 |
 |
 |
| |
Darunavir EvaLuation in Pediatric HIV-1-Infected treatment-experienced patients, DELPHI
(Trial TMC114-C212)
|
| |
| |
Reported by Jules Levin
15th CROI, Feb 3-6, 2008, Boston
R Bologna, S Rugina, P Cahn, P Flynn, S Blanche, B Van Baelen, R De Greef, L Lavreys, T Van De Casteele, S Spinosa-Guzman
AUTHOR CONCLUSIONS
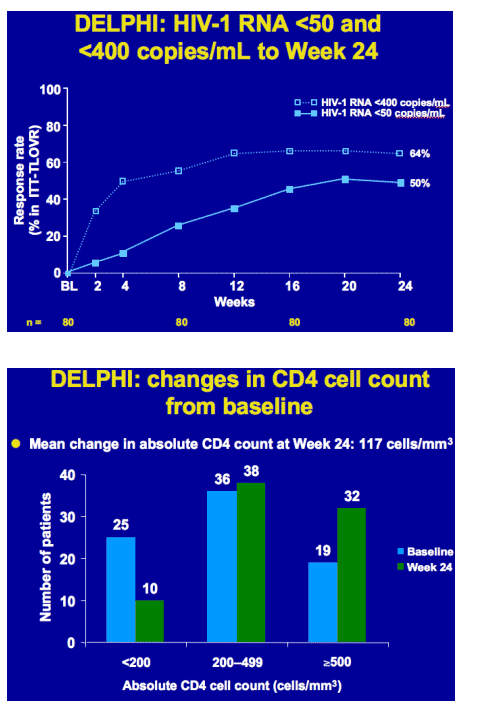
In treatment-experienced HIV-1-infected children and adolescents at Week 24, DRV/r showed
-- good virologic response rates (64% <400, 50% <50 c/ml)
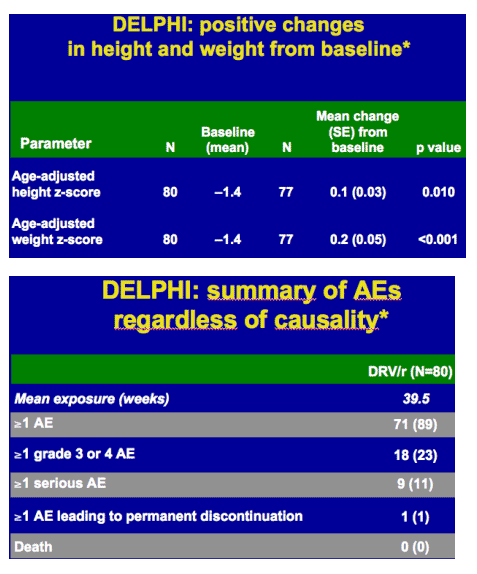
-- positive clinical outcomes
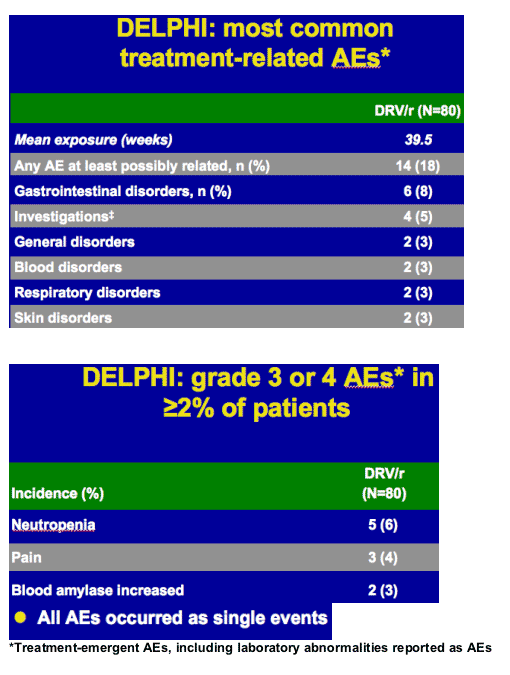
-- favorable safety and tolerability
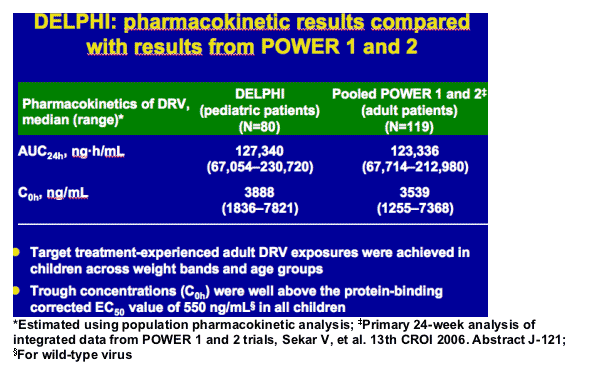
-- comparable exposure to adults and confirmed appropriate dose selection in this pediatric population
-- DRV/r is a new therapeutic option for treatment-experienced HIV-1-infected pediatric patients
DELPHI: open-label, Phase II study
Week 24 primary analysis (TMC114-C212)
evaluation of safety, tolerability and efficacy of DRV/r plus an OBR over 48 weeks in ARV treatment-experienced male and female pediatric patients


|
| |
|
 |
 |
|
|