 |
 |
 |
| |
Metformin with Peginterferon Alfa-2a and Ribavirin in the Treatment of Naive Genotype 1 Chronic Hepatitis C Patients with Insulin Resistance (TRIC-1): Final Results of a Randomized and Double-Blinded Trial
|
| |
| |
Reported by Jules Levin
AASLD Nov 4 2008, San Francisco, CA
M. Romero-Gomez, M. Diago, RJ Andrade, JL Calleja, J. Salmeron, CM Fernandez-Rodriguez, R. Solà, JM. Herrerias, J. Garcia-Samaniego, R. Moreno-Otero, A. Olveira, O. Núñez, M de la Mata, F. Jorquera, RM Morillas, B. Dalmau, R. Martin-Vivaldi, JI Arenas-Ruiz, E. Rodriguez, S. Duran, P. Giner.
Unidad de Gestion Clinica de Enfermedades Digestivas y CIBEREHD, Hospital Universitario de Valme, Sevilla. On behalf of the TRIC-1 Spanish group.
AUTHOR CONCLUSIONS
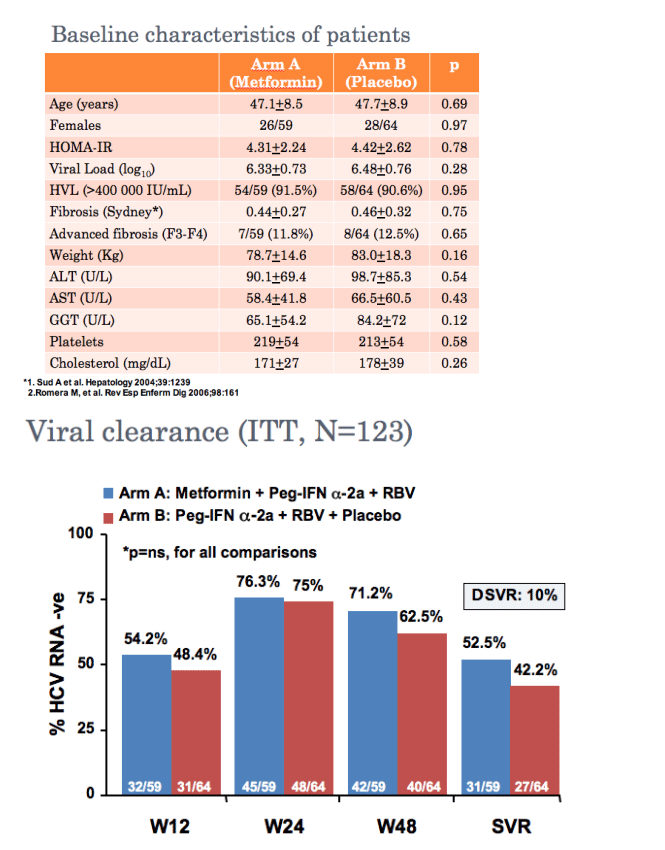
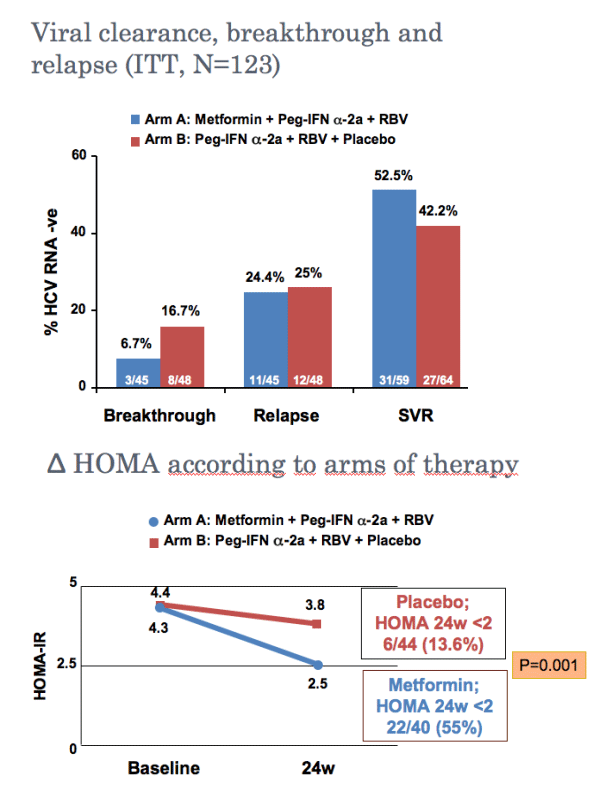
