 |
 |
 |
| |
Metabolic Complications: A CROI 2008 Summary
|
| |
| |
David Alain Wohl, MD - The University of North Carolina
CROI Feb 2008, Boston, MA
Procrastination smacks of inexcusable sloth and is justifiably included among the seven deadly sins, but an advantage of waiting a few weeks after the 15th CROI to write a summary of the findings presented at the conference is the luxury of quiet contemplation of the data away from the inescapable and droning chatter that enveloped Boston for those few days in February. As anyone well into their 4th decade of life can attest, things are often better seen at a distance. Additionally, there is the benefit to be had by listening to the conversations that follow all such meetings - after the heat of initial surprise has dissipated.
A few interesting, if not controversial, presentations regarding metabolic complications of HIV were presented at CROI 2008, and what follows is a belated overview of the most noteworthy.
DAD Spells Trouble for ABC
One of the most brow-raising, if not brow-furrowing, presentations at the conference was a poster presentation by investigators from the D:A:D study - a large prospective observational study of several cohorts of HIV-infected patients in Europe, Australia and the US - examining the association between NRTI exposure and myocardial infarction (MI) (1). This work follows previous investigation by this team establishing a link between protease inhibitors and cardiovascular disease (CVD) (2). Although expecting to find a role for thymidine analogues (i.e. stavudine and zidovudine) in the development of MIs among study participants, the investigators were admittedly shocked when their analyses pointed to significant associations between exposure to either abacavir (ABC) or didanosine (ddI) and risk of MI.
The study includes data from as many as 33,347 individual HIV-infected patients with over 157,000 person-years of follow-up during which 517 MIs were recorded. Of these events, 192 were among persons who were receiving or recently had taken ABC. In a series of analyses looking at the impact of cumulative, recent and past exposure to ABC it appeared that only recent use (within the past 6 months) was associated with heart attack. Overall, recent ABC exposure was associated with a 90% increased risk of MI over and above baseline risk; among those with recent ddI use the risk of MI increased by 49%. However, the risk of MI with ABC or ddI seemed to be influenced greatly by underlying CVD risk. Those at highest CVD risk (i.e. >20% 10 year risk of MI as determined by the Framingham algorithm) appeared to suffer the greatest from exposure to these NRTIs.
The poster concludes that the risk of MI with ABC disappears with cessation of the drug; however, this finding seems to be deduced from the additive modeling of the timing and duration of ABC and ddI exposure the investigators performed. This analysis indicated that cumulative and past experience with the drug was not associated with MI risk when recent use was introduced to the model.
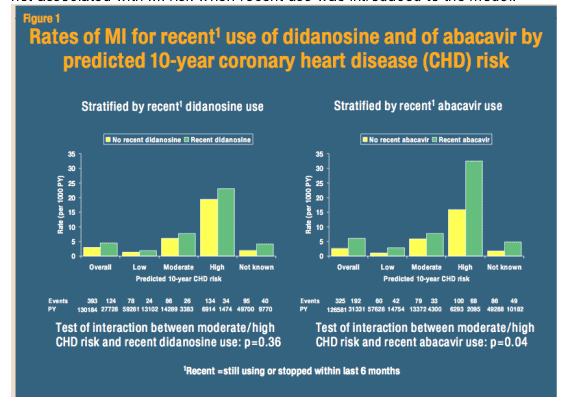
Importantly, it is clear that those who received ABC and those who did not in this non-randomized cohort study were not quite equal in terms of underlying CVD risk. Compared to patients receiving other NRTIs, those with recent ABC treatment were more likely to be male, older, diabetic and have personal or family history of CVD, hypertension, dyslipidemia or moderate to high risk of CVD - suggesting that such patients might have been channeled to ABC, given the perception that this drug might be more metabolically neutral than most other NRTIs. Recognizing this potential bias, the analyses controlled for these CVD risk variables, as well as HIV RNA levels and CD4 cell counts, and found that the associations between both ABC and ddI and the risk of MI persisted. Interestingly, no association between ABC or ddI and stroke was observed; however, only 195 such events were diagnosed during the study period.
