 |
 |
 |
| |
A Multicenter, Open-Label Study Of Efficacy And Safety Of Peginterferon Alfa-2a (40KD) In Korean Patients With HBeAg-Positive CHB Harboring Lamivudine-Resistant YMDD Mutants
|
| |
| |
Reported by Jules Levin
59th Annual Meeting of the American Association for the Study of Liver Diseases, October 31-November 4 2008, San Francisco, USA
DJ Suh,1 HC Lee,1 KS Byun,2 M Cho,3 YO Kweon,4 WY Tak,4 CY Chon,5 KC Koh,6 Y-S Lee7
1Asan Medical Center, Seoul, South Korea; 2Korea University Guro Hospital, Seoul, South Korea; 3Pusan National University Hospital, Busan, South Korea; 4Kyungpook National University Hospital, Daegu, South Korea;
5Severance Hospital, Seoul, South Korea; 6Samsung Medical Center, Seoul, South Korea; 7Catholic University of Korea, Holy Family Hospital, Bucheon, South Korea
SUMMARY
Korean patients, typically infected with HBV genotype C, harboring HBV strains with YMDD mutations responded just as well to 48 weeks of peginterferon alfa-2a treatment as treatment-naïve patients
Overall rates of HBeAg seroconversion and HBV DNA suppression were similar
Biochemical benefits were also observed with almost 40% of patients achieving
ALT normalization
Adverse events reported were those known to be associated with interferon-based therapy and the number of adverse events experienced did not differ between the two groups
CONCLUSION
Patients who develop lamivudine resistance can be treated effectively with peginterferon alfa-2a (40KD) with response rates approaching those in patients without prior therapy. This therapeutic approach appears to be safe and results in an HBeAg seroconversion rate similar to that seen in lamivudine-treatment-naïve Korean patients
Background
Development of resistance to lamivudine occurs at a high rate - in up to 70% of patients harboring lamivudine-resistant YMDD mutant viral strains after 5 years of treatment1
The virological breakthrough that follows the development of lamivudine resistance may have serious clinical consequences with severe, acute and sometimes fatal exacerbation of liver disease2,3
Patients who are harboring hepatitis B virus (HBV) strains with YMDD mutations following treatment with lamivudine, therefore, represent an important target group for therapy
Treatment with other antivirals, such as entecavir, is a problem due to cross-resistance
Small studies have suggested that treatment with peginterferon alfa-2a may offer an effective approach for these patients4,5
OBJECTIVE
To evaluate the efficacy and safety of peginterferon alfa-2a (40KD) in Korean patients with HBeAg-positive chronic hepatitis B (CHB) who have developed
genotypic resistance to lamivudine
METHODS
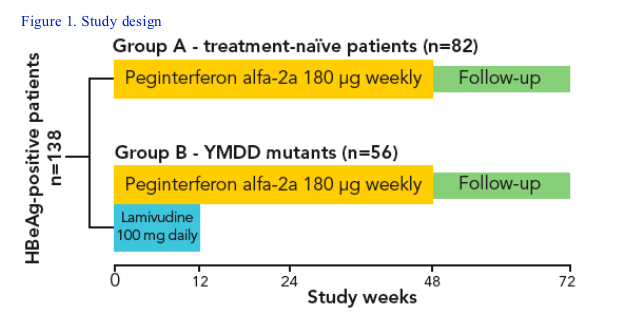
This was a non-randomized, open-label study conducted at various study centers in Korea. Korean patients are typically infected with HBV genotype C Patients were stratified into two groups:
--'Treatment-naïve' patients had not received any antiviral therapy for CHB in the 6 months before enrolment (Group A)
--'YMDD mutant' patients had received in the past or were currently receiving
lamivudine and had genotypic evidence of YMDD mutations (Group B)
All patients received 48 weeks of once-weekly treatment with peginterferon
alfa-2a (40KD) [PEGASYS]
Patients in Group B who were receiving lamivudine therapy at enrolment received overlapping peginterferon alfa-2a (40KD) treatment with lamivudine for 12 weeks (Figure 1)
Primary endpoints were HBV DNA <100,000 copies/mL (measured using Roche TaqMan assay) and loss of HBeAg. HBeAg seroconversion was a secondary endpoint. All were assessed 24 weeks after the end of treatment
