 |
 |
 |
| |
A Phase 1 Study of the Safety, Tolerability, and Pharmacokinetics of Single Ascending Oral Doses of the NS3/4A Protease Inhibitor ITMN-191 in Healthy Subjects
|
| |
| |
Reported by Jules Levin
AASLD Nov 4 2008 San Francisco, CA
B. Bradford1 , C. Rubino2, S. Porter1, A. Forrest2, L. Blatt1, A. Patat3
1InterMune, Inc., Brisbane, CA ; 2ICPD/Ordway Research Institute, Inc., Albany, NY, SUNY School of Pharmacy, Buffalo, NY; 3 Biotrial, Rennes, France
BACKGROUND & AIMS
ITMN-191 is a highly potent and highly selective macrocyclic inhibitor of the HCV NS3/4A serine protease that achieves high liver concentrations with only modest systemic exposure. Preclinical studies demonstrate an EC50 of 1.8 nM in an HCV genotype 1 replicon assay and liver:plasma concentration ratios of 10:1 and
120:1 in rats and primates, respectively. The aim of the present study was to evaluate the safety, tolerability, and pharmacokinetics (PK) of single ascending oral doses of ITMN-191 and examine the effect of food on ITMN-191 plasma PK in healthy adults.
AUTHOR SUMMARY & CONCLUSIONS
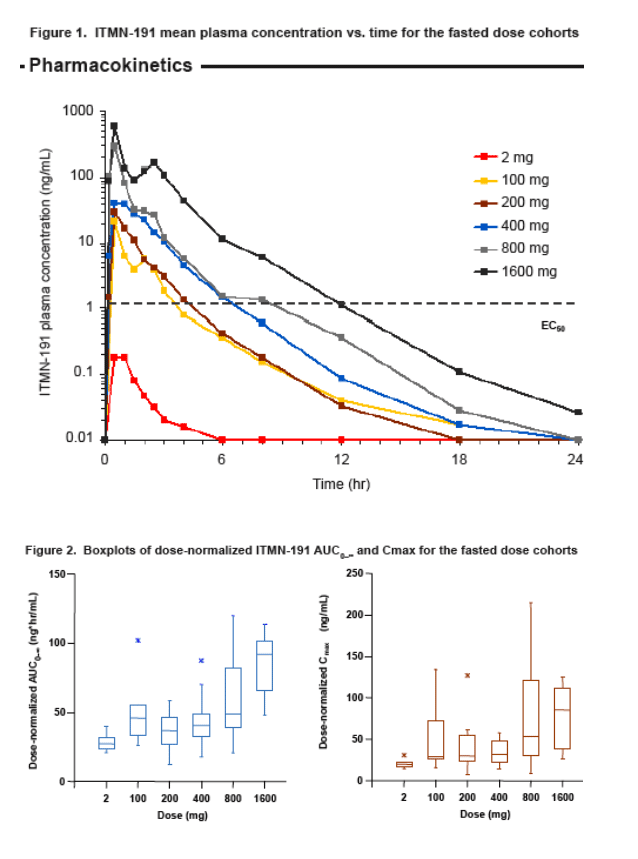
ITMN-191 was safe and generally well tolerated over the range of studied doses. Adverse events were generally mild and transient with no evidence of clinically significant laboratory abnormalities or ECG changes
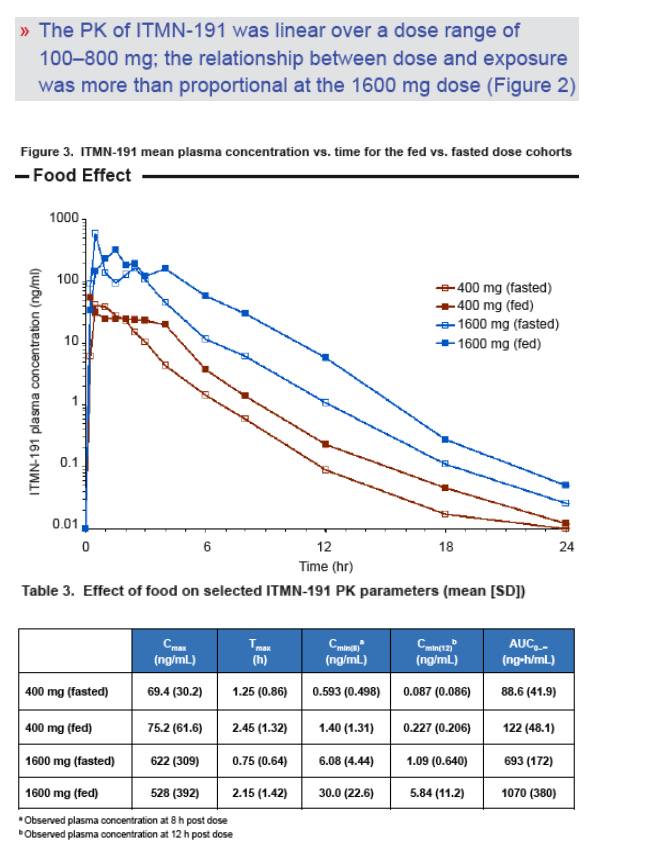
The pharmacokinetics of ITMN-191 appear linear over a dose range of 100-800 mg; the relationship between dose and exposure is more than proportional at the 1600 mg dose
