 |
 |
 |
| |
Study M05-730 Primary Efficacy Results at Week 48:
Phase 3, Randomized, Open-Label Study of Lopinavir/ritonavir (LPV/r) Tablets Once Daily (QD) versus Twice Daily (BID), Co-Administered with Tenofovir DF (TDF) + Emtricitabine (FTC) in Antiretroviral-Naive (ARV) HIV-1 Infected Subjects
|
| |
| |
View the PDF version of the M05-730 study here
Reported by Jules Levin
CROI 2008, Boston
Gathe J1, da Silva BA2, Loutfy M3, Podzamczer D4, Rubio R5, Gibbs S2, Marsh T2, Naylor C2, Fredrick L2, Bernstein B2
1Therapeutic Concepts, Houston, Texas, USA; 2Abbott Laboratories, Abbott Park, Illinois, USA; 3Maple Leaf Medical Clinic,
Toronto, Ontario, Canada; 4Hospital Universitari de Bellvitge, Barcelona, Spain; 5Hospital 12 Octubre, Madrid, Spain
15th Conference on Retroviruses and Opportunistic Infections (CROI) · February 3-6, 2008 · Boston, Massachusetts
AUTHOR CONCLUSIONS
QD and BID dosing of LPV/r tablets have similar overall safety and tolerability.
The antiviral activity of LPV/r tablet dosed QD is non-inferior to BID dosing in ARV-naive subjects. Response to therapy was independent of baseline viral load or CD4 cell count and no PI or tenofovir resistance was noted in either the QD or BID LPV/r treatment groups.
"....In the primary efficacy analysis at Week 48, 77% of QD-treated and 76% of BID-treated subjects achieved a viral load <50 copies/mL by ITT NC=F analysis (Figure 2a).....The difference in response rates (QD minus BID) and 95% CI was 1% (-5% to 8%), which confirmed the non-inferiority of the QD regimen to the BID regimen, as the lower bound of the confidence interval was within the pre-specified margin of -12%. The on treatment data confirms this finding (Figure 2b).....A sensitivity analysis adjusting for the baseline imbalance in HIV-1 RNA level was consistent with the primary analysis, with an estimated difference (95% CI) in response rates of 1% (-6% to 7%).....Within subgroups defined by baseline HIV-1 RNA (<100,000 copies/mL or ≥100,000 copies/mL) or baseline CD4+ T-cell count (<50 cells, 50-200 cells and ≥200 cells/mm3), a similar proportion of QD- and BID-treated subjects achieved HIV-1 RNA <50 copies/mL at Week 48 (Figure 3)..... Rate of study drug-related diarrhea of moderate or greater severity was similar between groups (QD 17%, BID 15%, p=0.671). Rate of other events of interest and grade 3+ lab abnormalities were also similar in the two treatment groups (Table 4).....
....Comparison of SGC to Tablet through Week 8:
There were no statistically significant differences between the tablet and SGC formulations in the following areas: the number of subjects discontinuing due to gastrointestinal adverse events or other adverse events; the incidence of treatment-emergent adverse events of diarrhea of any severity and of moderate or greater severity and related to the study drug; the proportion of subjects with Grade 3+ lab abnormalities; or the mean change from baseline for total cholesterol or triglycerides at any time point during the first 8 weeks of treatment......In those subjects who switched from SGC to tablet, subjects overwhelmingly preferred the LPV/r tablet over the SGC (p< 0.001), with 75% (QD) and 80% (BID) preferring the tablet and only 3% (QD) and 5% (BID) preferring the SGC."
AUTHOR DISCUSSION
Previous studies have demonstrated patient preference for the tablet formulation after switching from the SGC.2-4 Consistent with this data, questionnaire data after the switch from SGC to tablet in Study M05-730 also demonstrated that patients preferred the tablet formulation over the SGC.
