 |
 |
 |
| |
Pharmacokinetics of the next-generation NNRTI etravirine (ETR; TMC125) in HIV-infected children between 6 and 17 years, inclusive
|
| |
| |
Reported by Jules Levin
CROI 200, Boston
Thomas N Kakuda,1 Christoph Konigs,2 Cornelia Feiterna-Sperling,3 Claudio Viscoli,4 Raffaella Rosso,4 Rekha Sinha,5 Ingeborg Peeters,5 Monika Peeters,5 Katrien Janssen,5 Richard MW Hoetelmans,5 Katia Boven1
1Tibotec, Inc, Yardley, PA, USA; 2J W Goethe University, Frankfurt, Germany; 3Charite Campus Virchow-Klinikum, Berlin, Germany; 4Azienda Ospedaliera Universitaria San Martino, Genoa, Italy;
5Tibotec BVBA, Mechelen, Belgium
Abstract
Background
ETR is a next-generation NNRTI that has demonstrated significant antiviral activity in two large Phase III trials, DUET-1 and 2, conducted in HIV-infected adults with evidence of NNRTI resistance. Pediatric dosing of ETR has not yet been established. The objective of this study is to determine the weight-based dose of ETR that will achieve exposures in children comparable to those in adults.
Methods
HIV-1-infected children between 6 and < /=17 years with at least two consecutive viral loads of <50 copies/mL on a lopinavir/ritonavir (LPV/r)-containing regimen,
were enrolled. ETR 4mg/kg bid was added for 7 days followed by a morning dose and 12-hour pharmacokinetic (PK) assessment on Day 8. Both 25 and 100mg tablets were used. ETR PK was assessed using non-compartmental analysis; Cmin and AUC12h were compared to parameters previously established in adults administered 200mg bid on a LPV/r-containing regimen. Safety and tolerability were assessed throughout the study up to 30 days postdosing.
Results
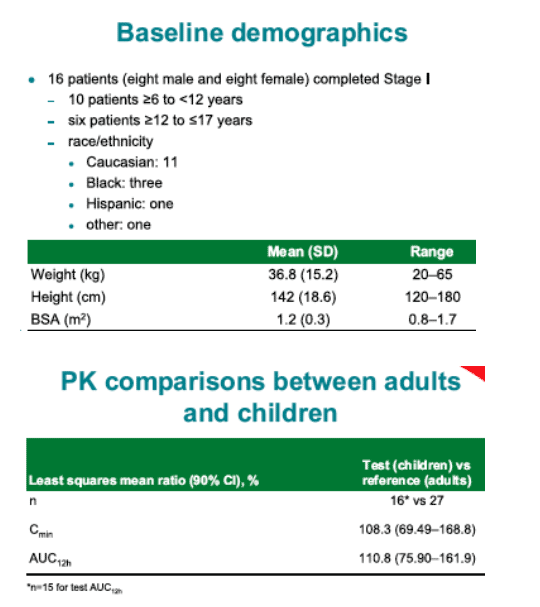
Seventeen children were enrolled in the study; PK data were available in 16: 10 between the ages of 6 and < 12 and six between the ages of >/=12 and
Author Conclusions
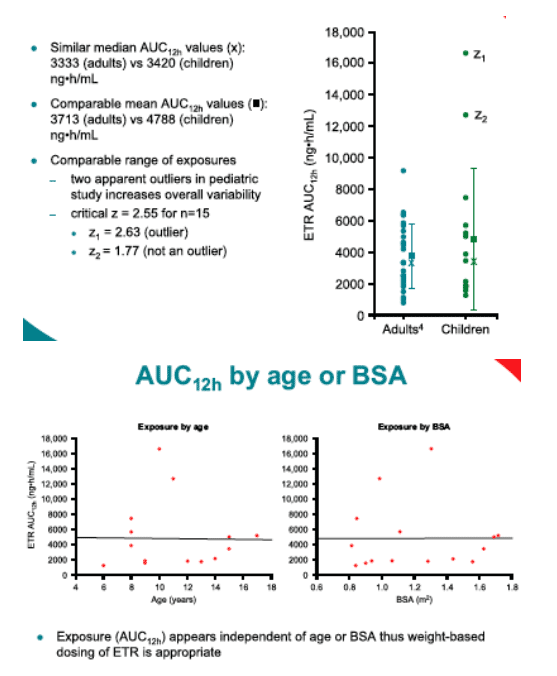
· ETR administered at 4mg/kg bid following a meal provides comparable exposure in children (age 6-17, inclusive) to 200mg bid in adults
· ETR was generally safe and well tolerated
- two patients developed a transient rash of mild-to-moderate severity
- no apparent association with ETR exposure (AUC12h)
· Given the general concern for under dosing of antiretrovirals in children5,6 and lack of safety signal during Stage I, Stage II of this trial with a 30% higher dose (5.2mg/kg bid) was started and recruitment is ongoing
· A Phase II trial to further determine pharmacokinetics, safety and efficacy in
treatment-experienced children will begin after final dose selection





|
| |
|
 |
 |
|
|