 |
 |
 |
| |
GS-8374, a Novel HIV Protease Inhibitor, Does Not Alter Peripheral Glucose Disposal in a Healthy Rodent Model System
|
| |
| |
Reported by Jules Levin
15th CROI, Feb 3-6, 2008, Boston
C Callebaut,1 Q Yan,2 J Koster,2 L Tsai,1 T Cihlar,1 and P Hruz2,3
1Gilead Sciences, Inc. Foster City, CA; 2Department of Pediatrics; 3Department of Cell Biology and Physiology, Washington University School of Medicine, St. Louis, MO
INTRODUCTION
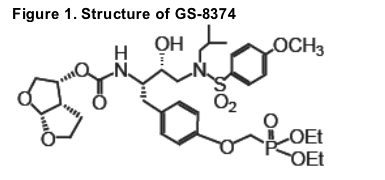
GS-8374 is a novel HIV-1 protease inhibitor (PI) with a unique diethylphosphonate motif incorporated into its molecule (Fig. 1). We previously showed that GS-8374 is a potent and selective PI, and that its
phosphonate moiety favorably affects its resistance profi le.1,2
The clinical use of HIV PIs in combination with other antiretroviral drugs dramatically reduced the morbidity and mortality of HIV infection.3 However, adverse metabolic effects induced by antiretroviral therapies remain one of the major factors often limiting their long-term clinical benefi t.4,5 Notably, many of the first generation HIV PIs are known to contribute to the development of lipodystrophy and peripheral insulin resistance.6
In vitro studies have demonstrated that some PIs can inhibit lipogenesis and adipocyte differentiation.7 Furthermore, it has been shown that PIs such as ritonavir and lopinavir induce insulin resistance in vitro by reducing glucose transport mediated by the facilitative glucose transporter GLUT4;8 conversely, atazanavir has been shown to have minimal effects on insulin sensitivity in vitro,9 and these profi les were respectively confi rmed in vivo.10,11
This presentation summarizes the in vitro effects of GS-8374 on adipocyte functions and characterizes the in vivo potential of GS-8374 to affect glucose disposal in a rat model, in comparison with key marketed PIs.
AUTHOR CONCLUSIONS
Treatment of human primary adipocytes with GS-8374 or atazanavir with concentrations up to 30 _M showed no effect on normal lipid accumulation; under the same conditions, ritonavir and lopinavir affected lipid accumulation with EC50 values of 16 and 17 _M, respectively.
Treatment of differentiated OP-9 mouse adipocytes with 10 _M GS-8374 or atazanavir showed no effect on the insulin-stimulated deoxyglucose uptake; ritonavir and lopinavir caused 50 and 33% reductions in deoxyglucose uptake, respectively, under the same conditions.
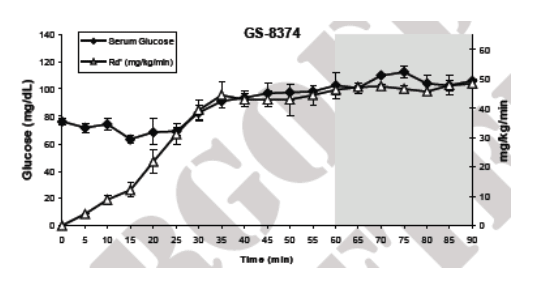
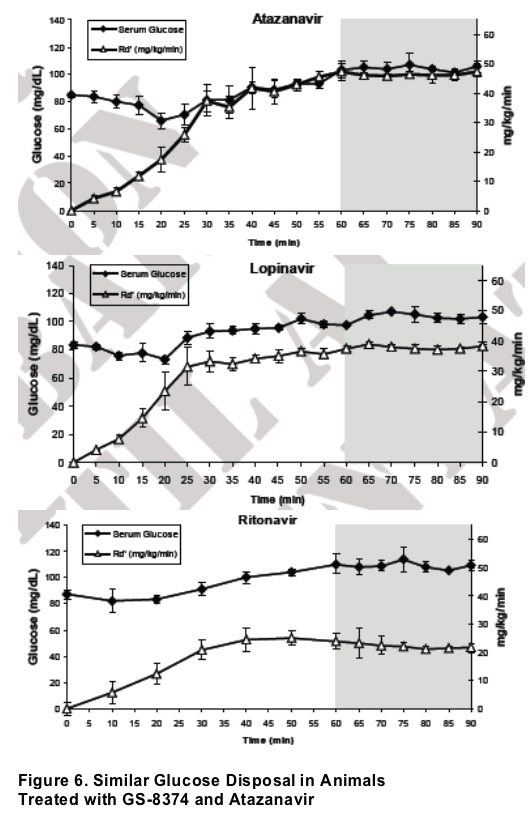
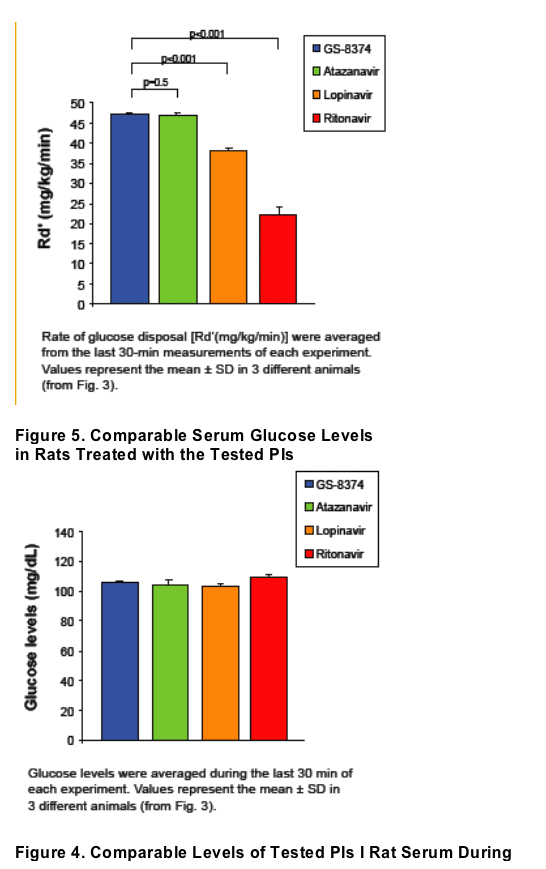
In rats, sustained serum levels of 8 _M GS-8374 or atazanavir had no effect on peripheral glucose disposal. Consistent with previous studies, comparable serum levels of ritonavir or lopinavir produced acute reduction of glucose disposal.
