 |
 |
 |
| |
Efficacy and Safety of Boosted Once-Daily Atazanavir and Twice-Daily Lopinavir Regimens in Treatment-Naive HIV-1 Infected Subjects
CASTLE: 48-Week Results
|
| |
| |
Reported by Jules Levin
CROI 2008, Boston
Jean-Michel Molina1, Jaime Andrade-Villanueva2, Juan Echevarria3, Ploenchan Chetchotisakd4, Jorge Corral5, Neal David6, Marco Mancini7, Lisa Percival7, Alexandra Thiry7, Donnie McGrath7
1Hopital Saint-Louis, Paris, France; 2Hospital Civil De Guadalajara, Guadalajara, Mexico; 3Hospital Nacional Cayetano Heredia, Lima, Peru; 4Khonkaen University, Khonkaen, Thailand; 5Hospital Interzonal Gral. De Agudos Oscar Alende, Buenos Aires, Argentina; 6Brooklyn Medical Centre, Western Cape, South Africa; 7Bristol-Myers Squibb, Wallingford, CT, USA
ABSTRACT
Background
ATV/r is as effective as LPV/r with more favorable lipid and GI profiles in treatment-experienced HIVinfected patients. Comparative data in ARV-naive patients are needed.
Methods
CASTLE is a randomized, open-label, multicenter, ongoing 96-week study to assess non-inferiority (10% margin) of ATV/r 300 mg/100 mg once-daily (QD) versus LPV/r 400 mg/100 mg twice-daily, both in combination with fixed-dose TDF 300 mg/FTC 200 mg QD, in treatment-naive patients. The primary end point was the proportion of patients with HIV RNA < 50 c/mL at week 48; planned secondary assessments included percent with HIV RNA < 400 c/mL, CD4 cell count change, and safety.
Results
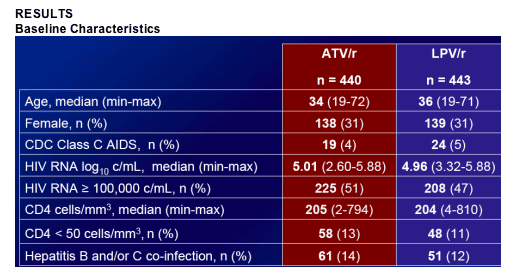
883 patients randomized, 878 treated. Baseline (BL) demographics and characteristics were well balanced. Median CD4 205 cells/mm3; median plasma HIV RNA 4.98 log10 c/mL.
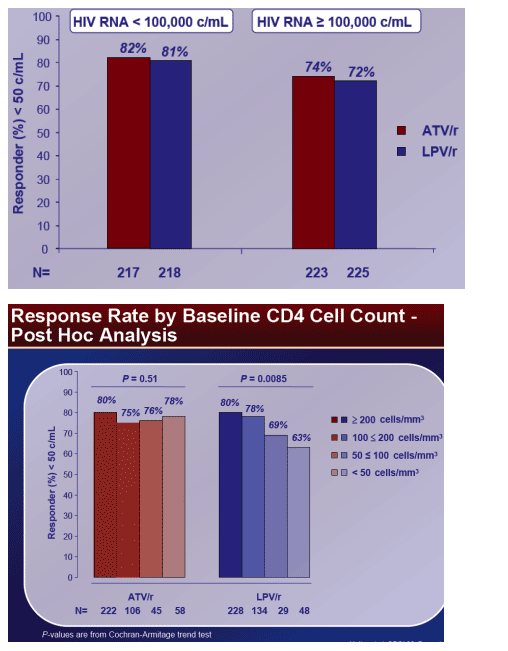
Confirmed Viral Response (CVR) (ITT), Noncompleters=Failure for <50 c/ml in this table immediately below is 78% for ATV/r vs 76% for LPV/r. For <400 c/ml, its 86% for ATV/r vs 82% for LPV/r. CVR for baseline CD4 <50 cells is 78% for ATV/r (n=58) vs 63% for LPV/r (n=48).
