 |
 |
 |
| |
Antiviral Efficacy of the HCV RNA Polymerase Inhibitor ANA598 in the Chimpanzee Model of HCV Infection
|
| |
| |
Reported by Jules Levin
AASLD San Francisco Nov 2008
Kevin R. Steffy1, Leonid Kirkovsky1, Robert E. Lanford2, Richard E. Showalter1, Maria Sergeeva1, Jingjing Zhao1, Devron R. Averett1 and James R. Appleman1
1Anadys Pharmaceuticals, Inc., San Diego CA
2 Department of Virology and Immunology, Southwest Foundation for Biomedical Research, San Antonio, TX
AUTHOR CONCLUSIONS
ANA598 exhibits substantial inhibitory effect against HCV genotype 1a and 1b virus in HCV infected chimpanzees following single and/or mul-tiple oral doses and was well tolerated throughout the course of the study.
Plasma concentrations that exceed the HCV replicon EC95 adjusted for protein binding are readily achievable in HCV infected chimpanzees with once-daily oral administration of ANA598 and were achieved in humans (see poster LB13).
A genotypic change (G554D), which confers resistance to ANA598 was observed in both HCV 1b-infected chimpanzees; this mutation has also been detected during in vitro resistance selection studies with ANA598 and shown to remain fully susceptible to interferon-a and several direct antivirals (see poster 1908).
These results support the further development of ANA598 for the treat-ment of genotype 1 HCV infection.
Introduction
ANA598 is a potent and specific non-nucleoside inhibitor of the hepatitis C virus RNA polymerase currently in clinical development for chronic HCV infection. The combination of antiviral potency in vitro (replicon EC50 50 nM and 3 nM for genotypes 1a and 1b, respectively) with robust plasma ex-posures in multiple preclinical species after oral administration prompted assessment of ANA598 in chimpanzees chronically infected with HCV.
Results
To determine the in vivo efficacy of ANA598 against HCV replication, we chose the chimpanzee model of HCV infection, a useful system for the investigation of the antiviral effects and resistance profiles of HCV poly-merase inhibitors. The antiviral efficacy of ANA598 was evaluated in two sets of chimpanzees infected with HCV genotypes 1a and 1b.
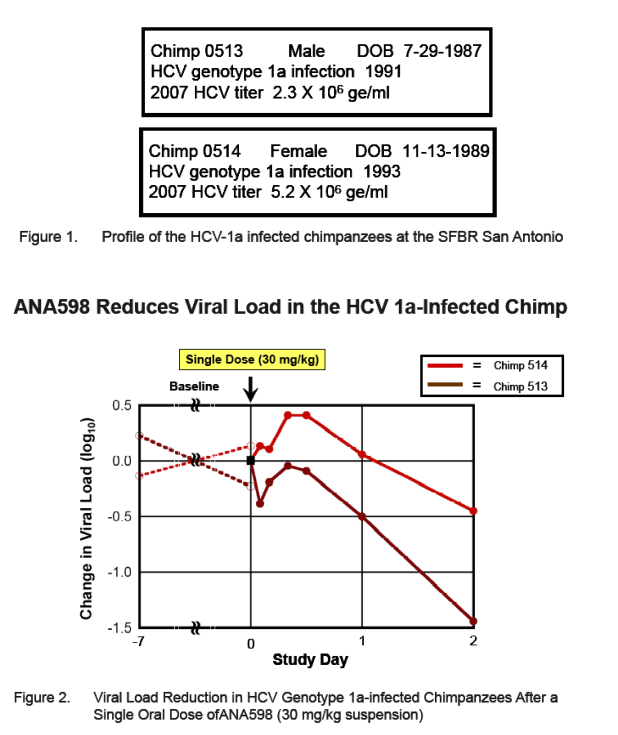
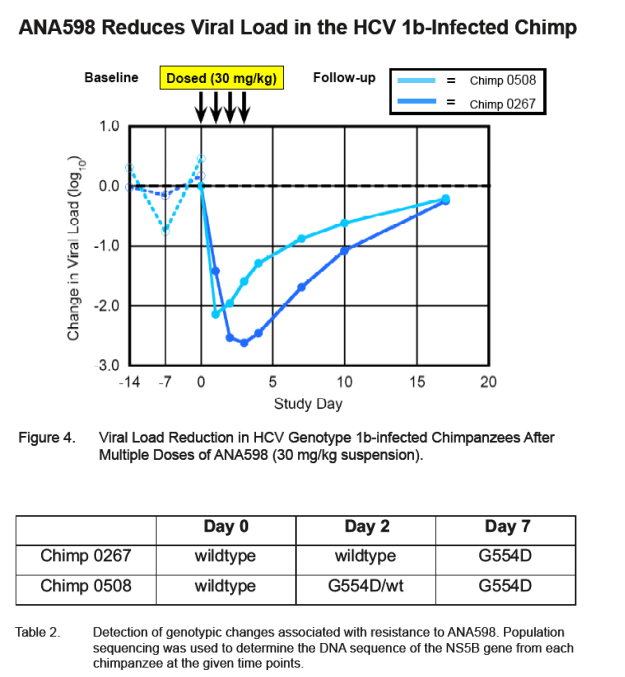
Two HCV 1a-infected chimpanzees were given a single oral dose at 30 mg/kg. This single dose of ANA598 produced 0.5 and 1.4 log10 reductions in viral load in the respective animals at 48 hours post-dose. ANA598 was adminis-tered to two HCV-1b infected chimpanzees as a once daily dose of 30 mg/kg for 4 consecutive days. Serum samples to monitor the viral titer were collected daily for 5 days as well as periodically during a 16 day follow-up period. Maximum viral load reductions of 2.2 and 2.6 log10 copies RNA/ml for each 1b-infected chimpanzee were observed within 24-48 hours after the initiation of treatment.