What does this all mean? The D:A:D poster, and now the recently published paper, have led to the issuance of an FDA statement, re-examination of current treatment guidelines, a review of internal data by GlaxoSmithKline of patients receiving ABC (no increased MI risk seen, albeit sample size and duration of follow-up smaller than in D:A:D) and a flurry of editorial comments. Certainly, the attention reflects the surprising nature of the findings of the analysis and the seriousness of the adverse event being connected to these NRTIs, as well as the strengths of the D:A:D cohort.
While this study found a very concerning association between ABC and ddI and MI risk, the absolute risk of an MI in this study was small. In particular, the added MI risk of either ABC or ddI for those with little underlying predisposition to CVD was low. However, any increase in MI risk due to an antiretroviral is troubling (especially if you are one of the 'excess' MIs). But, before casting ABC from our midst it is important to recognize two points: a) this was a cohort study, not a randomized trial, and although the analyses attempted to adjust for imbalances between those receiving ABC and those who did not, it is unknown if other influences were not unaccounted for (i.e. statin use, the role of HLA-B*5701 genotype), and b) shelving of ABC leaves tenofovir (TDF) as the NRTI left standing yet, the D:A:D study did not have sufficient data to include this agent in their analysis of MI risk. So, although there is no indication at all that TDF is associated with MI, it has yet to be studied in D:A:D and reliance on this drug as an alternative to ABC in patients in whom CVD is a concern requires an assumption that it is free of the links to MIs that was found for ABC and ddI.
Therefore, these findings should be treated with a healthy dose of respect but it must also be recognized that they are, so far, singular in their conclusions, and have no clear biological explanation. Coupled with the news from AIDS Clinical Trials Group (ACTG) study A5202 - during which ABC/3TC performed sub-optimally compared to TDF/FTC among treatment naive individuals with HIV RNA levels above 100,000 c/mL - cautious use of ABC is advised until further data, perhaps from other large clinical cohorts, become available. Until then, care should be taken when considering initiation of ABC for those at greatest CVD risk and those with HIV RNA levels around 100,000 or more copies/mL. For those on the drug and doing well clinically, a discussion of the risks and benefits of continued therapy is warranted as is heightened attention to other modifiable CVD risk factors.
Inflammation SMARTs
The dramatic finding of the SMART Study was that CD4 cell count guided treatment interruption was more dangerous than continued exposure to HAART (3). The posited explanation for these results was that cessation of HIV therapy permitted a surge in viremia, triggering a reactive inflammatory response that was harmful to organ and host. Support for this pro-inflammatory hypothesis was found in at least two presentations, including one examining baseline markers of inflammation and thrombosis among SMART Study participants and a treatment interruption trial conducted in Thailand.
The Strategies for the Management of Antiretroviral Therapy (SMART) Study, is a seminal trial examining the effect of CD4 cell count guided ART treatment interruption on clinical outcomes. As has been well described, this large study found that treatment discontinuation was associated higher rates of mortality and adverse events, including those categorized as being cardiovascular, renal and hepatic, compared to continued HIV therapy. To help explain these results, the investigators examined baseline markers of inflammation and coagulation among the 85 (55 in the treatment interruption arm) deaths that occurred prior to the halting of the study by a data and safety monitoring board (4). These participants were matched by country, age, gender and randomization date with two controls who survived. Follow-up specimens were available for over 80% of cases who died and controls.
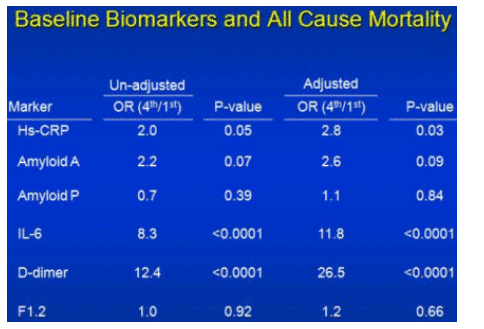
Compared to the controls, those who died had significantly lower CD4 cell counts, a history of diabetes or treatment for hypertension, and prior CVD. Importantly, many more of those who died were current smokers compared to controls (58% versus 32%, respectively). In addition to these baseline characteristics, the investigators examined the association between death and baseline levels of markers of inflammation and clotting that have been associated with CVD in HIV-uninfected individuals. These included the inflammation markers high sensitivity-C reactive protein (hsCRP), interleukin-6 (IL-6), serum amyloid A, and amyloid P and coagulation markers D-dimer and prothrombin fragment 1+2.
