 |
 |
 |
| |
Results of A Phase I Safety, Tolerability and Pharmacokinetic Study of ANA598, A Non-nucleoside NS5B Polymerase Inhibitor, in Healthy Volunteers
|
| |
| |
Reported by Jules Levin
AASLD San Francisco Nov 2008
Mohamad H. Rahimy, Constance A. Crowley, James L. Freddo, Maria V. Sergeeva, Brent Golec
Anadys Pharmaceuticals, Inc. San Diego, CA
AUTHOR CONCLUSIONS
ANA598 was very well tolerated after administration of single oral doses up to 3000 mg.
There were no serious AEs or discontinuations from the study.
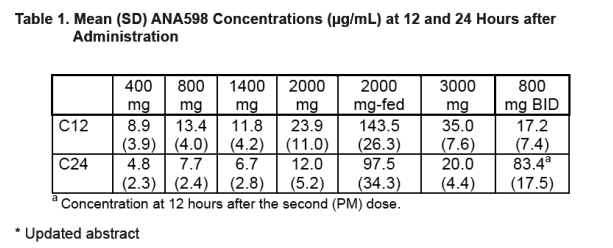
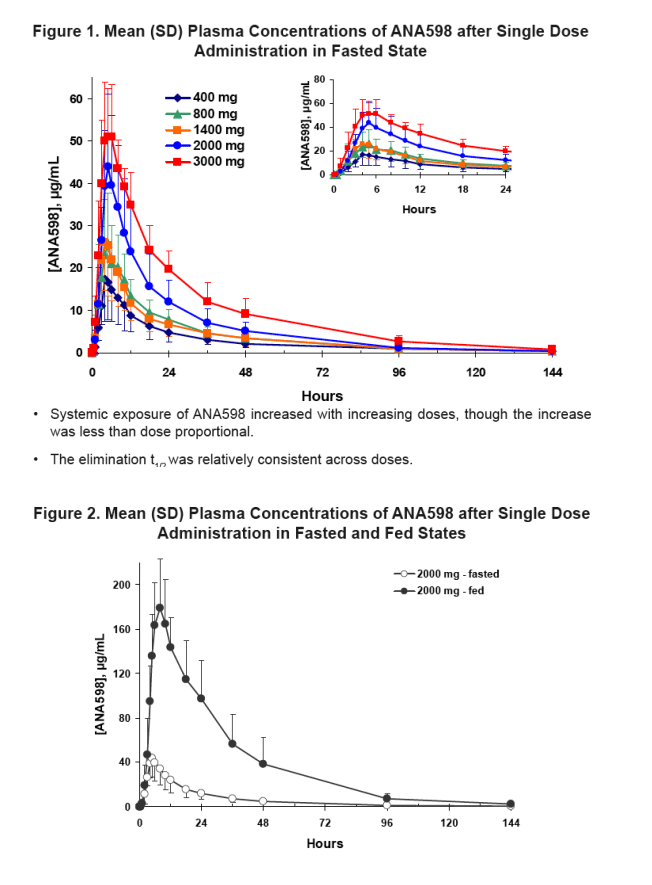
ANA598 systemic exposure (Cmax and AUC) generally increased with increasing doses, though the increase was less than dose proportional under fasting conditions.
ANA598 plasma elimination t1/2 ranged from 22 to 31 hours and was relatively consistent across doses.
Following consumption of a high fat-meal, both Cmax and AUC increased significantly relative to the fasted state.
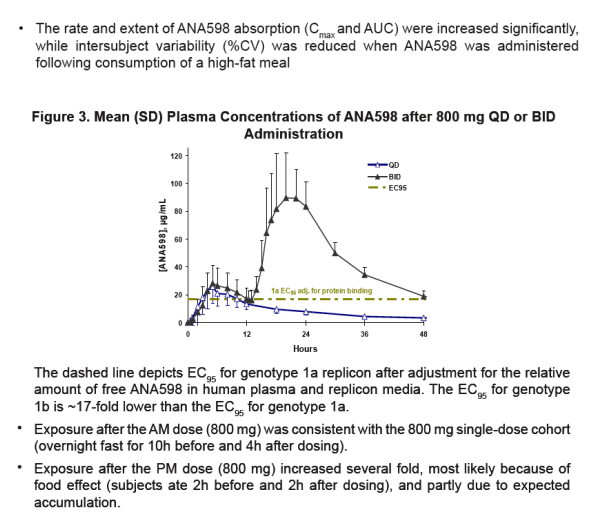
Exposure was higher after the PM dose (800 mg) in the BID cohort, most likely because of food effect and partly due to expected accumulation.
The favorable PK and tolerability profiles of ANA598 support QD or BID
administration.
The results support continued development of ANA598 for patients with chronic HCV infection.