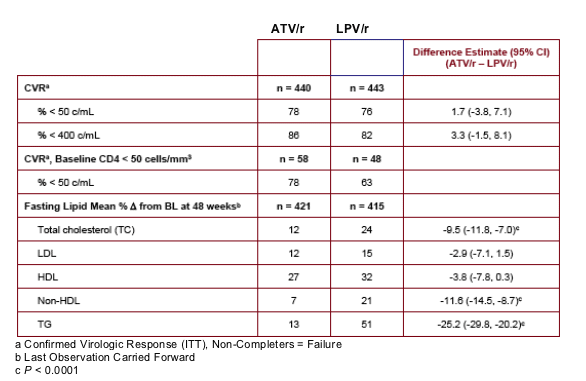
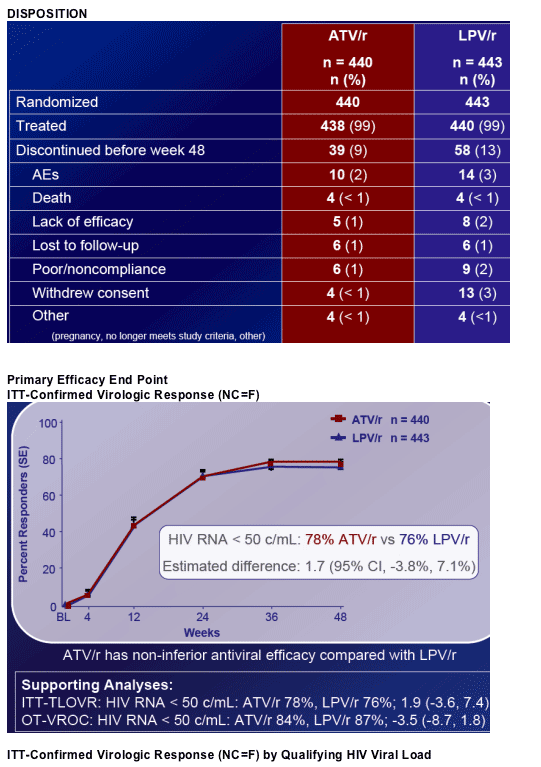
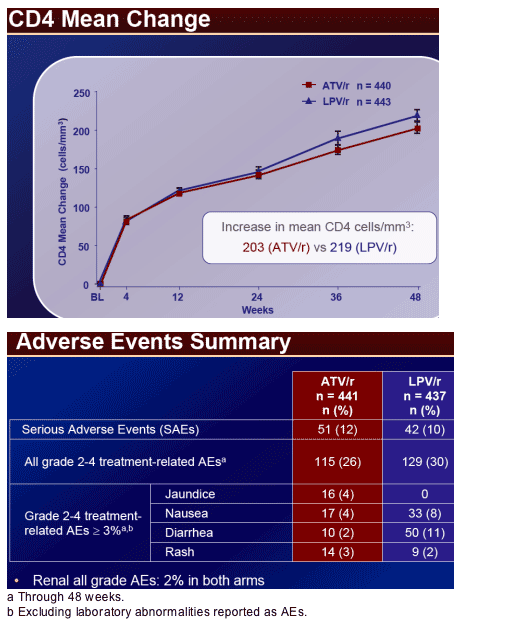
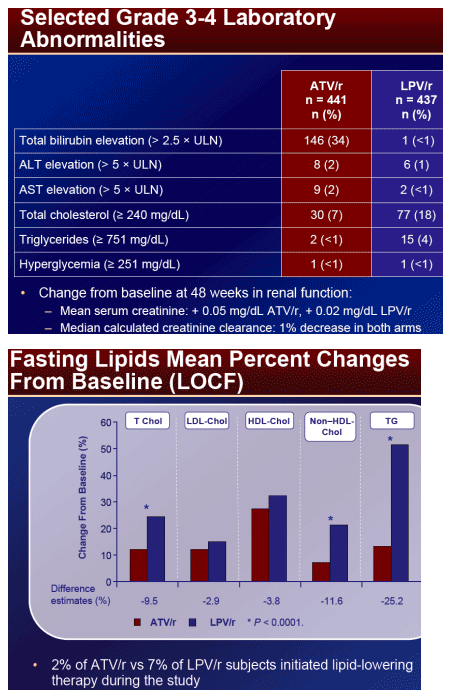
At week 48, mean CD4 increases from BL for ATV/r and LPV/r were 203 and 219 cells/mm3, respectively. Fewer patients on ATV/r (2%) than LPV/r (7%)* initiated lipid lowering therapy. The proportion of patients with a TC:HDL ratio > 5 at week 48 was 12% and 20% on ATV/r and LPV/r, respectively. Patients on ATV/r had a lower incidence of grade 2-4 treatment-related diarrhea (2% vs 11%) and nausea (4% vs 8%) than LPV/r. Grade 3-4 ALT/AST elevations were low (˛ 2%) in both arms. Discontinuations prior to week 48 were: ATV/r, 9%; LPV/r, 13%. AE-related discontinuations were 2% and 3% on ATV/r and LPV/r, respectively. Three patients (< 1%) discontinued ATV/r due to jaundice/hyperbilirubinemia.
Conclusions
In treatment-naive patients, ATV/r demonstrated similar efficacy, a lower incidence of GI-related AEs, and a significantly better lipid profile (TC, TG, non-HDL) compared to LPV/r. In combination with TDF and FTC, both ATV/r and LPV/r were generally well tolerated with few discontinuations through 48 weeks.
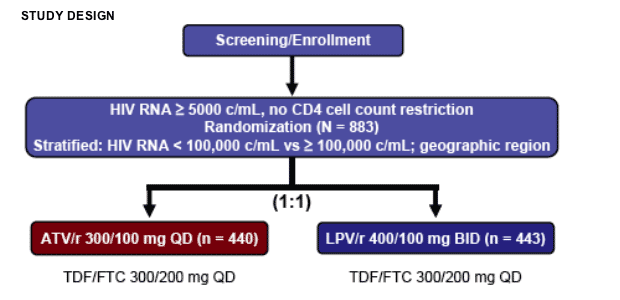
STUDY OBJECTIVES
Primary End Point
Proportion of subjects with HIV RNA < 50 c/mL at week 48
- Principal analysis: ITT-Confirmed Virologic Response (CVR) - (NC = F)
- Supportive analyses:
· ITT-TLOVR
· On-treatment-Virologic Response Observed Cases (OT-VROC)
Primary Objective
Demonstrate noninferiority of ATV/r once daily vs LPV/r twice daily based on primary end point
- _ (delta) -10%, ATV/r - LPV/r
Secondary End Points
--Immunologic response
--Safety and tolerability
--Changes in fasting lipids

|
| |
|
 |
 |
|
|