In an analysis adjusting for participant demographics, CVD risk factors, HIV viral load, CD4 cell count and viral hepatitis, two baseline biomarkers separated from the pack in terms of increasing the odds of death: IL-6 and D-dimer levels (adjusted odds ratio = 11.8 and 26.5, respectively; p <.0001 for both). For D-dimer, there was a strong and significant trend correlating the levels at baseline and the odds of death.
In a separate analysis, the authors examined the correlation between the change in these particular biomarkers over the first month of the study and death among a random subset of almost 250 participants in each of the study groups. IL-6 and D-dimer levels rose significantly more in the treatment discontinuation group than the treatment continuation group. D-dimer levels were found to correlate with month 1 viral loads such that the greatest increases were seen among those with the higher HIV RNA levels. Changes in IL-6 and D-dimer levels, as well as in hsCRP and amyloid P levels, were associated with risk of subsequent death. Importantly, the association between the biomarkers and death included non-CVD related mortality, suggesting deleterious effects of inflammation beyond the cardiovascular system and involving endothelium in disparate organ systems.
Another presentation at the conference from the STACCATO Study team reinforced the same general message regarding the link between inflammation during HIV treatment interruption and risk of badness (5). This was a trial in which antiretroviral therapy naive participants were treated with HAART for six months and then randomized to continue their regimen or undergo structured interruptions of their therapy based on CD4 cell count.
An analysis of blood specimens from 145 participants in Thailand (97 in the treatment interruption arm, 48 in the continuous therapy arm) was conducted to examine changes from baseline to week 12 in an array of biomarkers including those of endothelial dysfunction (s-VCAM-1 and p-selectin), inflammation (CRP, IL-6, IL-10, MCP-1, and MIP 1-alpha), and adipokines (leptin and adiponectin). For those who had interrupted their HIV therapy, samples from 12 weeks after resumption of treatment were also analyzed.
When looking at changes in markers between study arms, significant increases in MCP-1 and MIP 1-alpha, as well as a decrease in adiponectin were observed in the treatment interruption arm compared to the continued therapy arm. Among those that interrupted their HIV treatment, the levels of MCP-1 and s-VCAM-1 increased but when treatment resumed only a modest return to on-treatment levels was observed. Similarly, IL-10 and adiponection declined during treatment interruption and did not rise after treatment was resumed. Therefore, treatment interruption seemed to cause lasting changes in these markers that were not completely reversed by the re-initiation of HIV therapy.
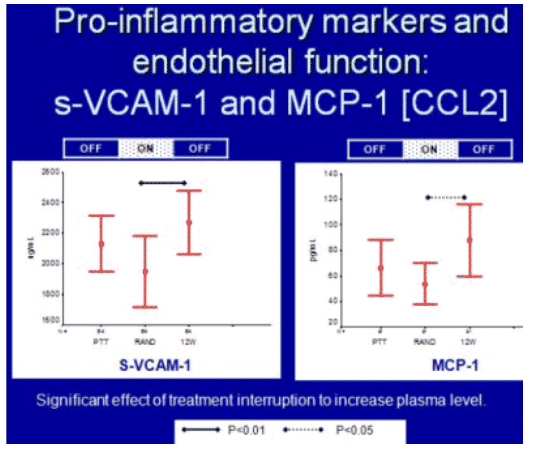
As in the SMART Study, there was a strong relationship between HIV RNA level and these biomarkers; however, curiously, in this study IL-6 was not correlated with HIV viremia.
