 |
 |
 |
| |
On-Treatment HBsAg Decline During Peginterferon alfa-2a (40KD) ± Lamivudine in Patients With HBsAg-Positive CHB As A Potential Predictor Of Durable Off-Treatment Response
|
| |
| |
Reported by Jules Levin
59th Annual Meeting of the American Association for the Study of Liver Diseases, October 31-November 4 2008, San Francisco, USA
GK Lau,1 P Marcellin,2 M Brunetto,3 T Piratvisuth,4 H-P Kapprell,5 P Button,6 R Batrla7
1Department of Medicine, The University of Hong Kong, Queen Mary Hospital, Hong Kong, China; 2Service d'Hepatologie, Hôpital Beaujon, Clichy, France; 3UO Epatologia, Azienda Ospedaliero Universitaria Pisana, Pisa, Cisanello, Italy;
4NKC Institute of Gastroenterology and Hepatology, Department of Internal Medicine, Songklanagarind Hospital, Prince of Songkla University, Hat Yai, Thailand; 5Abbott GmbH & Co, Wiesbaden-Delkenheim, Germany;
6Roche Products Pty Ltd, Dee Why, NSW, Australia; 7Hoffmann-La Roche Ltd, Basel, Switzerland
Background
HBsAg clearance is the ultimate goal of therapy in patients with chronic hepatitis B, being associated with favorable outcome
Patients with HBeAg-positive disease who achieve a sustained posttreatment response to interferon-based therapy have an increasing chance of developing subsequent HBsAg clearance during longterm follow up1
Previously, we have shown that a 48-week course of treatment with peginterferon alfa-2a (alone or in combination with lamivudine)
-- but not lamivudine alone - resulted in HBsAg clearance and seroconversion in patients with HBeAg-positive CHB2
It has been suggested that on-therapy reduction in serum HBsAg level may be a useful marker of response to therapy in patients with chronic hepatitis B3,4
Early on-treatment quantification of serum HBsAg represents a new diagnostic tool that allows us to assess the relationship between HBsAg decline by response to therapy
OBJECTIVE
To identify the potential association between reduction in HBsAg - during treatment with peginterferon alfa-2a ± lamivudine - and posttreatment response
AUTHOR SUMMARY
Peginterferon alfa-2a treatment, either alone or in combination with lamivudine, resulted in a considerable decline in HBsAg level from baseline
