy, R Ghalib, W Deng, S Noviello, JK Albrecht, AJ Muir
on Behalf of the IDEAL Study Team
Author Conclusions
As in previous studies, African American patients had lower SVR rates than non-African American patients
SVR rates:
-- Low baseline viral load > high baseline viral load
-- Fasting glucose <100 mg/dl
-- Baseline Hemoglobin
-- PEG-IFN alfa-2a + RBV > low-dose PEG-IFN alfa-2b 1.0 + RBV
-- PEG-IFN alfa-2b 1.5 + RBV = PEG-IFN alfa-2a + RBV
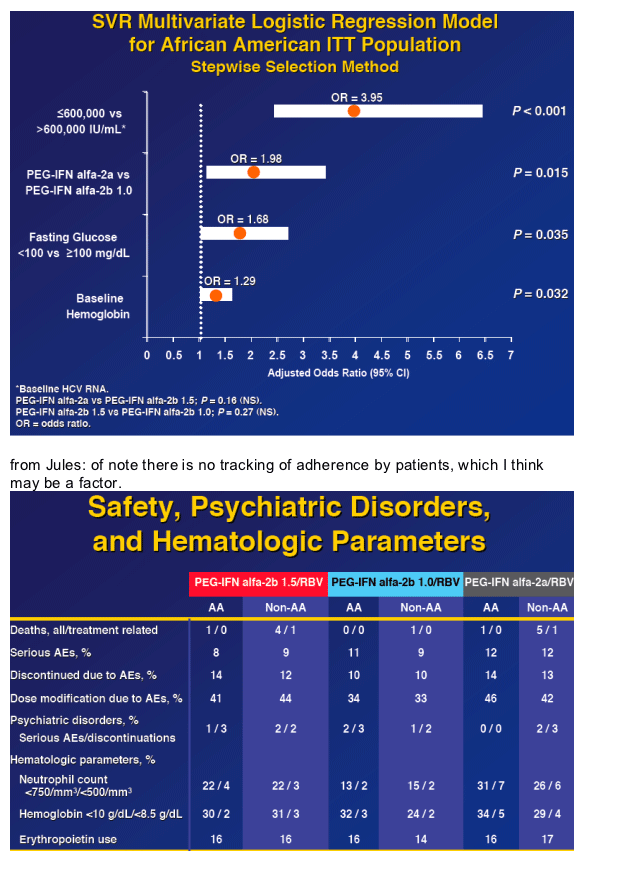
(from Jules: low baseline VL, baseline fasting glucose <100 mg/dl, and baseline hemoglobin were predictive of SVR rate for AAs).
Early response in African American patients was an important factor in predicting SVR in each of the 3 treatment arms (from Jules: also of interest is that AAs had relatively high undetectable viral load rates compared to non-AAs at weeks 2 and 4 which fell off relative to non-AAs by end-of-treatment; see comments and slides below)
Safety and tolerability were similar among African American patients and non-African American patients in the 3 treatment groups

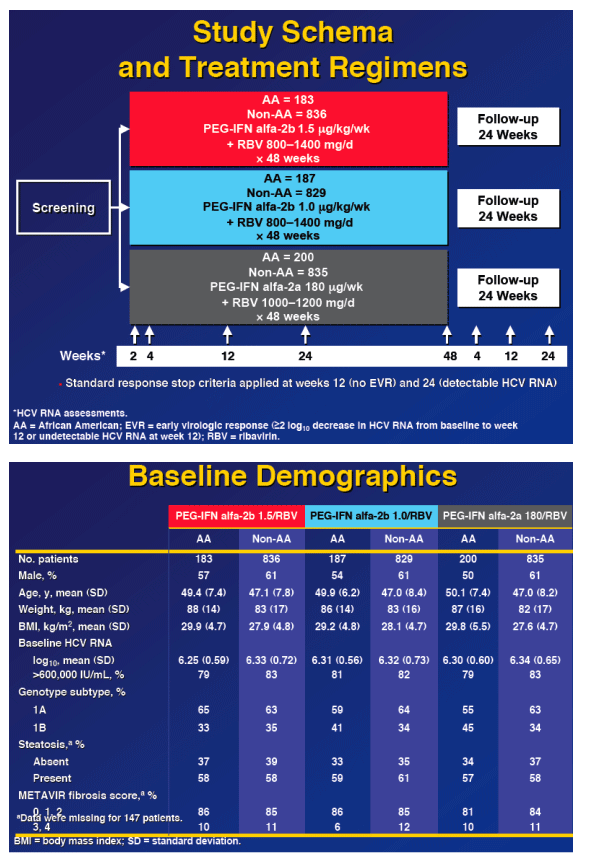
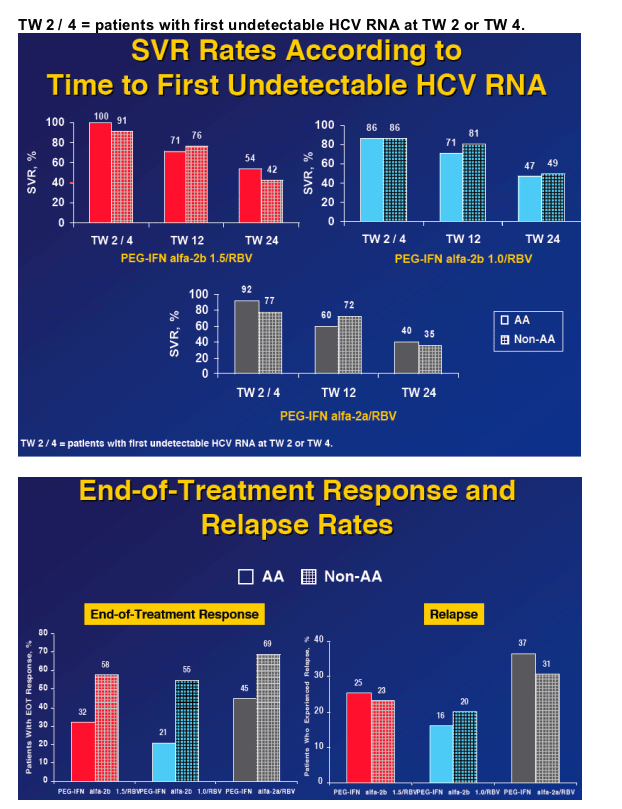
from Jules: you'll notice in the following slides SVR was higher by 3 percentage points with PEG-IFN alfa-a for AAs (26 vs 23%), but at the end of 24 weeks in 2nd slide below PEG-IFN alfa-2b had higher rates of undetectable HCV RNA 54% in PEG-IFN alfa-2b 1.5/RBV, 47% with PEG-IFN alfa 2b 1.0/RBV, and 40% with PEG-IFN alfa-2b/RBV. But, end of treatment responses for AAs were different: 32% for Peg-2b 1.5/RBV, 21% for Peg-2b 1.0/RBV, and 45% for Peg-2a/RBV. And, relapse rate was higher in Peg-2a group: 37% in Peg-2a, 25% for Peg-2b 1,5, and 16% for Peg-2b 1.0.