What does this all mean? These results provide strong support for the hypothesis that discontinuation of HIV therapy leads to inflammatory and endovascular reactions and that these in turn are associated with all-cause mortality. Further, if one was not already convinced of the hazards of treatment interruption these studies demonstrate concerning evidence that the resurgence of HIV viremia leads to the expression of evil humors. For the audience of infectious diseases and HIV specialists, the presenter of the SMART Study data, Dr. Kuller, an epidemiologist from the University of Pittsburg, framed the significance of the strength of the relationship between D-dimer and IL-6 and death by calling the findings "extraordinary" and of a much greater magnitude than that seen in HIV-uninfected cohorts. He raised the interesting question of whether, given these data, IL-6 and D-dimer levels or other markers should be monitored among individuals with HIV infection. Certainly, further study of the utility of these and other biomarkers of inflammation and coagulation can be expected. The close relationship between these markers and control of HIV viremia suggest they may be of little use in those successfully treated with HAART but they may be more meaningful for individuals not receiving HIV therapy. Application of interventions that aim to reduce inflammation are already under investigation and one small proof of concept study examining pentoxifylline, a TNF-alpha antagonist, was presented at CROI and found reductions in serum VCAM-1 and CXCL10 (a interferon-gamma induced protein), as well as improvement in flow mediated vasodiliation - a marker of endothelial function (6).
Bones
Brittle bones is a consequence of aging but may be accelerated during HIV infection and treatment. Several studies have demonstrated reduce bone density among HIV-infected persons naive to HIV therapy and such individuals often, but not always, have risk factors for osteopenia/porosis including history of low weight, smoking, sedentary lifestyle, vitamin D deficiency, among others (7). Further, declines in done density have been observed during treatment with HIV medications (8). A handful of studies at CROI examined bone health among those receiving HIV therapy including a small randomized trial of three different antiretroviral treatment strategies and two studies during which NRTI therapy was manipulated.
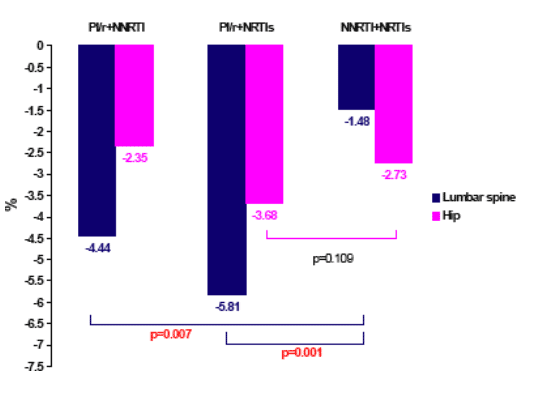
Bone mineral density (BMD) was assessed at the hip and lumbar spine of 71 treatment naive patients randomized 2:1:1 to PI/r+NNRTI vs. PI/r+2 NRTIs vs. NNRTI+2 NRTIs as part of a French ARNS trial (9). Lopinavir/r (LPV/r) + efavirenz (EFV) was the most commonly used combination in the NRTI-sparing arm. LPV/r or indinavir/r (IDV/r) in combination with ZDV/3TC was used in the PI/r + NRTI arm. EFV + AZT/3TC was the most commonly used combination in the NNRTI + NRTI arm. At baseline, 31% of the participants had osteopenia at either the hip and/or spine (PI/r + NNRTI (33%); PI/r + NRTI (32%); NNRTI + NRTI (25%) while only two patients (3%) had osteoporosis (all were in the PI/r + NNRTI arm). Baseline CD4 cell count was between 200 and 250/mm3, more than half the participants smoked and the body mass index was fairly low at around 23 kg/M2.
At 48 weeks of treatment there were significant declines in BMD in all study groups; however, the greatest declines, especially at the spine, were observed among those receiving a regimen containing a PI/r.
Another randomized trial also looked at NNRTI- vs. PI-based regimens and bone density and found very different results (10). Abbott 613 was a trial examining a strategy of antiretroviral simplification to LPV/r monotherapy during which 155 treatment naive patients were randomized 1:2 to receive ZDV/3TC+EFV or ZDV/3TC+LPV/r. Those in the latter group were eligible to simplify their regimen to LPV/r alone if they achieved lasting viral suppression. Whole body DEXA scans were conducted at baseline and 96 weeks and results were available from 74 LPV/r and 32 EFV assigned patients and the results of these scans were presented.
Mean total BMD at baseline was 1.17 +/- 0.10 g/cm2 and 1.19 +/- 0.12 g/cm2 for LPV/r and EFV, respectively. In a multivariable analysis, baseline BMD was independently associated with lower weight, non-black race and lower baseline HIV RNA levels.