Significantly more patients treated with peginterferon alfa-2a + lamivudine than with peginterferon alfa-2a alone had a reduction in HBsAg level from baseline to end of treatment of >1 log10 IU/mL
HBsAg levels at baseline and at treatment week 12 were associated with post-treatment response to peginterferon alfa-2a ± lamivudine
An HBeAg seroconversion rate of 50% was achieved 6 months post treatment in patients with week 12 HBsAg level ≦1500 IU/mL
HBV DNA level at week 12 is associated with post-treatment response but the cut-off level is dependent on treatment regimen
AUTHOR CONCLUSION
HBsAg level at baseline and during treatment was associated with response 6 months post treatment in patients with HBeAg-positive disease. HBsAg or HBV DNA determination on-treatment could be used as early predictors of durable
post-treatment response to peginterferon-based therapy in the individual patient
METHODS
This was an analysis of a total of 542 HBeAg-positive patients treated with peginterferon alfa-2a [40KD; PEGASYS] alone (180 _g once weekly, n=271) or in combination with lamivudine (100 mg daily, n=271)
Patients received treatment for 48 weeks followed by 24 weeks of off-treatment
follow up
Quantitative HBsAg in serum was measured pretreatment, on treatment (weeks 12, 24, 48) and 24 weeks post treatment using the Abbott Architect HBsAg assay
We investigated possible association between response 6 months post treatment and HBsAg level at baseline or week 12 and HBV DNA level at week 12. Definitions of response were: HBeAg seroconversion, HBV DNA ≦10,000 copies/mL, HBV DNA ≦10,000 copies/mL + HBeAg seroconversion, HBV DNA ≦400 copies/mL, and HBsAg clearance
RESULTS
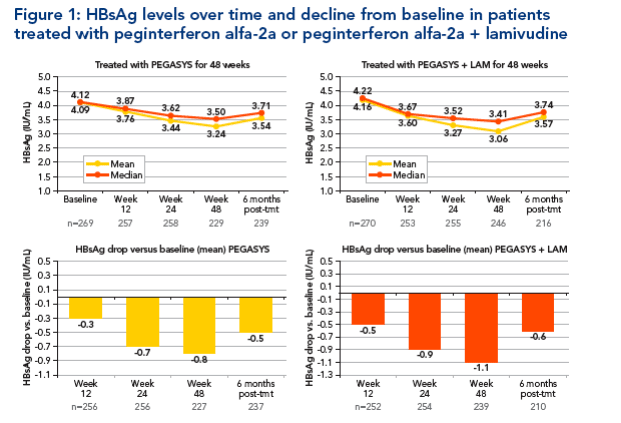
HBsAg level over time and decline from baseline
Baseline HBsAg levels in the two treatment groups were similar (Figure 1)
HBsAg levels decreased from baseline at weeks 12, 24 and 48 in both
treatment groups (Figure 1)
-- Decline in HBsAg level was slightly more pronounced in the peginterferon alfa-2a ± lamivudine group compared with peginterferon alfa-2a monotherapy
-- There was some rebound in HBsAg level between end of treatment and week 72
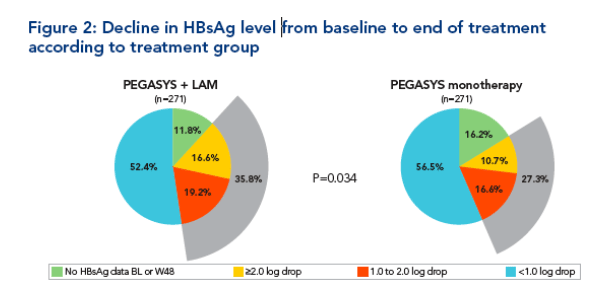
Decline in HBsAg level from baseline to end of treatment according
to treatment
Significantly more patients treated with peginterferon alfa-2a + lamivudine
