 |
 |
 |
| |
Kinetics Of HBsAg Decline in Patients With HBeAg-Negative Chronic Hepatitis B Treated With Peginterferon alfa-2a According to Genotype and its Association With Sustained HBsAg Clearance 4 Years Post Treatment
|
| |
| |
Reported by Jules Levin
59th Annual Meeting of the American Association for the Study of Liver Diseases, 31 October- 4 November 2008, San Francisco, USA
MR Brunetto,1 F Bonino,2 P Marcellin,3 P Button,4 R Batrla5
1UO Epatologia, Azienda Ospedaliero Universitaria Pisana, Pisa, Cisanello, Italy; 2Direzione Scientifica, Policlinico di Milano, IRCCS, Milano and Universita di Pisa, Pisa, Italy; 3Service d'Hépatologie, Hôpital Beaujon, Clichy, France;
4Roche Products Pty Limited, Dee Why, NSW, Australia; 5Hoffmann-La Roche Ltd, Basel, Switzerland
Background
HBsAg clearance/seroconversion (loss of HBsAg and appearance of anti-HBs) can be considered the closest we can achieve to clinical cure of chronic hepatitis B infection
Interferon-based therapy can induce HBsAg clearance/seroconversion and there is an indication that quantitative HBsAg analysis may be a useful marker of the patient's response to therapy1
A large-scale, phase 3 study in HBeAg-negative chronic hepatitis B, showed that a 48-week course of peginterferon alfa-2a ± lamivudine, was superior to lamivudine monotherapy for all response parameters assessed 6 months post treatment, including HBsAg clearance2
In that study HBsAg clearance was observed across all the major HBV genotypes A, B, C and D
End of treatment HBsAg level was associated with durable off-therapy response to peginterferon alfa-2a3
4 years post-treatment with peginterferon alfa-2a ± lamivudine HBsAg was cleared in 11% of patients (vs 2% with lamivudine; P=0.021) and suppression of HBV DNA to <400 copies/mL was achieved in 17% (vs 7% with lamivudine; P=0.042)4
OBJECTIVES
To analyze baseline levels of HBsAg and kinetics of HBsAg decline according to genotype in patients treated with a 48-week course of peginterferon alfa-2a ± lamivudine
To analyze HBsAg decline in patients with and without HBsAg clearance 4 years post treatment
AUTHOR SUMMARY
Baseline HBsAg levels varied across genotypes. Highest levels were observed in patients with genotypes A and D
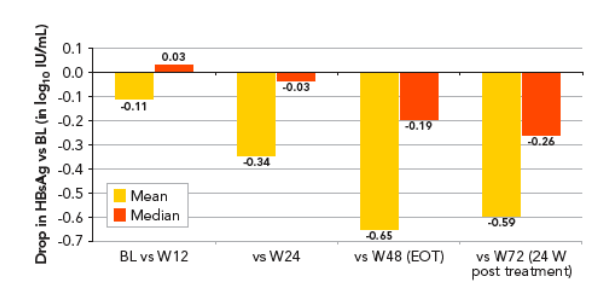
By end of treatment the overall mean (median) decline in HBsAg was -0.65 (-0.19) log10 IU/mL
The overall kinetics of HBsAg decline appeared to be genotype dependent with the most rapid and pronounced decline experienced by patients infected with HBV genotype B and the slowest decline in patients infected with HBV genotype D