After 96 weeks of treatment, similar mean decreases in BMD were observed in both the PI/r and NNRTI treatment groups: -2.5% in the LPV/r and -2.3% in the EFV group (p=0.86). Within treatment groups, changes from baseline in BMD were statistically significant (p<0.01) for each group.
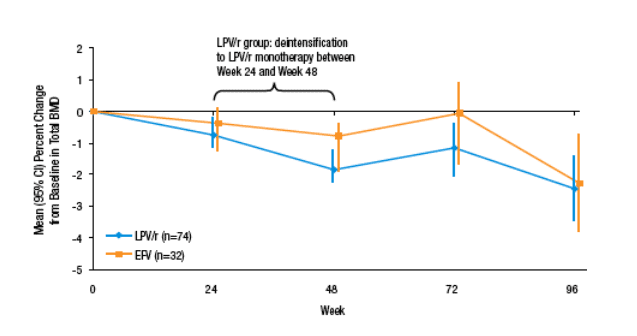
A 5% or greater loss of BMD was observed in 16 (15%) subjects. Subjects with lower baseline CD4 cell count, higher baseline fasting glucose and of non-black race demonstrated a higher risk for > 5% decrease in BMD. Changes in BMD were not correlated to baseline BMD, other parameters of glucose metabolism or changes in body composition. Likewise, simplification to LPV/r alone had no evident effect on BMD.
Lastly, investigators from the Swiss Cohort Study investigated serum (not bone-sepcific) alkaline phosphatase levels, a potential marker of bone demineralization, among their patients to determine whether TDF use was associated with changes on this biomarker (11). They examined data from183 treatment naive patients starting TDF-containing regimens, 515 starting TDF-sparing therapy, 180 reinitiated TDF and 200 reinitiated TDF-sparing regimens, as well as 73 who discontinued TDF. Serum alkaline phosphatase was measured at baseline, 1, 3 , 6, 12 months of therapy.
TDF use as opposed to TDF-sparing regimens, was associated with a significant increase in alkaline phosphatase both in naive and experienced patients through 12 months, while TDF discontinuation was associated with a significant decrease in this marker.
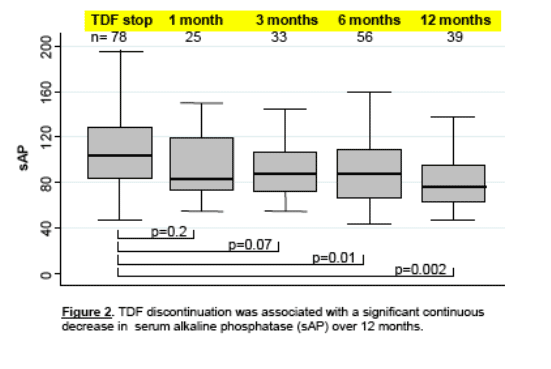
In a multivariate analysis increased risk of increased alkaline phosphatase included systolic and TDF. Increases in alkaline phosphatase, also a marker of liver damage, did not correlate with increased ALT; HCV infection or decreased glomerular filtration rate (GFR).
What does this all mean? The first two trials both found that pre-treatment osteopenia was prevalent but osteoporosis was, fortunately, rare. In both studies continued loss of bone density was observed during HIV therapy. Whether, the decline in BMD during HIV treatment is faster or slower than losses occurring prior to antiretroviral therapy is not clear. In the ARNS trial, PI treatment was associated with bone loss but this study was small and the different treatment regimens within each arm make the study messy and challenging to interpret. The results of the DEXA scan analyses from the Abbott 613 study demonstrated that patient characteristics influence bone loss during therapy with those who are non-black, having a lower CD4 cell count and glucose intolerance at greater risk. In this study there was no difference seen between the NNRTI and PI/r used, although the effect of earlier discontinuation of ZDV/3TC on the 96 week data is unclear.
The link between TDF and serum alkaline phosphatase levels adds further evidence of a role for this nucleotide analogue in reduced bone mineralization. Previous studies have demonstrated a greater loss of BMD with TDF compared to d4T and ZDV. Renal losses of phosphorus due to TDF, may trigger compensatory release of this mineral from bone stores, leading to reduced BMD in some individuals. (note from Jules: the strength of the cnnection between serum alkaline phosphatase levels and BMD may need further data).
