 |
 |
 |
| |
Inefficient Renal Transport of Nucleotide Reverse Transcriptase Inhibitor GS-9148 Indicates Its Low Potential for Nephrotoxicity
|
| |
| |
Reported by Jules Levin
15th CROI, Feb 3-6, 2008, Boston
G Laflamme,1 A Ray,1 R Fisher,2 R Mackman,1 and T Cihlar1
1Gilead Sciences, Inc., Foster City, CA; 2Vitron, Inc., Tucson, AZ
INTRODUCTION
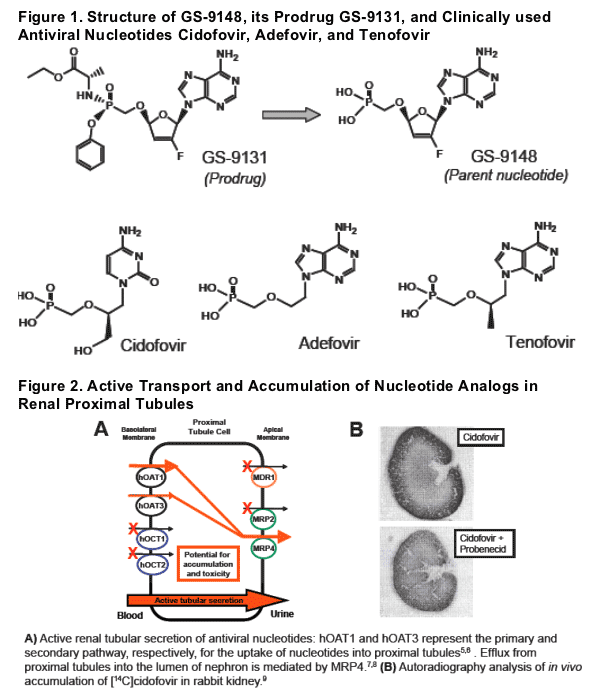
GS-9148 is a novel nucleotide HIV RT inhibitor active against a variety of NRTIresistant strains (M184V, K65R, L74V, and multiple TAMs).1
GS-9148 has been rationally designed to eliminate the mitochondrial toxicity of its structural precursor d4AP by incorporating a 2'-fl uorine substitution into its sugar moiety.1
GS-9131, an orally bioavailable amidate prodrug of GS-9148, effi ciently delivers GS-9148 into peripheral lymphocytes in vitro and in vivo,1,2 and is currently under clinical evaluation.
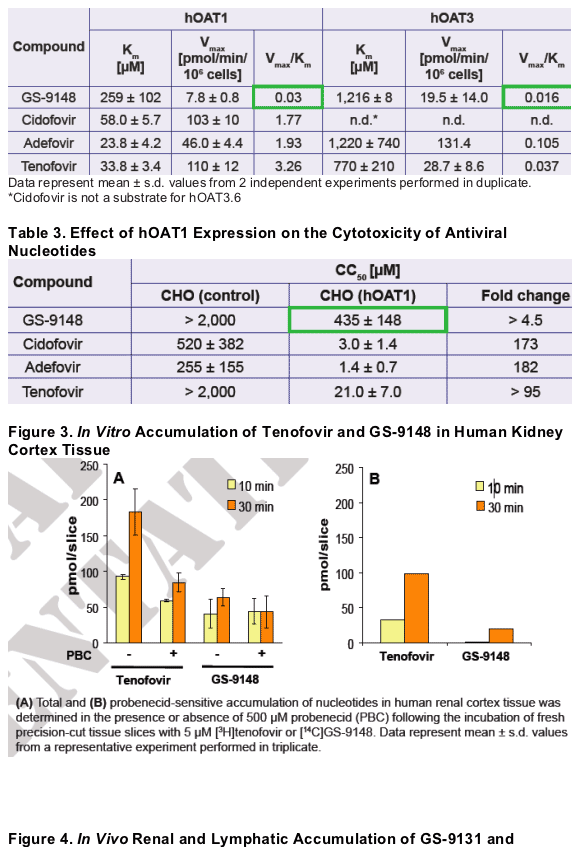
Renal tubular dysfunction occurs in a subset of patients treated with antiviral nucleotides, particularly cidofovir.3 This adverse effect is etiologically linked to the active uptake and accumulation of nucleotides in proximal tubules via organic anion transporters, primarily hOAT1 and hOAT3.4-6
This study assessed the in vitro and in vivo renal transport and accumulation of GS-9148 in comparison with clinically used antiviral nucleotides cidofovir, adefovir, and tenofovir.