This study is the largest study to date comparing QD and BID dosing of LPV/r, and the first utilizing the tablet formulation. Overall, during the first 48 weeks of treatment, no clinically important differences were identified in the safety or tolerability of QD vs. BID dosing of LPV/r. Notably, and in contrast to Study M02-418, differences in rates of diarrhea were not observed when QD and BID dosing of LPV/r were compared.5
In contrast to previously reported studies with smaller numbers of subjects receiving QD LPV/r and utilizing the SGC formulation of LPV/r, antiviral activity was independent of baseline HIV-1 RNA or CD4+ T-cell count.6-7
BACKGROUND
Lopinavir (LPV) is an HIV protease inhibitor (PI) that is co-formulated with ritonavir (r), which functions as a pharmacokinetic enhancer. Antiviral activity of LPV/r has been demonstrated in antiretroviral (ARV)-naive and PI-experienced patients.
LPV/r was initially approved as a twice-daily administered PI. Data from a prior LPV/r study (Study 418) compared once-daily (QD) and twice-daily (BID) LPV/r administration in antiretroviral-naive patients using the soft gel capsule (SGC).
The QD LPV/r regimen was shown to be non-inferior to the BID regimen; however, diarrhea rates were higher in the QD group.
The tablet formulation of LPV/r reduces the pill burden of the standard 800/200 mg LPV/r total daily dose from 6 capsules to 4 tablets, and, unlike the SGC, does not require refrigeration, and may be taken with or without food.
Study M05-730 was performed to assess the safety and tolerability of the tablet and SGC formulations dosed BID and QD through 8 weeks of treatment followed by a comparison of the LPV/r tablet dosed QD versus BID administered with NRTIs through 96 weeks of treatment. Efficacy and safety data are presented through 48 weeks, representing the pre-specified primary efficacy analysis timepoint.
METHODS
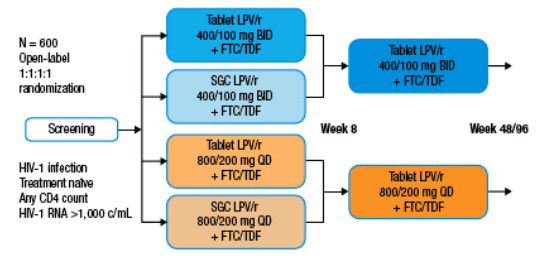
Study M05-730 is a 96-week Phase 3, open-label, randomized, multi-center, multi-country study that enrolled ARV-naive subjects with HIV-1 RNA ≥ 1000 copies/mL and any CD4+ T-cell count.
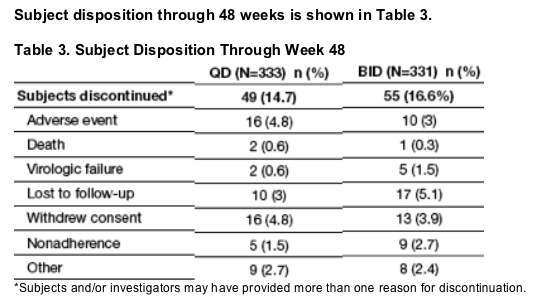
664 ARV-naive HIV-1 infected subjects were randomized 1:1:1:1 to LPV/r QD SGC, BID SGC, QD tablet, or BID tablet for 8 weeks (wks).
All subjects received emtricitabine (FTC) 200 mg QD and tenofovir disoproxil fumarate (TDF) 300 mg QD.
At Week 8, all subjects receiving SGC were switched to the tablet formulation maintaining their same randomized dosing schedule.
Subjects were evaluated every 2 weeks through Week 16, every 8 weeks through Week 48, then every 12 weeks through Week 96.
Figure 1. Study M05-730 Design
Primary endpoints
Efficacy: Proportion of subjects with plasma HIV-1 RNA level <50 copies/mL at Week 48
Safety: Proportion of subjects with adverse events of diarrhea during the first 8 weeks of study drug treatment
Efficacy Analysis:
Primary efficacy endpoint: proportion of subjects with HIV-1 RNA <50 copies/mL at Week 48, using an intent-to-treat, noncompleter=failure (ITT NC=F) approach comparing QD and BID groups. The pre-specified noninferiority margin was 12%.
Secondary analyses include the mean change from baseline in CD4+ T-cell count and the emergence of viral resistance through 48 weeks.
