 |
 |
 |
| |
Women, Younger Age & Black Race Associated with Shorter Time to Viral Failure: Effect of Baseline Characteristics on Treatment Outcomes in ACTG 5142: A Prospective, Randomized, Phase III Trial of NRTI-, PI-, and NNRTI-sparing Regimens for Initial Treatment of HIV-1 Infection
|
| |
| |
Reported by Jules Levin
Sharon A. Riddler*1, Richard H. Haubrich2, Greg DiRienzo3, Lynne Peeples3, William G. Powderly4, Kevin W. Garren5, David L. Butcher6, James F. Rooney7, Diane V. Havlir8, John W. Mellors1, and the AIDS Clinical Trials Group 5142 Study Team
University of Pittsburgh, Pittsburgh, PA, USA1; University of California, San Diego, San Diego, CA, USA2; Harvard School of Public Health, Statistical and Data Analysis Center, Boston, MA, USA3; University College, Dublin, Ireland4; Abbott
Laboratories, Abbott Park, IL, USA5; Bristol-Myers Squibb, Plainsboro, NJ, USA6; Gilead Sciences, Foster City, CA, USA7 and University of California, San Francisco, San Francisco, CA, USA8
BACKGROUND
· Current DHHS guidelines recommend efavirenz or ritonavir-boosted PI regimens as initial therapy for all patients.
· A5142 was a large randomized study of potent class sparing antiretroviral regimens for initial therapy of HIV infection.
· It is important to assess the impact of baseline characteristics on the virologic response and toxicity of these regimens.
· We explored the effect of baseline characteristics on the outcome of treatment in A5142.
Summary of Results
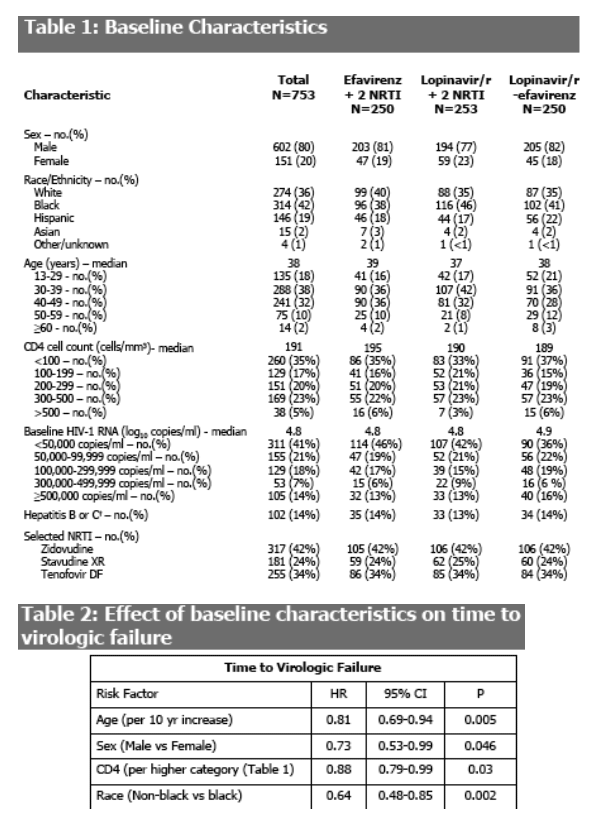
A shorter time to virologic failure was associated with younger age, female sex, lower CD4 and black race (Table 2).
Baseline HIV RNA (<100,000 vs ≥100,000 copies/ml) was not associated with the overall risk of virologic failure (log rank P=0.81).
None of the baseline factors examined was significantly associated with the risk of regimen completion.
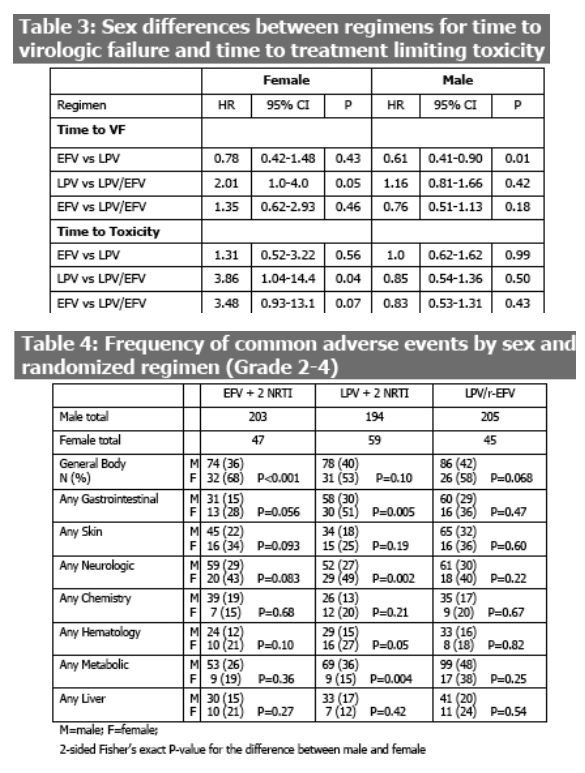
Sex differences by regimen were observed. For women, the risk of virologic failure was significantly lower for LPV/r-EFV compared to LPV/r (Table 3). In pairwise comparisons, the NRTI sparing regimen of LPV/r-EFV had longer time to toxicity related discontinuation than LPV/r + 2 NRTI (HR 3.86; P=0.04) and EFV + 2 NRTI (HR 3.48; P=0.07). For men, the differences between regimens were similar to the overall study results with a longer time to virologic failure for the EFV arm and no difference between arms for the time to toxicity.