The combined response consisted of loss of HBeAg and presence of anti-HBe,
HBV DNA <100,000 copies/mL and alanine aminotranserase (ALT) normalization
All efficacy results shown are from week 72 (24 weeks after the end of treatment) for the per protocol population; adverse events are shown for the randomized population
RESULTS
Patient recruitment and enrolment
The study took place from August 2005 to August 2007 and involved seven treatment centers in South Korea
A total of 150 patients were enrolled in the study. Data from 138 patients were included in this analysis; 82 were treatment-naïve (Group A) and 56 had genotypic evidence of YMDD mutants (Group B). Of these, 61 patients and 43 patients, respectively, had completed the study as planned at the time of the analysis (total n=104)
Baseline characteristics are shown in Table 1. The mean age of the enrolled
population was 35.5±8.9 years. A greater proportion of Group B was male (85.7%) than Group A (72.0%). Baseline ALT levels were higher in Group A (142.1±111.0 IU/L) than in Group B (113.6±142.3 IU/L). Levels of HBV DNA were similar in both groups. Liver biopsies showed that more patients in Group B had cirrhosis (13.3%) and bridging fibrosis (20.0%) than patients in Group A (2.8% and 13.9%, respectively)

* Data from eight patients missing; Data from one patient missing
Efficacy
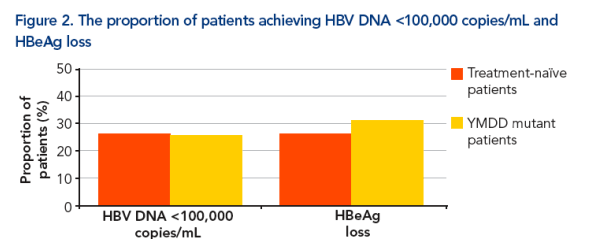
The rates of HBV DNA suppression to <100,000 copies/mL were similar in the two groups. A total of 16 patients in group A achieved this level of DNA suppression (26.2%, 95% CI 15.8%, 39.1%) and 11 patients in group B (25.6%, 95% CI 13.5%, 41.2%) (Figure 2)
The overall HBeAg loss rate was 28% at week 72 and there was no significant difference between the two groups. Of the 61 patients in Group A, 16 achieved HBeAg loss (26.2%, 95% CI 15.8%, 39.1%) and in group B, 13 of the 43 patients achieved HBeAg loss (30.2%, 95% CI 17.2%, 46.1%) (Figure 2)
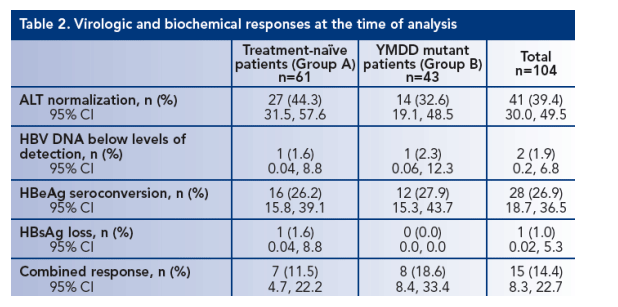
Overall, 39.4% of patients had normalized ALT levels: 44.3% of group A and 32.6% of Group B (Table 2)
HBV DNA levels were below the detection limit in one patient in each group
One patient in Group A had cleared HBsAg by the time of analysis
Seven patients in Group A (11.5%) and eight patients in Group B (18.6%) had achieved a combined response at the time of analysis (Table 2)
Safety
The adverse events profiles were similar between the two groups (Table 3)
although a higher proportion of patients in group B experienced serious
side effects
Adverse events reported were those known to be associated with interferon-based therapy, most frequently alopecia, asthenia, neutropenia and thrombocytopenia

REFERENCES
1. Dusheiko G et al. Treatment of hepatitis B. Gut 2008;57(1):105-24
2. Liaw Y-F. Management of YMDD mutations during lamivudine therapy in patients with chronic hepatitis B. J Gastroenterol Hepatol 2002;17 (Suppl 3):S333-7
3. Yuen MF et al. Clinical outcome and virologic profiles of severe hepatitis B exacerbation due to YMDD mutations. J Hepatol 2003;39:850-5
4. Shi XF et al. Peginterferon alfa-2a for patients with HBeAg-positive CHB who failed prior lamivudine treatment. Hepatol Intl 2007;1:(O-90)18
5. Xu D-Z et al. Efficacy and safety of peginterferon alfa-2a in Chinese patients with HBeAgpositive chronic hepatitis B. J Hepatol 2008;48(Suppl 2):(712) S266
DISCLOSURE INFORMATION
Editorial support for the development of this poster was funded by F. Hoffmann-La Roche Ltd, Basel, Switzerland
|
| |
|
 |
 |
|
|