Safety Analysis:
Primary safety endpoint: proportion of subjects reporting a treatment-emergent adverse event of diarrhea (without regard to severity or causality) during the first 8 weeks of dosing. The difference between tablet and SGC formulations was assessed using the Cochran-Mantel-Haenszel (CMH) test stratified by dosing group (QD vs. BID).
Additional analyses include the proportion of subjects reporting treatment emergent adverse events, grade 3+ lab abnormalities, and mean changes from baseline for lab determinations through 48 weeks.
Resistance Analysis:
The proportion of subjects who developed resistance to each study drug was compared between QD and BID dosing groups using Fisher's exact test.
Resistance to drugs is defined by the IAS-USA Panel.1
Lopinavir resistance was also more conservatively defined as the presence of one or more of the following mutations in protease: I47V/A, G48V, I50V, V82A/F/T/S, I84V, L90M, or the presence of 3 or more of the following: L10F/I/R/V, K20M/R, L24I, V32I, L33F, M36I, M46I/L, F53L, any change to I54, A71V/T, G73S.
RESULTS
Baseline demographics are shown in Tables 1 and 2.
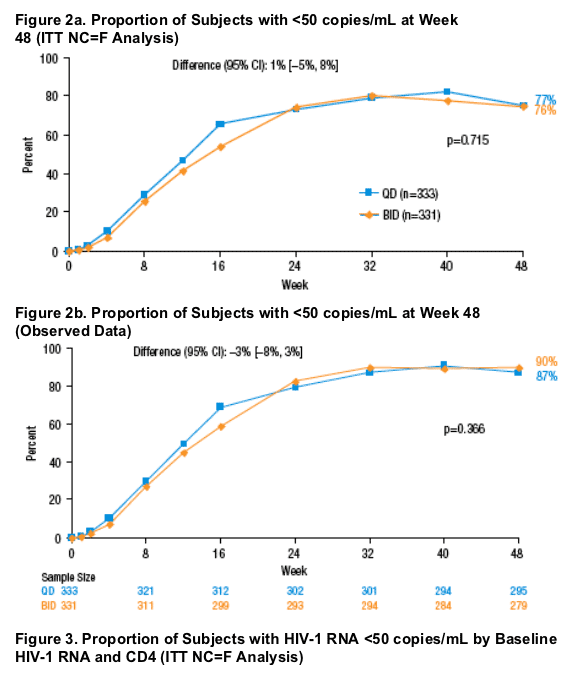
Primary Efficacy Endpoint Through Week 48
In the primary efficacy analysis at Week 48, 77% of QD-treated and 76% of BID-treated subjects achieved a viral load <50 copies/mL by ITT NC=F analysis (Figure 2a).
The difference in response rates (QD minus BID) and 95% CI was 1% (-5% to 8%), which confirmed the non-inferiority of the QD regimen to the BID regimen, as the lower bound of the confidence interval was within the pre-specified margin of -12%. The on treatment data confirms this finding (Figure 2b).
A sensitivity analysis adjusting for the baseline imbalance in HIV-1 RNA level was consistent with the primary analysis, with an estimated difference (95% CI) in response rates of 1% (-6% to 7%).
Within subgroups defined by baseline HIV-1 RNA (<100,000 copies/mL or ≥100,000 copies/mL) or baseline CD4+ T-cell count (<50 cells, 50-200 cells and ≥200 cells/mm3), a similar proportion of QD- and BID-treated subjects achieved HIV-1 RNA <50 copies/mL at Week 48 (Figure 3).
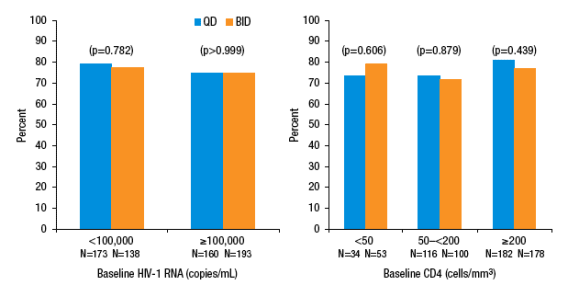
Similar mean increases from baseline in CD4+ T-cell count at Week 48 were observed in the QD and BID treatment groups (186 and 197 cells/mm3, respectively; p=0.350).
