 |
 |
 |
| |
A phase I study to explore the activity and safety of SCH 532706, a small molecule chemokine receptor-5 (CCR5) antagonist in HIV-1 infected subjects
|
| |
| |
Reported by Jules Levin
CROI 2008, Boston
Pett SL1,2,McCarthy M3, MacRae K1, Cooper DA1,2, Tendlokar A3, Norris R1, Tseng J3, Williams KM4,5, Emery S2.
S2.1HIV, Immunology and Infectious Diseases Clinical Services Unit, St. Vincent's Hospital, Sydney, Australia; 2National Centre in HIV Epidemiology and Clinical Research, University of NSW, Darlinghurst, Sydney, Australia; 3Schering-Plough Research Institute, Kenilworth, NJ, USA; 4Clinical Trials Centre, St. Vincent's Hospital, Sydney, Australia; 5Clinical Pharmacology Department, St. Vincent's Hospital and University of NSW, Darlinghurst, Sydney, Australia,
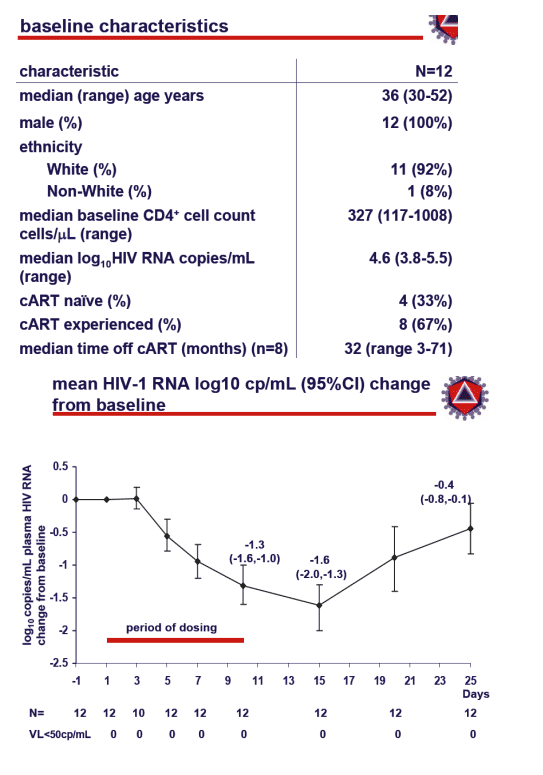
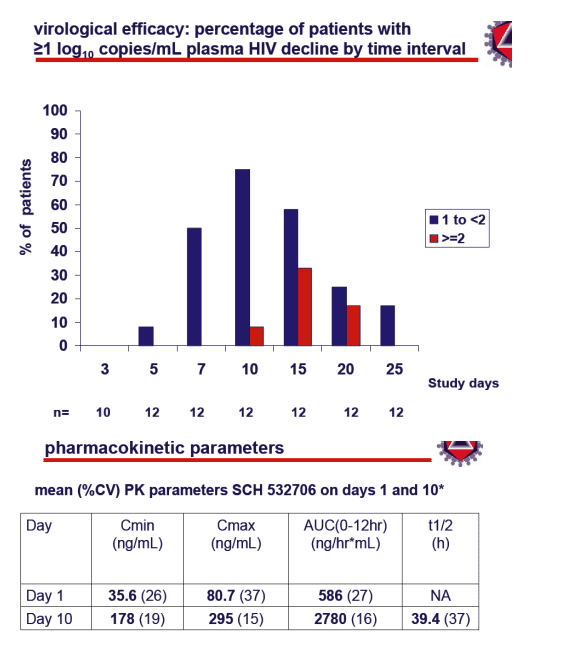
From Jules: There are several CCR5 inhibitors of note in clinical and preclinical development including this drug from Schering and ones from Pfizer and Incyte. Clearly CCR5 atagonists will play a role in ART therapy and CCR5 is not going away. Further characterization of their role is ongoing. Of particular note, and I reported on this & this report from CROI is now on the NATAP website, is that maraviroc was ot associated wih lipid elevations. After 10 days of monotherapy the mean viral load reduction was -1.3 log, after several more days after therapy stopped it was -1.6 probably due to continual binding of drug to receptors, 80% of patients had 1 log or more reduction in viral load after 10 days.
AUTHOR Conclusions
SCH 532706, in the presence of ritonavir, at a dose of 60mg bid was safe and well tolerated.
The drug was biologically active against HIV-1
SCH 532706 is suitable for once daily dosing in a ritonavir-containing regimen.
BACKGROUND
SCH 532706 is a small molecule CCR5-receptor antagonist
SCH 532706 in vitro data:
--high affinity for CCR5 receptor -Kd = 0.36 ±0.09 nM
--inhibits replication of HIV-1 primary isolates in PBMCs: IC50and IC900.2 and 1.8 nM, respectively
--long dissociation half life of 78 ±24 hours
--resistance has been difficult to generate
pre-clinical pharmacokinetic characteristics:
--good bioavailability in animal models
--moderate protein binding (80%)
--metabolism via CYP3A4; without potent inhibition/induction
--not a P-glycoprotein (P-gp) efflux substrate
--eliminated by urinary and biliary routes
--weak inhibition of hERG current (-8% in 5 _M solution)
SCH 532706 PK
in a prior rising multiple-dose study in healthy volunteers SCH 532706 was safe and well tolerated
the effects of administration with a potent CYP3A4 inhibitor were to:
--increase AUC 11 fold
--increase Cmin 18 fold
--prolong t1/2from 12 to 26 hours
--reduce PK variability from 30 to 10%
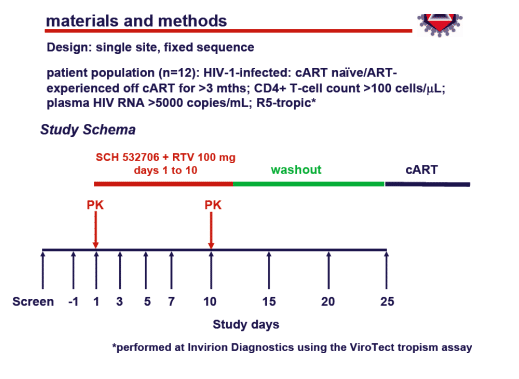
Primary study objective
to examine the antiviral activity of SCH 532706 in untreated, R5-tropic HIV-1-infected individuals about to begin or resume cART
Secondary study objective:
--to determine the PK profile of SCH 532706 in HIV-1-infected individuals;
--to explore the relationship between plasma drug exposure and virological response;
--to further document safety and tolerability of SCH 532706
Endpoints and analysis
Primary endpoint
change from baseline to Day 10 plasma HIV RNA (log10copies/mL)
Secondary endpoints
--proportion of patients with Day 10 plasma HIV RNA <50 copies/mL
--proportion of patients with ≥1 log10copies/mL decline at Day 10
--standard PK assessments after single and multiple doses
--changes in immunological parameters and tropism
--adverse events: clinical and laboratory
--ECG changes
Analysis
--ITT
--ANOVA model for the primary efficacy parameter i.e. log10change in HIV RNA from baseline to day 10
--Summary statistics for concentration data at each sampling time and the derived PK parameters
--safety parameters summarised

|
| |
|
 |
 |
|
|