AUTHOR CONCLUSIONS
Similar to tenofovir and in contrast to cidofovir and adefovir, GS-9148 exhibits low cytotoxicity in RPTECs (Table 1).
GS-9148 showed relatively inefficient transport by the renal uptake transporters hOAT1 and 3 (Table 2). GS-9148 was also markedly less cytotoxic than other
antiviral nucleotides in cells over-expressing hOAT1 (Table 3).
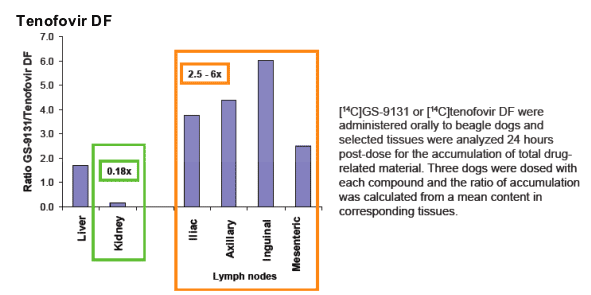
Lower in vitro active accumulation of GS-9148 was observed in fresh human renal tissue compared to that of tenofovir (Fig. 3).
When compared to tenofovir DF, reduced in vivo renal accumulation and enhanced lymphoid uptake of the drug-related material was observed following oral administration of GS-9131 to beagle dogs (Fig. 4).
Together, results of this study demonstrate a less effective renal transport of GS-9148 compared to cidofovir, adefovir, and tenofovir, suggesting a low potential for nephrotoxicity.
METHODS
GS-9148 and GS-9131 were synthesized as described.1,10 Cidofovir, adefovir, and tenofovir were provided by Gilead's Process Chemistry.
In vitro cytotoxicity was assessed in primary human renal proximal tubule epithelial cells (RPTECs; Clonetics-Cambrex, San Diego, CA) following a 5-day incubation with tested drugs. Cell viability was quantifi ed using a CellTiterGlo reagent (Promega, Madison, WI).
Kinetics of renal transport and its effect on the cytotoxicity of nucleotides was characterized in CHO and BHK cells stably transfected with human renal organic anion transporter type 1 (hOAT1) and 3 (hOAT3), respectively. Experiments were conducted as previously described.4-6
In vitro accumulation of nucleotides in human renal cortex was assessed using the incubation of precision-cut fresh tissue slices (Vitron, Tucson, AZ) with 5 _M [14C]GS-9148 or [3H]tenofovir. After incubation, tissue was washed and solubilized in 0.5 N NaOH. Radioactivity in tissue lysates was counted following their neutralization with HCl.
In vivo renal and lymphoid drug accumulation was determined 24 hours after the oral administration of [14C]GS-9131 or [14C]tenofovir DF to beagle dogs using standard tissue processing techniques.
RESULTS

REFERENCES
1. Cihlar T. et al., Antimicrobial Agents Chemother 52, E-publication ahead of print (2008).
2. Ray A. et al., Antimicrobial Agents Chemother 52, E-publication ahead of print (2008).
3. Lalezari J. et al., Ann Intern Med 126, 257-263 (1997).
4. Cihlar T. et al., Nucleosides Nucleotides Nucleic Acids 20, 641-648 (2001).
5. Ho E. et al., J Amer Soc Nephrol 11, 383-93 (2000).
6. Cihlar T. et al., 44th ICAAC, Abstr. A-443 (2004).
7. Ray A. et al., Antimicrobial Agents Chemother 50, 3297-304 (2006).
8. Cihlar T. et al., Antiviral Ther 12, 267-72 (2007).
9. Cundy K. et al., Drug Metab Dispos 24, 315-21 (1996).
10. Boojamra C. et al., Bioorg Med Chem Lett, E-publication ahead of print (2008).
|
| |
|
 |
 |
|
|