Co-administration of ITMN-191 with food results in a significant increase in bioavailability; concomitant food intake is recommended
The results of the present study support the continued clinical development of ITMN-191. Subsequent studies to assess the safety and antiviral activity of multiple ascending doses of ITMN-191 in patients with chronic HCV are currently underway
METHODS
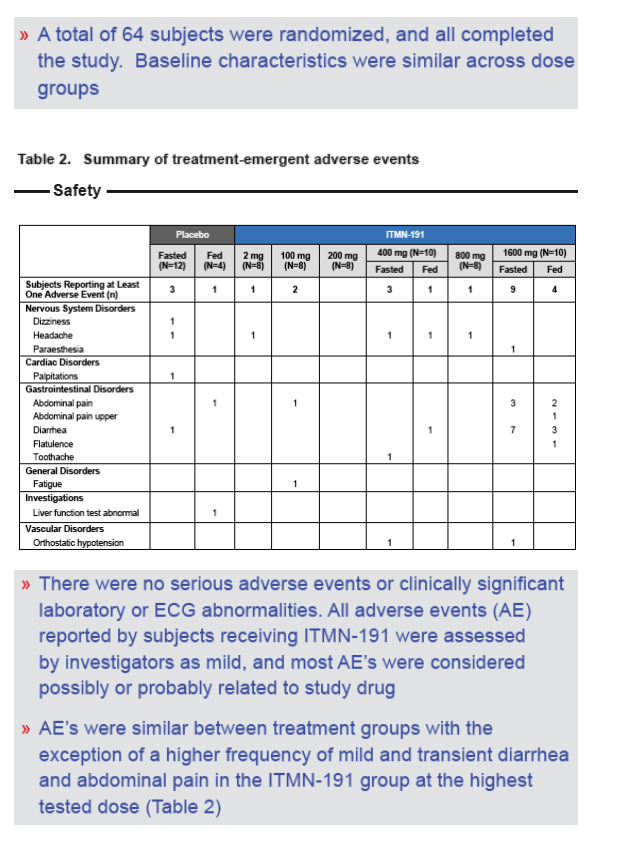
⇒ In a double-blind, placebo-controlled, first-in-man study, healthy adult subjects were randomized to receive a single oral dose of ITMN-191 or matched placebo in a Phase 1 research facility
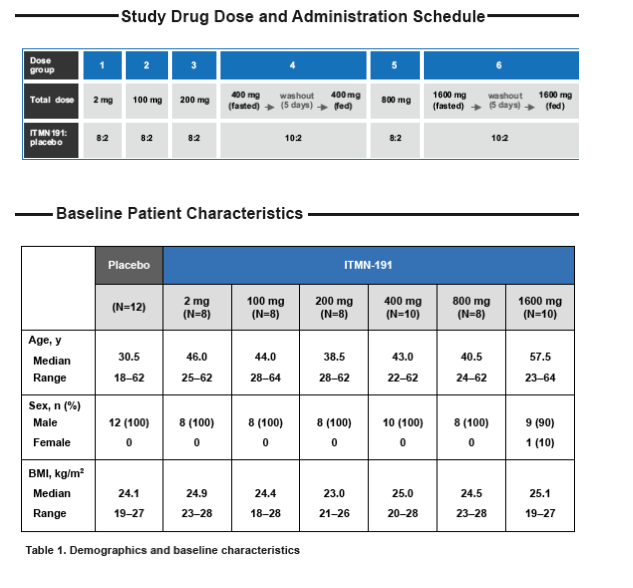
⇒ Study drug was administered orally in doses ranging from 2-1600 mg
⇒ In each of 4 cohorts, 8 and 2 subjects received ITMN-191 or placebo, respectively. In order to assess the effect of food on ITMN-191 PK, an additional 2 cohorts of 12 subjects each received ITMN-191 or placebo (10:2) in both the fed and fasted states, separated by a 5-day washout period
⇒ Plasma samples for PK analysis were collected 1 hr before dosing and at 0.25, 0.5, 1, 1.5, 2, 2.5, 3, 4, 6, 8, 12, 18, 24, and 32 hrs after dosing
⇒ PK parameter estimates were calculated using non-compartmental (slope-height-area-moment analysis) methods
⇒ PK modeling using compartmental analysis was performed to simulate plasma concentration profiles at steady state

|
| |
|
 |
 |
|
|