 |
 |
 |
| |
Two Schering CCR5 Antagonists Get Spotlight at HIV Conference
|
| |
| |
15th Conference on Retroviruses and Opportunistic Infections
February 3-6, 2008
Boston
Mark Mascolini
VICTOR E-1 investigators offered results of a double-blind, placebo-controlled trial supporting 30 mg of vicriviroc once daily as the best dose of this CCR5 antagonist for further development [1], while phase 1 findings on another CCR5 plugger, SCH532706, confirmed that this newer agent sticks tenaciously to CCR5 coreceptors after dosing stops [2]. The two studies headlined the opening press briefing at the 15th Conference on Retroviruses and Opportunistic Infections in Boston.
Barry Zingman (Montefiore Medical Center, Bronx, New York) and VICTOR E-1 colleagues randomized 39 heavily treatment-experienced people to 30 mg of vicriviroc once daily, 40 people to 20 mg once daily, and 35 people to placebo. Everyone also got an optimized background regimen (OBR) including a ritonavir-boosted protease inhibitor (PI). Both vicriviroc and SCH532706 need a ritonavir boost.
All study participants had tried drugs from at least three antiretroviral classes, carried mutations conferring resistance to both protease and reverse transcriptase inhibitors, and had a viral load above 1000 copies during at least 6 weeks of treatment with the same regimen. Everyone's background combination included a ritonavir-boosted PI they had not tried before and at least three antiretrovirals in all. Of the 116 people enrolled in the US and Brazil, 78% were men, 71% Latino, 68% Caucasian, and 5% coinfected with hepatitis C virus. Starting viral loads averaged 4.5 log (about 30,000 copies) and starting CD4 counts generally lay in the low 200s.
Viral load declines after 48 weeks of treatment (the primary endpoint) averaged 1.77 log in the 30-mg vicriviroc group, 1.75 log in the 20-mg arm, and 0.79 log in the placebo group. Respective CD4 average counts rose by 102, 136, and 63 cells. Fourteen people (40%) randomized to placebo endured virologic failure by week 48, compared with 5 (18%) in the 30-mg vicriviroc group and 3 (8%) in the 20-mg group.
More than half of study participants taking vicriviroc attained a 48-week viral load below 50 copies in an intent-to-treat analysis, compared with 14% in the placebo group (table). The vicriviroc groups controlled HIV better than the placebo group regardless of whether they had 0, 1 to 2, or 3 or more active drugs in their background regimen (table).
48-Week intent-to-treat <50-copy rate with vicriviroc in heavily experienced patients
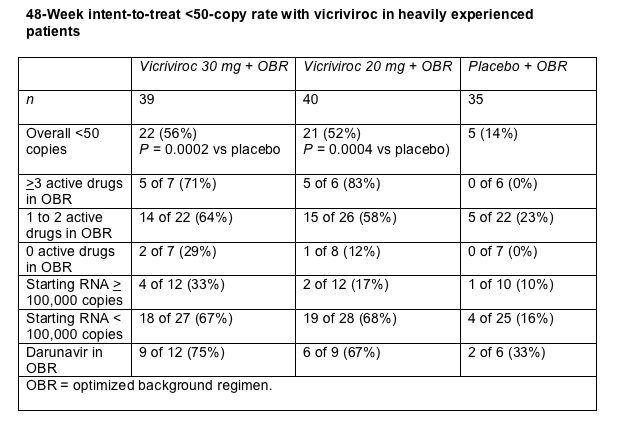
About one third of people beginning 30 mg of vicriviroc with a viral load above 100,000 copies reached a sub-50-copy load, compared with 17% taking 20 mg and 10% taking placebo (table). More people taking 30 mg than 20 mg had a vicriviroc trough concentration above 100 ng/mL (92% versus 82%). Because of these virologic and pharmacologic results, Schering will test 30 mg of vicriviroc once daily with a ritonavir-boosted PI in phase 3 trials.
Everyone enrolled in VICTOR E-1 had to have CCR5-using virus when screened for the trial. As in studies of maraviroc, the already-licensed CCR5 antagonist, a number of people screened with R5 HIV had dual/mixed or X4-using virus when they showed up to take their first dose. Researchers believe this apparently sudden turnaround means a person had dual/mixed or X4 virus all along, but at low levels that became detectable only on study day 1. Still, dual-mixed or X4 virus emerged about twice as often in the 30-mg vicriviroc group (23%) than in the 20-mg group (10%) or the placebo group (9%). Zingman told reporters that emergence of dual-mixed or X4 virus did not coincide with regimen failure. (from Jules: but there is no expectation that mixed or X4 will respond to CCR5 antagonist).
Grade 3 or 4 side effects were equivalent in the three study groups, affecting 21% taking 30 of vicriviroc, 20% taking 20 mg, and 20% taking placebo.
Schering's second CCR5 inhibitor in clinical trials still goes by its in-house number, SCH532706 [2]. A potential plus for this compound is its strong affinity for the CCR5 coreceptor; once it latches on, it's loath to let go.
Sarah Pett (University of New South Wales, Sydney) outlined results of a single-center, open-label, safety and efficacy study at a press briefing on the first day of the conference. The study involved 4 antiretroviral-naive and 8 experienced men with an average CD4 count of 385 and an average viral load of 4.56 log (about 35,000 copies). They all took 60 mg of SCH532706 twice daily plus 100 mg of ritonavir once daily for 10 days. But Pett and colleagues kept measuring their viral load after they stopped the drugs.
On day 10 viral load drops averaged 1.31 log (95% confidence interval 1.6 to 1.0). And viral loads continued falling through day 15, when the average decline stood at 1.62 log (95% confidence interval 2.0 to 1.3). That persistent viral decay probably means SCH532706 keeps HIV from binding to CCR5 for days after dosing stops. Pett speculated that SCH532706 may be a once-daily drug candidate. HIV populations in study participants continued using CCR5 throughout the trial.
Eleven of 14 people complained of side effects, often gastrointestinal problems that ritonavir may cause. About three quarters of side effects were mild, and two thirds were judged unrelated to study drugs. One serious problem, moderate pericarditis, arose 13 days after dosing stopped and disappeared spontaneously. No one had heart problems detectable by electrocardiography.
References
1. Zingman B, J Suleiman J, DeJesus E, et al. Vicriviroc, a next generation CCR5 antagonist, exhibits potent, sustained suppression of viral replication in treatment-experienced adults: VICTOR-E1 48-week results. 15th Conference on Retroviruses and Opportunistic Infections. February 3-6, 2008. Boston. Abstract 39LB.
2. Pett S, Emery S, MacRae K, et al. Safety and activity of SCH532706, a small molecule chemokine receptor 5 antagonist in HIV-1-infected individuals. 15th Conference on Retroviruses and Opportunistic Infections. February 3-6, 2008. Boston. Abstract 38.
|
| |
|
 |
 |
|
|