ABSTRACT
Background: ANA598 is a novel non-nucleoside inhibitor of the hepatitis C virus (HCV) genotype 1 NS5B polymerase in development for the treatment of patients with chronic HCV infection. In vitro studies have shown that ANA598 exhibits potent antiviral activity in genotype 1a and 1b replicon systems with EC50 values of 50 and 3 nM (28 and 1.7 ng/mL) and EC95 values of 500 and 30 nM (280 and 17 ng/mL), respectively. Results from a single-dose escalation trial in healthy subjects are reported here.
Methods: A phase I, randomized, double-blind, ascending, single-dose trial was conducted to evaluate the safety, tolerability and pharmacokinetics of ANA598 formulated as 200 mg capsule. Healthy subjects (18 to 55 years of age) were randomized into treatment cohorts consisting of 6 subjects receiving single ascending oral doses of ANA598 (400, 800, 1400, 2000 [fasted and fed], or 3000 mg) and 2 subjects receiving placebo. An additional cohort received 800 mg twice daily (BID) for 2 doses. All doses were administered in the fasted state with the exception of the 2000 mg-fed group. Safety and pharmacokinetics
were evaluated for 7 days after oral administration of ANA598 capsules or matching placebo.
Results: ANA598 was well tolerated at all doses administered. There were no serious adverse events and no withdrawals from the study. All reported adverse events were classified as mild (13 events) or moderate (1 event), with no apparent dose relationship and no pattern of events within any body system. ANA598 systemic exposure generally increased with increasing doses, though the increase was less than dose proportional under fasting conditions (Table 1). Both Cmax and AUC increased by several fold in subjects who received ANA598 after consuming a high-fat meal. ANA598 plasma elimination t1/2 ranged from 22 to 31 hours, and the t1/2 was independent of dose.
Conclusion: The favorable pharmacokinetic and tolerability profiles of ANA598 support QD or BID administration. The results support continued development of ANA598 for patients with chronic HCV infection
BACKGROUND
ANA598 is a novel non-nucleoside inhibitor of the HCV genotype 1 NS5B polymerase in development for the treatment of patients with chronic HCV infection. In in vitro studies, ANA598 has exhibited low nM potency against HCV genotype 1a and 1b replicon systems. Following oral administration of [14C]ANA598 to rats and monkeys, the parent compound in plasma accounted for approximately 99% of the total drug-related radioactivity concentrations, indicating no major circulating metabolites. In rats and monkeys, the majority of radioactivity (81 to 96%) was recovered in feces; this observation in conjunction
with high levels of ANA598 radioactivity seen in the liver and/or bile, suggested that biliary excretion is the major route of ANA598 elimination.
OBJECTIVES
To assess the safety and tolerability of ANA598 after oral administration of single ascending doses up to 3000 mg in healthy volunteers.
To determine the plasma PK of ANA598 after oral administration of single ascending doses in healthy volunteers.
To evaluate the effect of food on the plasma PK of ANA598 after oral administration of the 2000-mg dose following consumption of a high-fat meal.
To assess the PK of ANA598 after oral administration of two 800-mg doses given at 12 hours apart.
DESIGN & METHOD
Randomized, double-blind, placebo-controlled, single ascending dose
Healthy subjects were randomized into sequential cohorts consisting of 6 subjects administered ANA598 capsules and 2 subjects administered matching placebo
Safety assessments included physical examination, vital signs, hematology, blood chemistry, urinalysis, ECG and adverse event reporting throughout the study
For pharmacokinetic evaluations, serial plasma samples were collected for up to 144 hours (6 days) post dose and analyzed using a validated high performance liquid chromatography-tandem mass spectrometery (LC-MS/MS) method
RESULTS
Note: The clinic portion of the study was recently completed but the clinical database has not been locked. Final analysis of the safety data is pending database lock
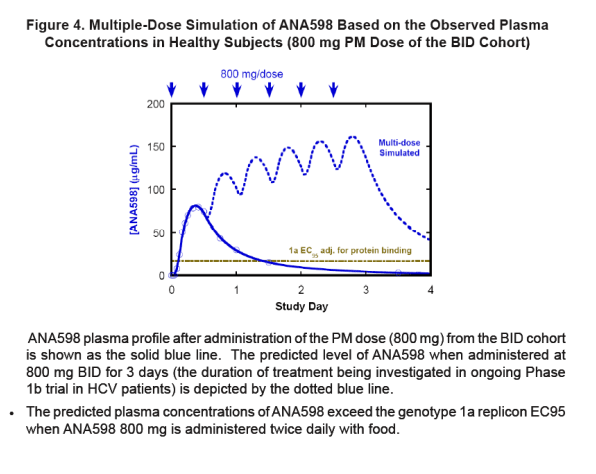
|
| |
|
 |
 |
|
|