HBsAg levels during treatment declined rapidly and to a far greater extent in patients who subsequently cleared HBsAg by 4 years post treatment compared with those who did not
-- Mean (median) decline from baseline to week 48 was -1.86 (-1.38) log10 IU/mL
vs -0.37 (-0.12) log10 IU/mL
In patients who cleared HBsAg, levels of HBV DNA declined rapidly and remained suppressed throughout treatment and beyond
AUTHOR CONCLUSION
On-treatment decline of HBsAg appears to be genotype dependent with the greatest decrease being observed in patients with genotype B. Rapid early on-treatment decline of HBsAg during peginterferon alfa-2a ± lamivudine was associated with HBsAg clearance 4 years post treatment. Quantification of early HBsAg level may help to identify which patients are most likely to achieve posttreatment cure (HBsAg loss) following peginterferon alfa-2a-based therapy
METHODS
This retrospective analysis of HBsAg levels was based on a subset of patients with available sera included in a large randomized trial of peginterferon alfa-2a who received peginterferon alfa-2a alone or combined with lamivudine for 48 weeks2
All participating centers in the initial study were invited to participate in a longterm observational follow-up study, in which patients were followed for up to 4 years after completing treatment. For a subset of the patients in this HBsAg analysis 4-year post-treatment follow up data were available. For the purpose of this analysis, patients completing the 4-year follow-up phase were classified as either HBsAg responders (with HBsAg clearance) or HBsAg non-responders (no HBsAg clearance)
Quantitative HBsAg in serum was measured retrospectively in available sera at baseline, on-treatment (weeks 12, 24, 48) and 24 weeks post treatment (week 72) using the Architect HBsAg assay (Abbott Diagnostics) Shown here are analyses for a total of 86 patients with available data at each of the time points
RESULTS
Baseline HBsAg levels and decline in HBsAg over time
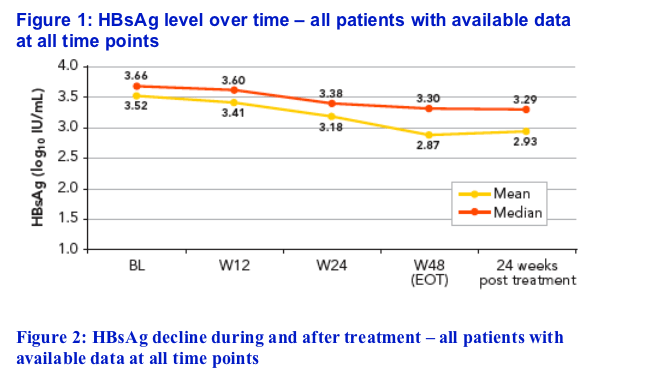
The mean (median) baseline HBsAg level for the 86 patients was 3.52 (3.66) log10 IU/mL
The HBsAg level over time is shown in Figure 1, with decline from baseline shown in Figure 2
Decline in HBsAg according to genotype
The HBV genotype distribution of the 86 patients was as follows:
-- Genotype A, n= 16; genotype B, n= 7; genotype C, n=20; genotype D, n=38
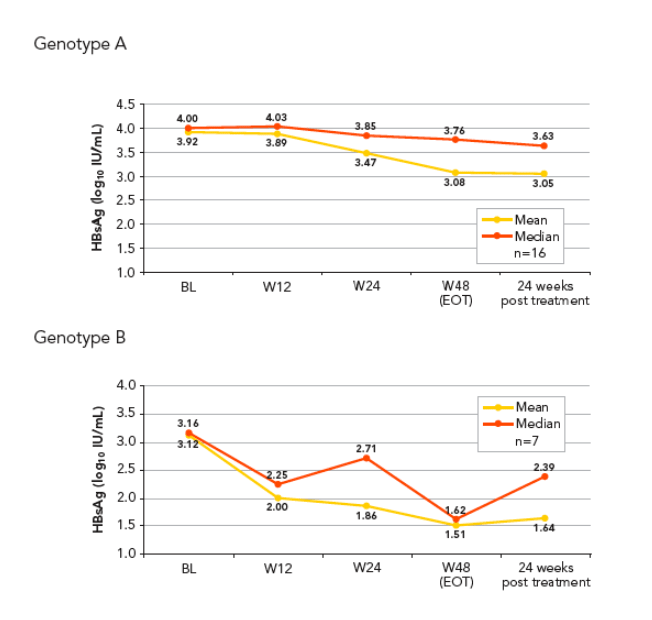
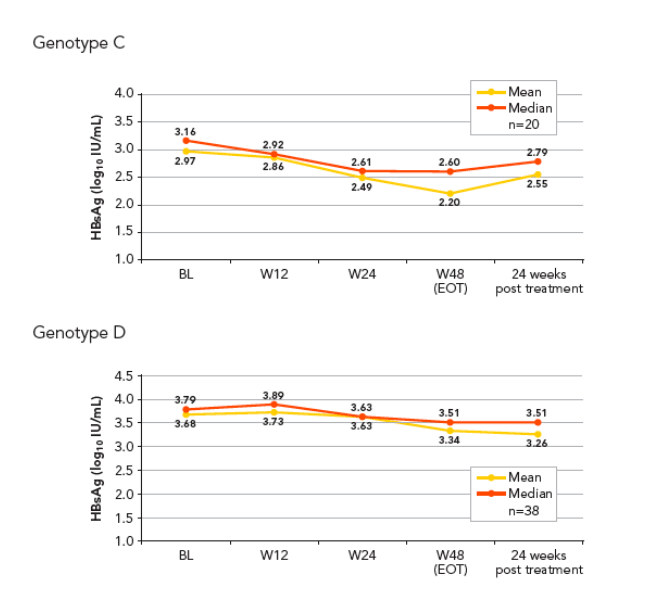
Baseline levels of HBsAg were higher in patients infected with genotypes A and D compared with genotypes B and C (Figure 3A_D)
The lowest levels of HBsAg at end of treatment (EOT) and 6 months post treatment were observed in patients infected with genotype B (Figure 3A_D, Table 1).