These results suggest a low potential of GS-8374 for metabolic adverse effects compared to some other PIs and confirm that these effects are not inherently linked to antiretroviral activity of PIs.
METHODS
Synthesis of GS-8374 and other protease inhibitors was performed at Gilead Sciences.
For lipid accumulation assay, primary human adipocytes (Cambrex) were cultivated with 10 _g/mL insulin, 1 _M dexamethasone, 200 _M indomethacin, 500 _M isobutyl-methylxanthine in presence of tested compounds. After 9 days, AdipoRed reagent (Cambrex) was added to the cells and fl uorescence was measured.
For glucose uptake, mouse OP9 stromal cell-derived differentiated adipocytes (ATCC) were treated with insulin and tested compounds (10_M). After one hour, [3H]-deoxy-D-glucose (0.2 _M) was added for 10 min followed by cell washing and lysis. Deoxyglucose uptake was determined by scintillation counting.12
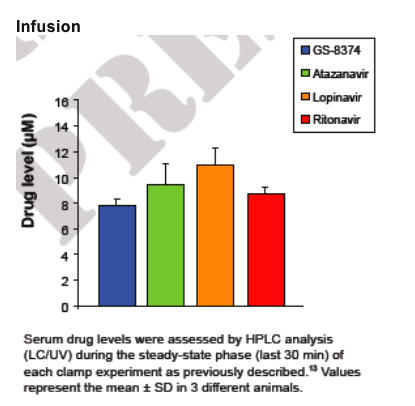
For in vivo studies, euglycemic hyperinsulinemic clamp experiments were performed in chronically catheterized healthy lean male Wistar rats. (all animal procedures were approved by the animal studies committee at Washington University School of Medicine) . Catheters were inserted into carotid arteries and jugular veins of ~200- g rats at least 5 days before the experiments. Intravenous infusion of the PIs was performed as previously described.13 Briefl y, a constant infusion of PI was started through the venous catheter at a rate of 10 _L/min for the duration of the clamp experiment. After 30 min of drug infusion, insulin (10 mU · kg-1 · min-1) was infused through the venous catheter. At 5 or 10-min intervals, determination of blood glucose levels was performed. Dextrose (50%) was infused through the venous catheter at a rate sufficient to maintain a plasma glucose level of ~105 mg/dL. Insulin sensitivity was determined by the average infusion rate during the final 30 minutes of each 90 minute clamp experiment (Fig. 2).
RESULTS
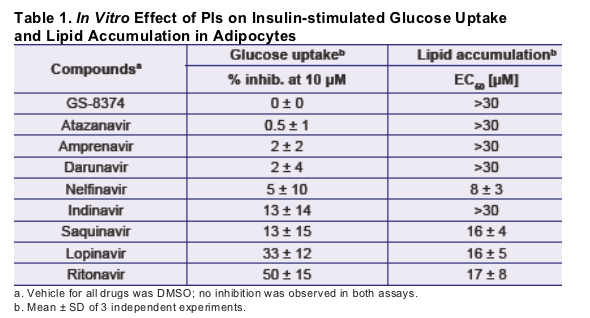
Figure 3. Acute In Vivo Effects of PIs on Peripheral Glucose Disposal
Euglycemic-hyperinsulinemic clamps were performed using 10 mU · kg-1 · min-1 of insulin. PIs were administered by constant intravenous infusion to maintain drug levels of 8-12 _M. Data represent the mean ± SD of measurements in 3 different animals performed during the glucose steady state of the experiment (grey area).
REFERENCES
1. Callebaut et al., (2007) Antiviral Research, 74 :A27.
2. Callebaut et al., (2007) Antiviral Therapy, 12:S18 #16.
3. Murphy et al., (2001) Ann. Intern. Med., 135: 17.
4. Carr et al., (1998) AIDS, 12:F51.
5. Sax et al., (2004) J. AIDS, 37:1111.
6. Murata et al., (2002) Current Drug Targets - Infectious Disorders, 2:1.
7. Dowell et al., (2000) J. Biol. Chem., 275:41325.
8. Hruz et al., (2005) JAIDS 40: 398-403.
9. Wolins et al., (2006) J. Lipid Res., 47:450.
10. Lee et al., (2005) Antiviral Therapy, 10:L6.
11. Noor et al., (2006) AIDS, 20:1813.
12. Hruz et al., (2002) Diabetes, 51:937.
13. Foisy et al., (1999) J. Chromatogr. Biomed. Appl., 721:239.
|
| |
|
 |
 |
|
|