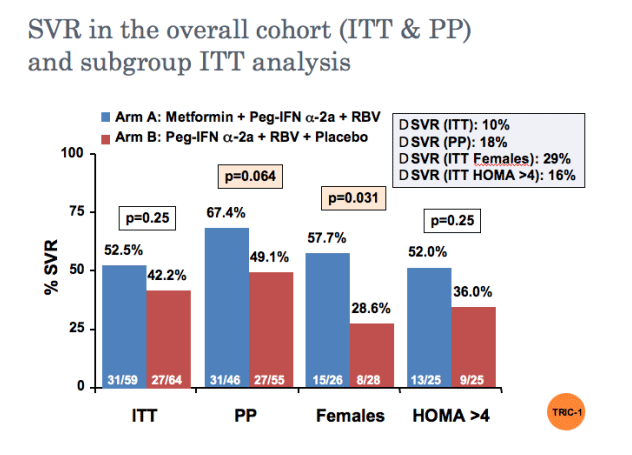
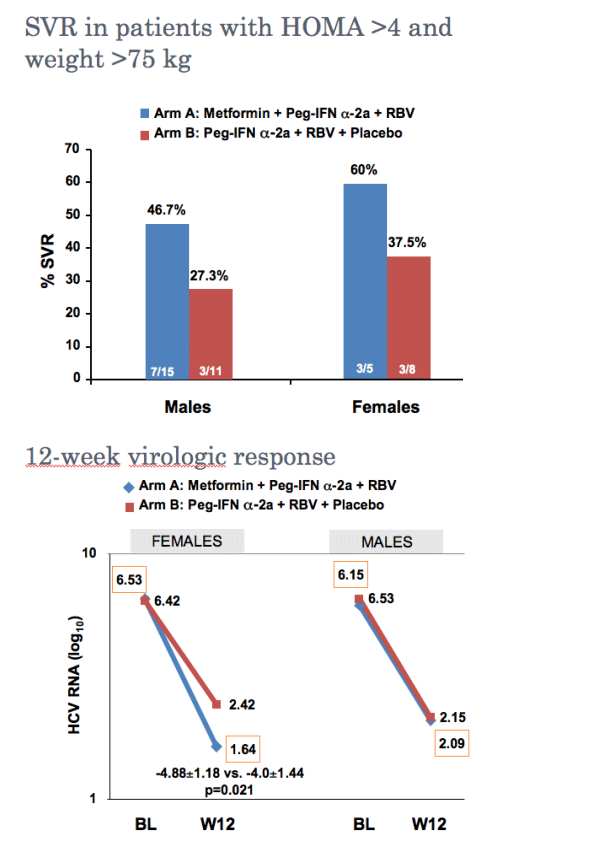
Metformin-based triple therapy is safe, improves insulin sensitivity and increases sustained virological response rate by 10% in patients with hepatitis C genotype 1 and insulin resistance (HOMA >2). This therapy was especially effective in females in whom metformin raised significantly the SVR rate.
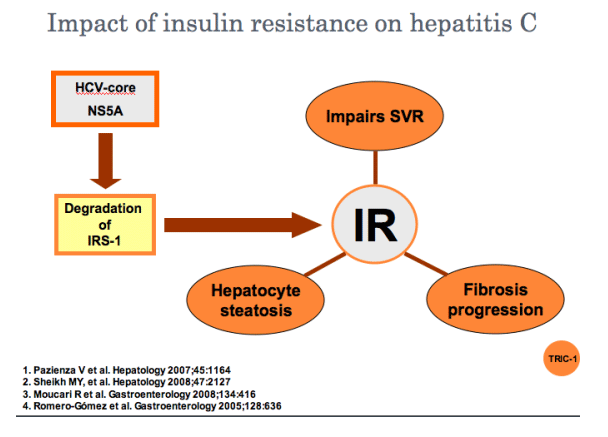
Insulin resistance seems to be a new target in the management of hepatitis C
INTRODUCTION
Metabolic abnormalities such as altered glucose homeostasis is not uncommon in patients with HCV
Patients with chronic hepatitis C have a significantly increased risk of diabetes mellitus compared with non-infected controls and patients infected with HBV
HCV eradication restores glucose homeostasis in many patients.