The highest HBsAg levels at EOT and end of follow-up were observed in genotype D infected patients who also had high levels at baseline. The median decline in HBsAg in genotype D patients was comparable to that seen in those infected with genotype A (Table 1)
The kinetics of HBsAg decline was different according to genotypes (Figure 3A_D)
-- Genotype B infected patients showed a rapid decline of HBsAg levels (1 log decline) occurring within the first 12 weeks of therapy
-- Genotype C infected patients showed a slower, but continuous decline of HBsAg levels, starting from the beginning of therapy
-- Genotype A and D infected patients showed a more gradual decline that started later than that seen in genotypes B and C

HBsAg decline according to HBsAg clearance 4 years post treatment
In the long-term observational follow-up study, a total of 25 patients cleared HBsAg 4 years post treatment (genotype A, n=2; genotype B, n=4; genotype C, n=14; genotype D, n=4; other, n=1)
We compared the kinetics of HBsAg decline in the patients with and without HBsAg clearance 4 years post treatment
-- A total of 16 patients who cleared HBsAg had available data at all of the time points
-- Genotype A, n=3; genotype B, n=2; genotype C, n=8; genotype D, n=2; other, n=1
-- Data from 70 patients who did not clear HBsAg by post-treatment year 4 were available for comparison
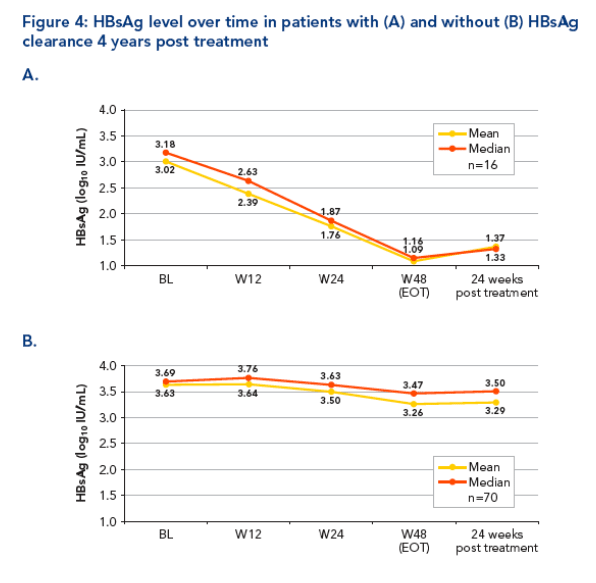
Mean (median) HBsAg levels were lower in those who cleared HBsAg than in
those who did not
-- Mean (median) 3.02 (3.18) log10 IU/mL vs 3.63 (3.69) log10 IU/mL
The overall decline in HBsAg from baseline observed in HBsAg responders was considerably more rapid and of greater magnitude than that observed in non-responders (Figure 4A and B)
-- By week 48, the mean (median) HBsAg decline from baseline was -1.86 (-1.38) log10 IU/mL for those who cleared HBsAg compared with -0.37 (-0.12) ) log10 IU/mL for those who did not clear HBsAg
HBV DNA decline according to HBsAg clearance 4 years post treatment
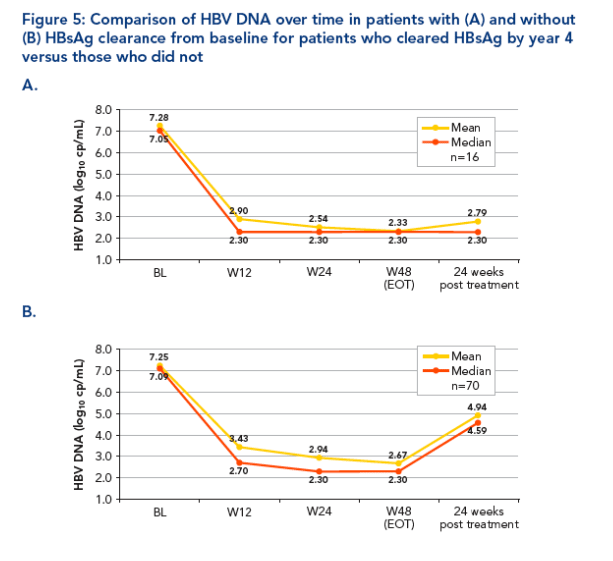
We compared the levels of HBV DNA over time in the patients who cleared HBsAg 4 years post treatment with those who did not
Mean (median) baseline HBV DNA levels were comparable for those who
cleared HBsAg and those who did not
-- 7.28 (7.05) log10 copies/mL vs 7.25 (7.09) log10 copies/mL
The mean (median) HBV DNA levels in those who cleared HBsAg declined rapidly during treatment and remained suppressed after treatment had stopped while those of the patients who did not clear HBsAg declined more slowly and rebounded after end of treatment (Figure 5)
REFERENCES
1. Janssen HL et al. Measurement of HBsAg to monitor hepatitis B viral replication in patients on alpha-interferon therapy. Antiviral Res 1994;23:251-7
2. Marcellin P et al. Peginterferon alfa-2a alone, lamivudine, and the two in combination in patients with HBeAg-negative-chronic hepatitis B. N Engl J Med 2004;351:1206-17
3. Marcellin P et al. Long-term follow-up of HBsAg clearance in patients with HBeAg-negative CHB treated with peginterferon alfa-2a (PEGASYS): increase in HBsAg clearance rate from 3% 6 months post-treatment to 8% after 3 years. Hepatology 2007;46(suppl):673A (Abstract 979)
4. Marcellin P et al. Virological and biochemical response in patients with HBeAg-negative chronic hepatitis B treated with peginterferon alfa-2a (40KD) with or without lamivudine: results of 4-year follow-up. J Hepatol 2008;28(suppl2):S46 (Abstract 103)
DISCLOSURE INFORMATION
This research was funded by F. Hoffmann-La Roche Ltd, Basel, Switzerland
ELE-
|
| |
|
 |
 |
|
|