After the initial drop in the viral load, one chim-panzee maintained suppression of viral replication that lasted throughout the treatment period. In the other chimpanzee, a gradual rise of plasma HCV RNA levels was observed beginning at day 2 and continuing through dosing (0.9 log increase from the nadir to 24 hr after last dose). Viral titers in both animals returned to within 2-fold of the baseline value by day 16.
DNA sequence analysis of the NS5B gene derived from chimp serum is currently in progress to determine if mutations associated with ANA598 drug resistance are present.

RESULTS
At 48 hours after dosing, ANA598 produced 0.5 and 1.4 log10 reductions from baseline in viral load in the respective chimpanzees (baseline is the average of day -7 and day 0 virus titers)
No genotypic changes associated with ANA598 drug resistance were observed in the viral RNA at the 24 hour time point for both chimpan-zees.
RNA samples for the 48 hour time point did not amplify for either pri-mate.

During the first 48 hours after initiation of dosing, the maximum viral load reductions from baseline in chimp 0267 and chimp 0508 were 2.6 log10 and 2.2 log10 respectively (baseline is the average of day -14, day -7 and day 0 virus titers).
In Chimp 0267 viral load reduction was sustained throughout the re-maining dosing period while in Chimp 0508 a modest rise in viral load was observed (0.9 log10) over days 3 and 4.
A genotypic change (G554D) associated with ANA598 drug resistance was detected in the viral RNA beginning at the day 2 time point for Chimp 0508 and was the predominant genotypic variant for both chimpanzees at the day 7 time point. The replicon EC50 for this mutant is 634 nM (see Poster 1908). Using the more sensitive ultra-deep pyrosequencing technology, we also observed a genotypic change (D559G) at a 7.8% frequency at day 7 for chimp 0508. Due to the long plasma half-life of ANA 598, chimps were exposed to suboptimal levels of ANA598 from the end of dosing through day 7.
ANA598 was well-tolerated in the chimpanzees with no safety con-cerns.
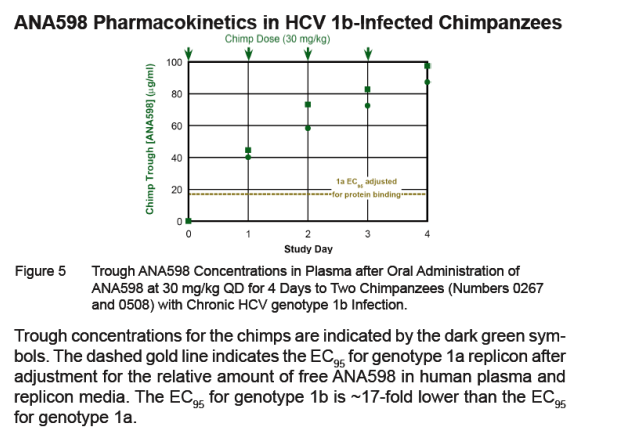
Trough ANA598 concentrations measured 24 hours after the first dose of ANA598 were 40 and 45 μg/ml and increased with each subsequent dose, reaching 87 and 97 μg/ml 24 hours after the fourth dose. This in-crease is consistent with a long plasma half-life similar to that observed in humans (see poster LB13). These trough levels are well in excess of the minimal concentrations predicted to achieve viral load reduction.
Disclosure
Kevin Steffy - Employee of: Anadys Pharmaceuticals, Inc., Stock Holder of: Anadys Pharmaceuticals, Inc.: Leonid Kirkovsky - Former Em-ployee of: Anadys Pharmaceuticals, Inc., Stock Holder of: Anadys Pharmaceuticals, Inc.; Robert Lanford - Employee of: Southwest Founda-tion for Biomedical Research; Richard Showalter - Employee of: Anadys Pharmaceuticals, Inc., Stock Holder of: Anadys Pharmaceuticals, Inc.; Maria Sergeeva - Employee of: Anadys Pharmaceuticals, Inc., Stock Holder of: Anadys Pharmaceuticals, Inc.; Jingjing Zhao - Former Employee of: Anadys Pharmaceuticals, Inc., Stock Holder of: Anadys Pharmaceuticals, Inc.; Devron Averett - Consultant of: Anadys Phar-maceuticals, Inc., Stock Holder of: Anadys Pharmaceuticals, Inc.; James Appleman - Employee of: Anadys Pharmaceuticals, Inc., Stock Holder of: Anadys Pharmaceuticals, Inc.
|
| |
|
 |
 |
|
|