All three studies strongly suggest that monitoring of BMD should be more widespread. Clearly, too few patients with HIV infection are being screened for low BMD and recent results from an AIDS Clinical Trials Group (ACTG) study demonstrating the safety and efficacy of alendronate for the treatment of reduced BMD in HIV+ patients supports the use of this medication in this population (12). The ACTG trial developed a check list for investigators to use when considering which patients to screen with DEXA scans for low BMD. This list can also be useful to clinicians and includes:
1. Does the patient smoke?
2. Does the patient weigh less than 70 kg or have a body mass index (BMI) of less than 25?
3. Has the patient been treated with corticosteroids at any time for a total of at least 14 days? (Note that systemic steroid treatment for 4 weeks or more within the 6 months prior to study entry is an exclusion criterion.)
4. Has the patient received antiretroviral treatment of any kind for a total of at least 2 years?
5. Has the patient ever experienced a significant weight loss during adult life?
The more 'yes' answers, the more likely the patient has low BMD.
NATAP's Jules Levin is spearheading efforts to invigorate the HIV research community to tackle bone issues as part of a strategy for helping those with HIV live not only longer but healthier lives. It is certain that additional work will be done to understand the contribution of individual antiretrovirals to BMD changes and approaches to minimize and prevent loss of bone that can lead to devastating and debilitating fractures.
Body Shape and ZDV/3TC
ACTG study A5142 convinced us that ZDV/3TC is associated with lipoatrophy (13). The SWEET Study examined the effect of switching from ZDV/3TC to TDF/FTC on limb fat among 250 patients on stable therapy with ZDV/3TC and EFV (14). Participants were randomized 1:1 to continue their pre-study regimen or swap the ZDV/3TC for TDF/FTC. A hundred subjects underwent DEXA scanning to look at body fat changes subsequent to the switch. While whole body fat increased in both study groups during the study, limb fat declined among those remaining on ZDV/3TC compared to an rise among those randomized to start TDF/FTC. Importantly, among the participants who had been on ZDV/3TC for 3 years or more (about half the cohort), switching to TDF/FTC produced only a slight increase in limb fat while those continuing on ZDV/3TC saw little change in limb fat - perhaps because any ZDV/3TC induced fat loss had already occurred.
Changes in lumbar and hip BMD were not significant in either study arm during 48 weeks of study. As in other studies, TDF lead to small but statistically significant greater decreases in estimates of glomerular filtration compared to ZDV/3TC.
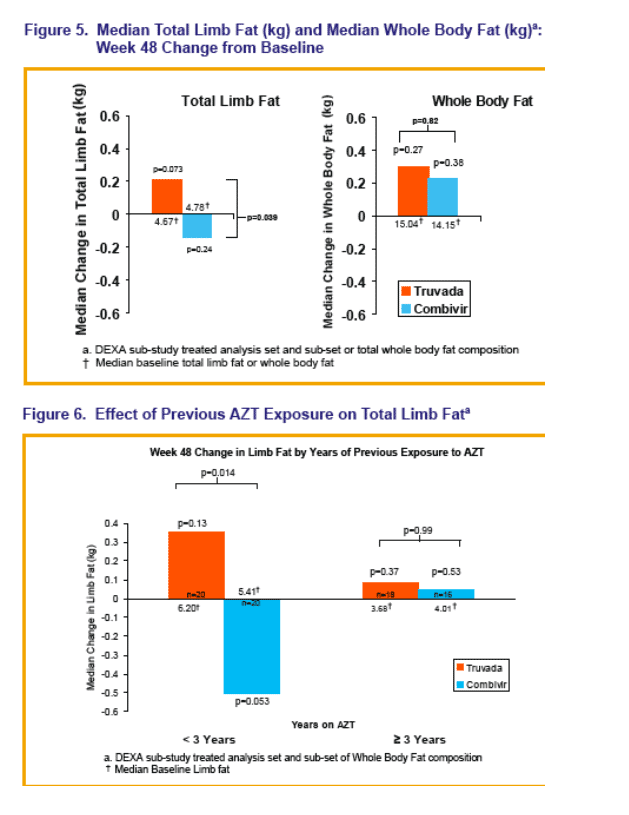
What does this mean? ZDV/3TC can cause limb fat loss and that switching to TDF/FTC can reverse some of this loss but has limited effect over 48 weeks on lipoatrophy among those receiving ZDV/3TC for 3 or more years.