had a reduction in HBsAg level from baseline to end of treatment of
>1 log10 IU/mL (Figure 2)
-- 35.8% of patients treated with peginterferon alfa-2a + lamivudine vs 27.3% treated with peginterferon alfa-2a monotherapy (P=0.034)
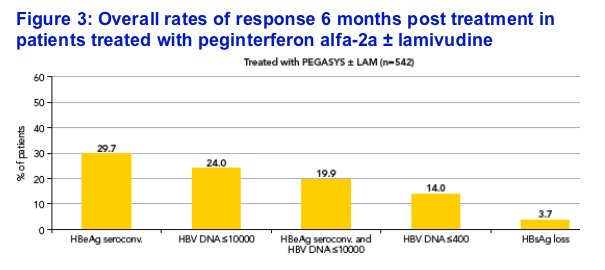
Response rates 6 months post treatment (peginterferon alfa-2a ± lamivudine n=542)
The overall rates of response for the 542 patients treated with
peginterferon alfa-2a ± lamivudine are shown in Figure 3
-- HBeAg seroconversion was achieved in 29.7%; 3.7% of patients had cleared HBsAg 6 months post treatment
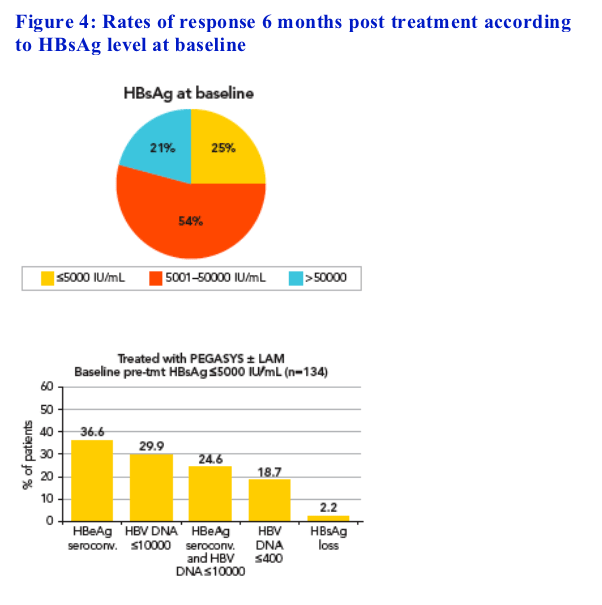
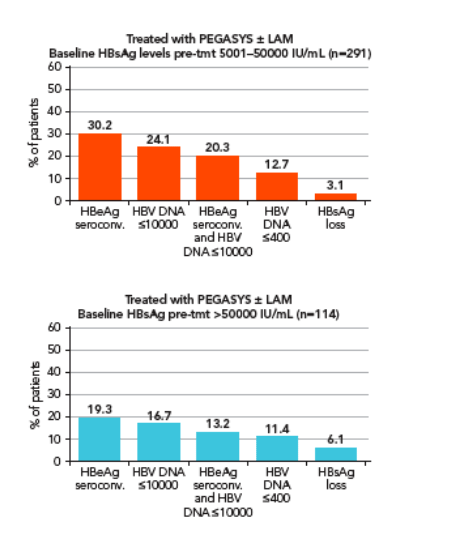
Analysis of response rates according to baseline HBsAg level
Patients were divided in three groups according to baseline HBsAg levels
The HBsAg cut-off levels chosen to delineate the three groups were 5000 IU/mL and 50,000 IU/mL, which correspond to rounded values
approximating to the first and fourth quartiles of the entire population
(5066 and 43,577 IU/mL, respectively) (Figure 4)
-- 25% of patients had HBsAg levels ≦5000 IU/mL
The rates of response to peginterferon alfa-2a ± lamivudine were
associated with baseline HBsAg levels (Figure 4)
-- Patients within the lowest quartile of HBsAg level (≦5000 IU/mL) had higher response rates than those with higher levels
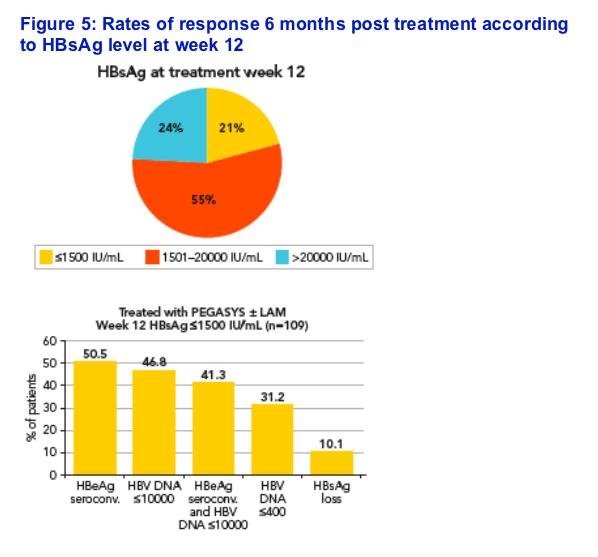
Potential association of HBsAg level at week 12 with response 6 months post treatment
Patients were divided in three groups according to week 12 HBsAg levels
The HBsAg cut-off levels chosen to delineate the three groups were 1500 IU/mL and 20,000 IU/mL, which correspond to rounded values approximating to the first and fourth quartiles of the entire population (1863 and 19,269 IU/mL, respectively) (Figure 5)
21% of patients had HBsAg levels ≦1500 IU/mL
The rates of response to peginterferon alfa-2a ± lamivudine were associated with week 12 HBsAg levels (Figure 5)
-- Highest response rates were observed in the patients in the lowest baseline HBsAg category (≦1500 IU/mL)
Importantly, this association was equally valid for the peginterferon alfa-2a
and the peginterferon alfa-2a + lamivudine patients