Also noteworthy is although SVR rates for non-AAs are similar for all 3 Peg/RBV treatment groups, at week 24 Peg-2a rates of undetectable are lower compared to the 2 Peg2b arms, but at the end of treatment Peg-2a SVR rate appears higher for Peg-2a than for the 2 Peg-2b arms, despite that relapse rate is higher. So what happened between weeks 24 to 48?
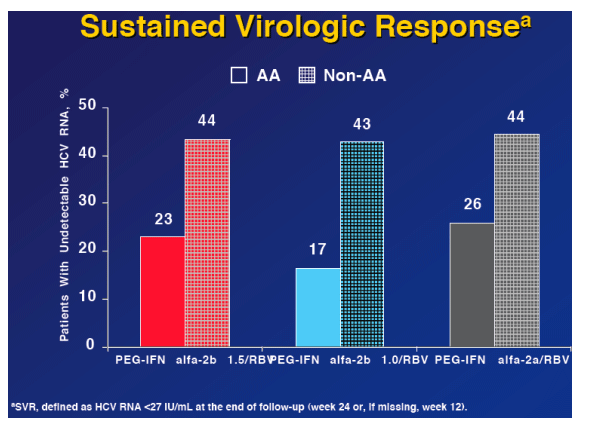
from Jules: you will note that AAs had high rates of viral response (undetectable) at weeks 2 and 4 and relatively performed well also at weeks 12 and 24. But by the end-of-treatment rates of response fell off for AAs and their relapse rate appeared to be higher compared to non-AAs in the Peg-2a group (37% vs 31%).
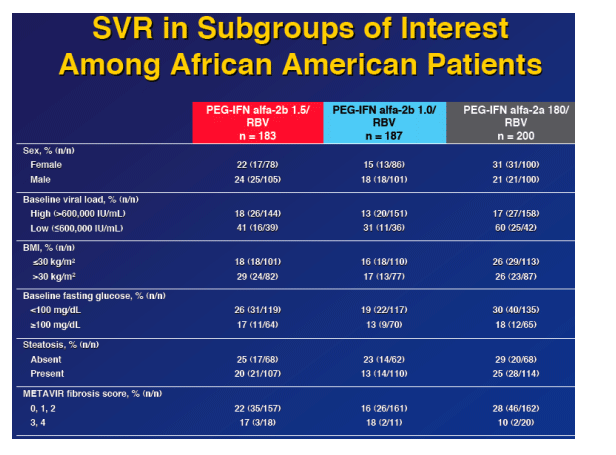
In the multivariate anaysis baseline viral load, baseline fasting glucose, and baseline hemoglobin predict SVR in AAs.
from Jules: of interest in this slide is - in patients with low baseline viral load (<600,000 IU/ml) Peg-2a had a higher SVR rate compared to Peg-2b 1.5 (60% vs 41%), but in high baseline viral loads SVR rates were the same 18% for Peg-2b 1.5 vs 17% for Peg-2a. Similar results is seen for BMI: for patients with low baseline BMI (<30 kg/m2) SVR rates is 26% for Peg-2a and 18% for Peg 2b 1.5, but for high baseline BMI (>30 kg/m2) SVR rates were 29% for Peg-2b 1.5 and 26% for Peg-2a. Similarly or patients regarding METAVIR fibrosis score, patients with fibrosis scores of 0, 1, or 2 SVR rate for Peg-2b 1.5 was 22% and for Peg-2a it was 28%, but for patients with fibrosis scores of 3 or 4 SVR rate was 17% for Peg-2b 1,5 and 10% for Peg-2a. The numbers of patients get small in the latter group. Below is the multivariate analysis.
|
| |
|
 |
 |
|
|