References
1. Sabin C, et al. Do Thymidine Analogues, Abacavir, Didanosine and Lamivudine Contribute to the Risk of Myocardial Infarction? The D:A:D Study. Proceedings of the 15th Conference on Retroviruses and Opportunistic Infections 2008. Boston, MA: Abstract 957c.
2. Friis-Moller N, Reiss P, Sabin CA, et al. Class of antiretroviral drugs and the risk of myocardial infarction. N Engl J Med. 2007;356:1723-1735.
3. Strategies for Management of Antiretroviral Therapy (SMART) Study Group, El-Sadr WM, Lundgren JD, et al. CD4+ count-guided interruption of antiretroviral treatment. N Engl J Med. 2006;355(22):2283-2296.
4. Kuller L, et al. Elevated levels of interleukin-6 and D-dimer are associated with an increased risk of death in patients with HIV. Proceedings of the 15th Conference on Retroviruses and Opportunistic Infections 2008. Boston, MA: Abstract 139.
5. Calmy A, et al. HIV activates markers of cardiovascular risk in a randomized treatment interruption trial: STACCATO. Proceedings of the 15th Conference on Retroviruses and Opportunistic Infections 2008. Boston, MA: Abstract 140.
6. Gupta S, et al. A pilot study of the TNF-alpha inhibitor pentoxifylline to improve HIV-related endothelial dysfunction. Proceedings of the 15th Conference on Retroviruses and Opportunistic Infections 2008. Boston, MA: Abstract 955.
7. Brown TT and McComsey GA. Osteopenia and osteoporosis in patients with HIV: a review of current concepts. Curr Infect Dis Rep. 2006. Mar;8(2):162-70.
8. Pozniak AL, Gallant JE, Dejesus E, et al. Tenofovir disoproxil fumarate, emtricitabine, and efavirenz versus fixed-dose zidovudine/lamivudine and efavirenz in antiretroviral-naive patients: virologic, immunologic, and morphologic changes-a 96-Week analysis. J Acquir Immune Defic Syndr. 2006;43:535-540.
9. Duvivier C, et al. First-line PI-containing Regimens Enhance Decreased Bone Mineral Density Greater than NNRTI-containing Regimen in HIV-1-infected Patients: A Substudy of the HIPPOCAMPE-ANRS 121 Trial. Proceedings of the 15th Conference on Retroviruses and Opportunistic Infections 2008. Boston, MA: Abstract 967.
10. Brown T, et al. Bone Mineral Density 96 Weeks after ART Initiation: A Randomized Trial Comparing Efavirenz-based Therapy with a Lopinavir/Ritonavir-containing Regimen with Simplification to LPV/r Monotherapy. Proceedings of the 15th Conference on Retroviruses and Opportunistic Infections 2008. Boston, MA: Abstract 966.
11. Fux C, et al. Tenofovir Use Is Associated with an Increase in Serum Alkaline Phosphatase in the Swiss HIV Cohort Study. Powderly W. Contributions of Age-Related Comorbidities. Proceedings of the 15th Conference on Retroviruses and Opportunistic Infections 2008. Boston, MA: Abstract 968.
12. McComsey GA, Kendall MA, Tebas P, et al. Alendronate with calcium and vitamin D supplementation is safe and effective for the treatment of decreased bone mineral density in HIV. AIDS. 2007 Nov 30;21(18):2473-82.
13. Haubrich R, Riddler S, DiRienzo G, et al. Metabolic outcomes of ACTG 5142: a prospectiv, randomized, phase III trial of NRTI-, PI-, and NNRTI-sparing regimens for intial treatment of HIV-1 infection. Paper presented at: Abstracts and Posters of the 14th Conference on Retroviruses and Opportunistic Infections, February 25-28, 2007; Los Angeles, CA. Abstract 38.
14. Moyle G, et al. Switching from Combivir to Truvada preserves limb fat: Results of a DEXA sub-study of the 48 week randomized study. Proceedings of the 15th Conference on Retroviruses and Opportunistic Infections 2008. Boston, MA: Abstract 938.
|
| |
|
 |
 |
|
|