 |
 |
 |
| |
Resistance After First Failure Looms as Larger Problem in Poor vs Rich Countries
|
| |
| |
15th Conference on Retroviruses and Opportunistic Infections
February 3-6, 2008
Boston
Mark Mascolini
Resistance to first-line nonnucleosides (NNRTIs) may prove more common in resource-poor countries than in wealthy nations, according to a comparison of 22 studies by top British resistance experts [1].
The researchers acknowledge that their analysis has distinct limits because it compares results from 18 randomized clinical trials in rich countries with data from four cohort studies in Malawi, Uganda, Cameroon, and Thailand. But they caution that rare access to viral load monitoring and less intense patient follow-up in low-income countries could make patients there more vulnerable to resistance.
Deenan Pillay (University College London) and coworkers analyzed results from 18 clinical trials testing a first-line NNRTI or ritonavir-boosted protease inhibitor (PI) in rich countries and four African or Thai cohort studies involving people starting the NNRTI nevirapine in their first regimen. All studies enrolled people over 13 years old, had 36 to 80 weeks of follow-up, and used genotyping to find resistance mutations after virologic failure.
Eight NNRTI trials enrolled 4212 people, 10 PI trials signed up 3063 patients, and four cohort studies tracked 2684 people. Pretreatment median CD4 counts ranged from 99 to 114 in the African and Thai cohort studies, compared with starting median CD4s of 137 to 363 in the 18 trials. Median pretreatment viral loads ranged from 4.66 to 5.21 log (about 40,000 to 170,000 copies) in the randomized trials and lay above 100,000 copies in the two cohort studies that measured pretreatment viral load.
Looking first only at the clinical trials, Pillay found no major resistance mutations at failure among 56.9% of patients starting a boosted PI versus 38% starting an NNRTI. Resistance rates at failure proved substantially higher in the four cohorts than in the trials for every resistance category assessed:
· NNRTI resistance rate: 5.93% cohorts, 2.35% trials
· PI resistance rate: 0.10% trials
· M184V/I: 5.55% cohorts, 1.61% NNRTI trials, 0.80% PI trials
· AZT/d4T mutations: 1.86% cohorts, 0.03% NNRTI trials, 0.03% PI trials
· K65R: 0.40% cohorts, 0.13% NNRTI trials, 0.00% PI trials
After up to 80 weeks of follow-up, more than 80% of genotyped African and Thai cohort members had the M184V/I mutation provoked by 3TC and about 90% of genotyped patients had an NNRTI mutation.
Possible reasons for the higher resistance rates in the cohort studies, Pillay suggested, "include time left on HAART after virological failure due to monitoring differences and the choice of nucleoside backbones." Everyone in the cohorts took 3TC plus d4T or AZT as their nucleosides. The trials used an array of nucleoside pairs including d4T/3TC, AZT/3TC, 3TC/tenofovir, 3TC/abacavir, and tenofovir/emtricitabine.
Reference
1. Ravinda K. Gupta, Andrew Hill, Will Sawyer, Deenan Pillay. Drug resistance after virological failure of first-line HAART in resource rich and poor settings--a meta-analysis. 15th Conference on Retroviruses and Opportunistic Infections. February 3-6, 2008. Boston. Abstract 891.
ABSTRACT
Drug Resistance after Virological Failure of First-line HAART in Resource-rich and Resource-poor Settings, a Meta-Analysis
Ravindra Gupta*1, A Hill2, and D Pillay1,3
1Univ Coll London, UK; 2Univ of Liverpool, UK; and 3Hlth Protection Agency, UK
Background: Drug resistance at first-line HAART failure may compromise future options, but there have been few systematic assessments of the incidence/characteristics of resistance between first line regimens. Such work is also of importance for developing optimal rollout programmes in resource poor settings.
Methods: A systematic MEDLINE search identified 18 clinical trials in resource-rich countries with 48-week data on genotypic resistance after first-line HAART failure, using either 2NRTI+ ritonavir-boosted protease inhibitor (PI/r) (n=3282) or 2 NRTI+NNRTI (n = 4426). In addition, 2 African cohorts (n = 498) and 1 Thai study evaluated resistance after failure of first-line HAART with 2 NRTI+NNRTI (there were no eligible clinical trials in resource poor settings). The number with M184V, at least 1 thymidine analogue mutation (TAM), major NNRTI or International AIDS Society (IAS) -USA PI mutation per trial at week 48, was calculated as a percentage of successfully genotyped patients (genotype analysis [GA]) or treated patients (intent to treat [ITT]). The meta-analysis used inverse-variance weighting to create robust estimates of overall prevalence per group, with exact 95% confidence intervals.
Results: Virologic failure (ITT, viral load >400) at 48 weeks occurred in 6.7% (2 NRTI/PI/r), 7.0% (2 NRTI/NNRTI), and 15.7% (2 NRTI/NNRTI, the African cohorts), and 87%, 80%, and 85% of failures were successfully genotyped. Resistance at failure was as follows:
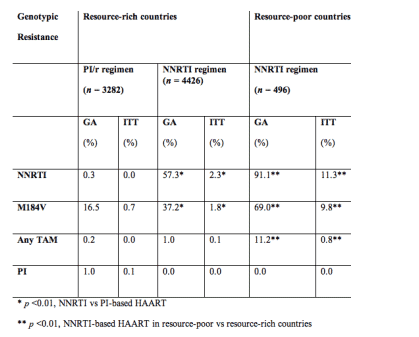
Conclusions: Given the caveats of meta-analysis, the prevalence of M184V at virologic failure is higher in patients starting first-line NNRTI-based HAART vs PI/r-based HAART. Genotypic resistance to lamivudine, NRTI (TAM), and NNRTI appears higher in resource-poor cohorts of first-line NNRTI-based HAART than NNRTI-based HAART in resource-rich countries. Possible reasons for this difference include time left on HAART after virologic failure due to different intensities of monitoring and the choice of nucleoside backbone. The implications of these findings must be placed in the context of availability/composition of second-line regimens, as well as the effect of resistance on clinical disease progression.
|
| |
|
 |
 |
|
|