Resistance Analysis Through Week 48
Seventeen subjects had resistance testing available (10 QD and 7 BID) through Week 48. No resistance to lopinavir or tenofovir DF was observed. Three subjects (2 QD and 1 BID) developed the M184V mutation.
Safety Analysis Through Week 48
Rate of study drug-related diarrhea of moderate or greater severity was similar between groups (QD 17%, BID 15%, p=0.671). Rate of other events of interest and grade 3+ lab abnormalities were also similar in the two treatment groups (Table 4).
While mean increases were observed in all lipid parameters in both groups, the mean LDL-C:HDL-C ratio decreased substantially in both groups (Table 5).

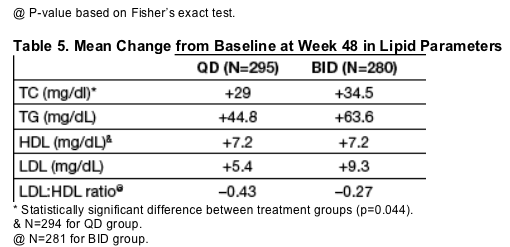
Comparison of SGC to Tablet through Week 8
There were no statistically significant differences between the tablet and SGC formulations in the following areas: the number of subjects discontinuing due to gastrointestinal adverse events or other adverse events; the incidence of treatment-emergent adverse events of diarrhea of any severity and of moderate or greater severity and related to the study drug; the proportion of subjects with Grade 3+ lab abnormalities; or the mean change from baseline for total cholesterol or triglycerides at any time point during the first 8 weeks of treatment.
In those subjects who switched from SGC to tablet, subjects overwhelmingly preferred the LPV/r tablet over the SGC (p< 0.001), with 75% (QD) and 80% (BID) preferring the tablet and only 3% (QD) and 5% (BID) preferring the SGC.
References
1. Johnson VA, Brun-VŽzinet F, Clotet B, et al. Update of the drug resistance mutations in HIV-1: 2007. Topics in HIV Medicine. 2007;15:119-125.
2. Schrader S, Chuck SK, Rahn LW, et al. Switching to lopinavir/ritonavir (LPV/r) tablets once daily (TAB-QD) from soft-gel capsule dosed BID/QD (SGC-BID/SGC-QD) led to significant improvements in tolerability, diarrhea, anti-diarrheal medication use, and satisfaction. 8th International Workshop on Adverse Drug Reactions and Lipodystrophy in HIV. 2006. San Francisco, CA.
3. Ofotokun I, Chuck SK, Rivas M, et al. Switching from lopinavir/ritonavir (LPV/r) soft gel capsule (SGC) to tablet formulation improves tolerability in indigent AIDS clinic. 45th Annual Meeting of the Infectious Disease Society of America. 2007. San Diego, CA.
4. Gathe JC, Lipman BA, Mayberry C, et al. Tolerability and Therapy Preference of Lopinavir/ritonavir (Kaletra) Soft-gel Capsules and Tablets as Single Agent in a Cohort of HIV Positive Adult Patients (IMANI 2). Eighth International Congress on Drug Therapy in HIV Infection. Glasgow, UK. 2006.
5. Johnson MA, Gathe JC, Podzamczer D, et al. A once-daily lopinavir/ritonavir-based regimen provided noninferior antiviral activity compared with a twice-daily regimen. Journal of Acquired Immune Deficiency Syndrome. 2006;43:153-160.
6. Mildvan D, Tierney C, Gross R, et al. Randomized comparison in treatment-naive patients of once-daily vs. twice-daily lopinavir/ritonavir-based ART and comparison of once-daily self-administered vs. directly observed therapy. 14th Conference on Retroviruses and Opportunistic Infections. 2007. Los Angeles. Poster 138.
7. Clumeck N, van Lunzen J, Chiliade P, et al. ARTEMIS: Efficacy and safety of lopinavir (BID vs. QD) and darunavir (QD) in antiretroviral-naive patients. 11th European AIDS Conference. 2007. Madrid, Spain. Abstract LBPS7/5.
|
| |
|
 |
 |
|
|