For women, the time to regimen completion was longer for LPV/r-EFV group compared with LPV/r (P=0.014) and EFV (P=0.08).
Racial differences between regimens were not observed.
Among subjects with baseline CD4<50 cells/mm3 (N=177), the median time to CD4>200 cells/mm3 was 55 weeks and was not different between arms (log rank P=0.77).
Toxicity related discontinuation of one or more regimen components occurred in 23/151 (15%) of women and 111/602 (18%) of men. The specific first treatment limiting toxicities were similar between men and women with the exception of fasting triglycerides which occurred in 15 men (11 LPV/r-EFV and 4 LPV/r) but none of the women.
In general, Grade 2 or higher symptoms were more frequent in women compared
to men (Table 4), however, rates of laboratory toxicities were similar, except for metabolic events in the LPV/r group which were more frequent in men.
Conclusions
In this large randomized trial of class-sparing regimens:
Virologic failure was more likely with lower baseline CD4 cell count,
black race, younger age and female sex.
Women had a longer time to virologic failure and treatment limiting toxicity with the nucleoside sparing regimen of LPV/r-EFV
All three regimens had a similar time to reach CD4 cell count >200 among participants with baseline CD4<50 cells/mm3
Women were more likely to experience (or report) symptoms though the proportion of men and women with treatment limiting toxicity were similar
OBJECTIVES & ENDPOINTS
OBJECTIVES:
To assess the impact of baseline characteristics on the time to virologic failure, regimen completion and toxicity related discontinuation
To describe sex related differences in toxicity to antiretroviral therapy in A5142
ENDPOINTS
Virologic failure was defined as any of the following: lack of suppression by 1.0 log10 or rebound before week 32, or failure to suppress to <200 copies/mL afte week 32
Regimen completion was defined as the first occurrence of virologic failure or toxicity related discontinuation of any regimen component (including within class switch)
METHODS
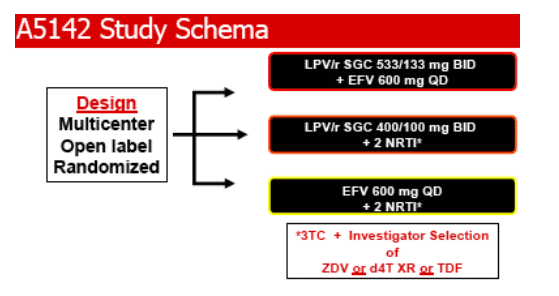
· Phase III, randomized, multicenter, open-label, 96 week trial
· 753 subjects (20% women) were treated on study in three arms:
· Efavirenz + 2 NRTI (EFV)
· Lopinavir/r + 2 NRTI (LPV/r)
· Lopinavir/r + Efavirenz (LPV/r-EFV)
· NRTI were selected prior to randomization from ZDV, d4T XR or TDF, each with 3TC
· Randomization stratified on: HIV RNA (<100,000 or ≥100,000 copies/mL), hepatitis B or C coinfection, and NRTI choice
· Major inclusion criteria
Antiretroviral naive
Any CD4
HIV RNA ≥2000 copies/mL
· Study visits occurred at pre-entry, entry, and weeks 1, 4, 8, 12, 16, 20, 24, and every 8 weeks thereafter
· Adverse clinical and laboratory events were assessed and graded using the DAIDS adverse event grading scale (1992). Treatment limiting toxicity was determined by the site investigator.
· Statistical considerations:
--All analyses were intent-to-treat
--Cox proportional hazard models were used to analyze associations of preselected baseline characteristics with risk of virologic failure, regimen completion and treatment limiting toxicity. The models were stratified on the three randomization strata.
|
| |
|
 |
 |
|
|