-- For example, 50.9% of patients treated with peginterferon alfa-2a and 50.0% of those treated with peginterferon alfa-2a + lamivudine with week 12 HBsAg ≦1500 IU/mL achieved HBeAg seroconversion 6 months post treatment
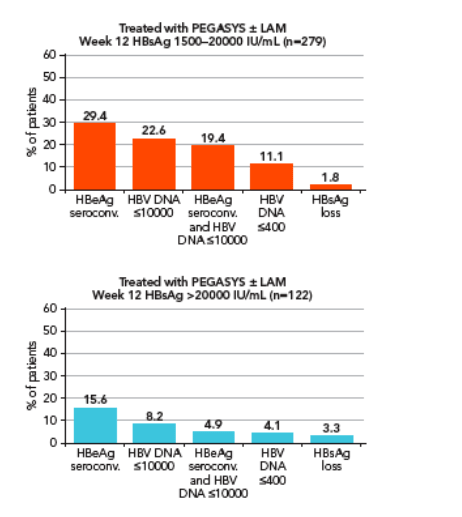
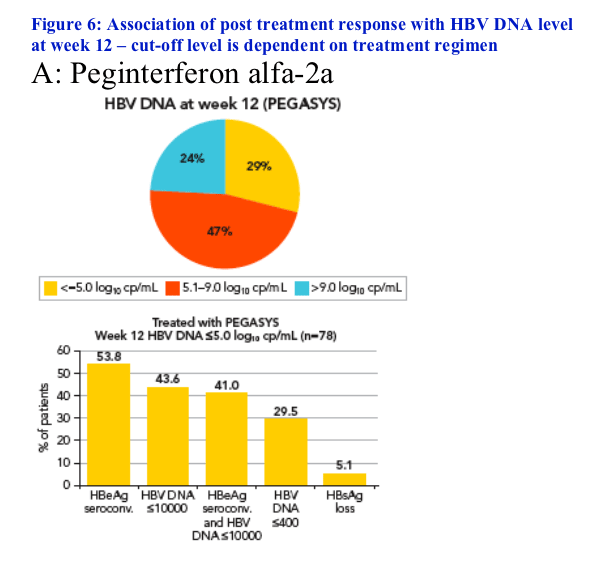
Potential association of HBV DNA level at week 12 with response 6 months post treatment
Since peginterferon alfa-2a alone and peginterferon alfa-2a + lamivudine suppress HBV DNA to different extents this analysis was conducted separately for the 2 treatment groups using different cut-offs
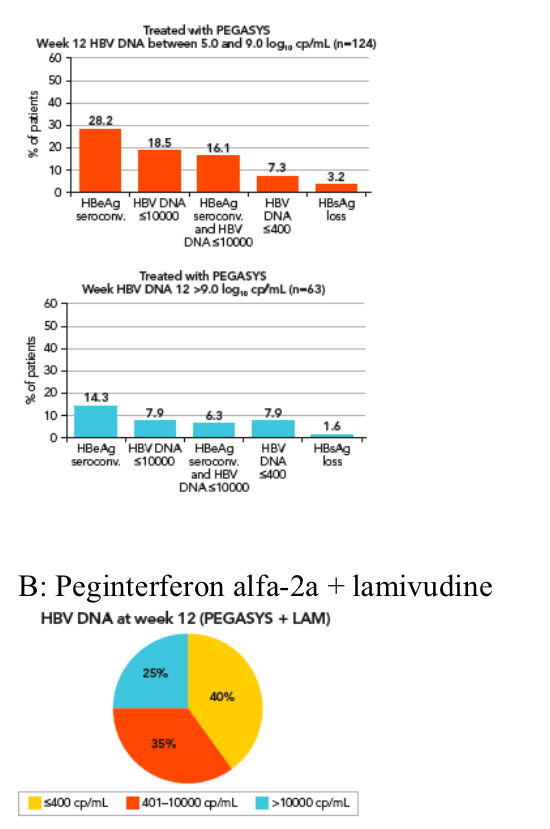
In the peginterferon alfa-2a group patients were divided according to week 12 HBV DNA values ≦5 log10 and >9 log10 copies/mL (Figure 6A)
-- 29% of these patients had HBV DNA ≦5 log10 copies/mL at week 12.
Response rates for peginterferon alfa-2a-treated patients based on
week 12 HBV DNA level are shown in Figure 6A
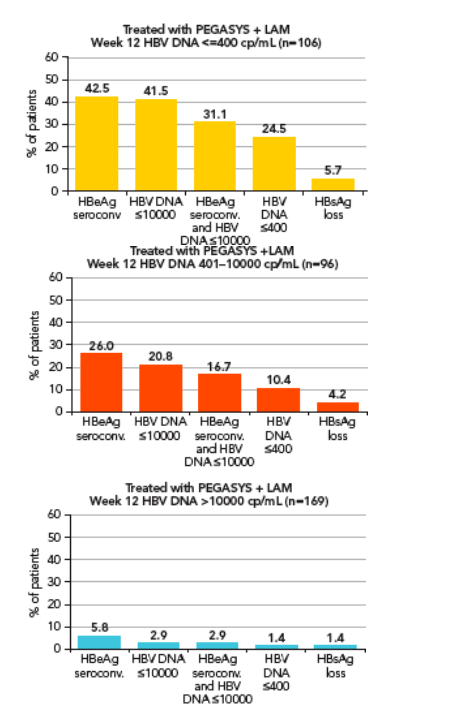
In the peginterferon alfa-2a + lamivudine group patients were divided according
to week 12 HBV DNA values ≦400 copies/mL and 10,000 copies/mL (Figure 6B)
-- 40% of these patients had HBV DNA ≦400 copies/mL at week 12.
Response rates for peginterferon alfa-2a-treated patients based on
week 12 HBV DNA level are shown in Figure 6B
Week 12 HBV DNA level is associated with post-treatment response but is not as appropriate as an on-treatment predictor of response as HBsAg since the cut-off level varies depending on treatment regimen
REFERENCES
1. Korevaar A et al. High rates of HBsAg seroconversion in chronic hepatitis B
patients responding to interferon therapy - a long-term follow-up study. AASLD 2007, P991
2. Lau GK et al. Peginterferon alfa-2a, lamivudine, and the combination for HBeAgpositive chronic hepatitis B. New Engl J Med 2005;2682-2695
3. Janssen HL et al. Measurement of HBsAg to monitor hepatitis B viral replication in patients on alpha-interferon therapy. Antiviral Res 1994;23:251-7
4. Marcellin P et al. In patients with HBeAg-negative chronic hepatitis B, HBsAg
serum levels early during treatment with peginterferon alfa-2a predict HBsAg
clearance 4 years post treatment. AASLD 2008, P919
DISCLOSURE INFORMATION
This research was funded by F. Hoffmann-La Roche Ltd, Basel, Switzerland
|
| |
|
 |
 |
|
|