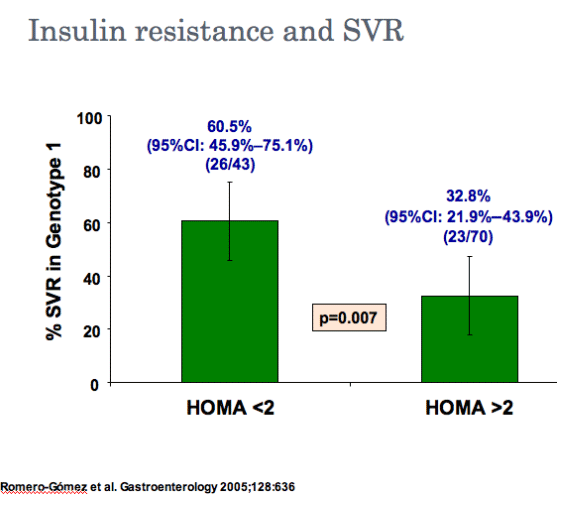
SVR rates are significantly lower in patients with insulin resistance or diabetes
It remains to be determined whether restoration of insulin sensitivity before or during treatment, can increase SVR rates
TRIC-1 study: aim
To determine if the addition of metformin to the standard of care could improve SVR in treatment naive patients with genotype 1 chronic hepatitis C and insulin resistance
TRIC-1 study: design
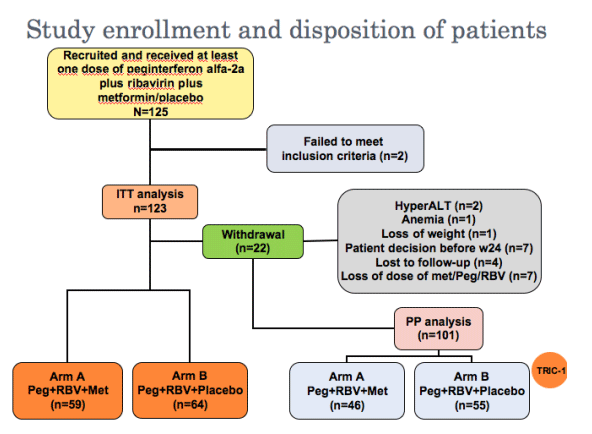
Prospective, multicenter, randomized, double-blinded, placebo-controlled trial
-- Sample size estimation was based on the assumption that metformin would improve insulin resistance and would increase the SVR rate by 28%
Estimated sample size was 124 patients with a 10% rate of loss to follow-up, type I error of 0.05 and type II of 0.2 (Fisher exact two-sided test)

TRIC-1 study: endpoints
Primary end-point: sustained virologic response (SVR) defined as viral clearance (HCV RNA
<10 IU/mL) 24 weeks after end of therapy
Secondary end-points:
--Viral clearance (HCV RNA <10 IU/mL) at Week 12, 24 and 48
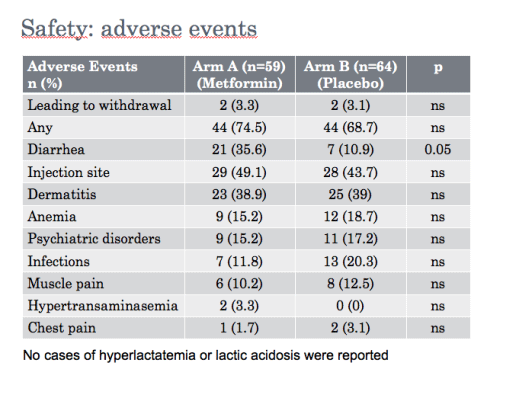
--Changes in HOMA during the first 24 weeks Safety

|
| |
|
 |
 |
|
|