 |
 |
 |
| |
Co-Receptor Tropism, ENV Genotype, and in vitro Susceptibility to CCR5 Antagonists During Monotherapy Study with INCB9471
|
| |
| |
Reported by Jules Levin
15th CROI, Feb 3-6, 2008, Boston
S. Erickson-Viitanen1, K. Abremski1, K. Solomon1, R. Levy1, E. Lam2, J. Whitcomb2, R. Lloyd, Jr.3, R. Mathis3, J. Reeves2 and D. Burns3
1Incyte Corporation, Wilmington, Delaware, USA; 2Monogram BioSciences, Inc., South San Francisco, CA, USA and 3Research Think Tank, Inc., Buford, GA, USA
Three new CCR5 antagonists were the subject of presentations at CROI 2008 from Pfizer, Schering, and this one from Incyte confirming that CCR5 is not going away. Research will I think over the next year provide an understanding on how and when CCR5 antagonists can be used for patients. The Maraviroc lipid profile in naives was reported at CROi and was very good, so maraviroc and this class of drugs appear to provide appealing possibilities.
Incyte told me at CROI they are planning a next study to begin soon.
AUTHOR CONCLUSIONS
_ INCB 9471, when given at 200 mg or 300 mg QD showed a significant antiviral response in a 14-day monotherapy study.
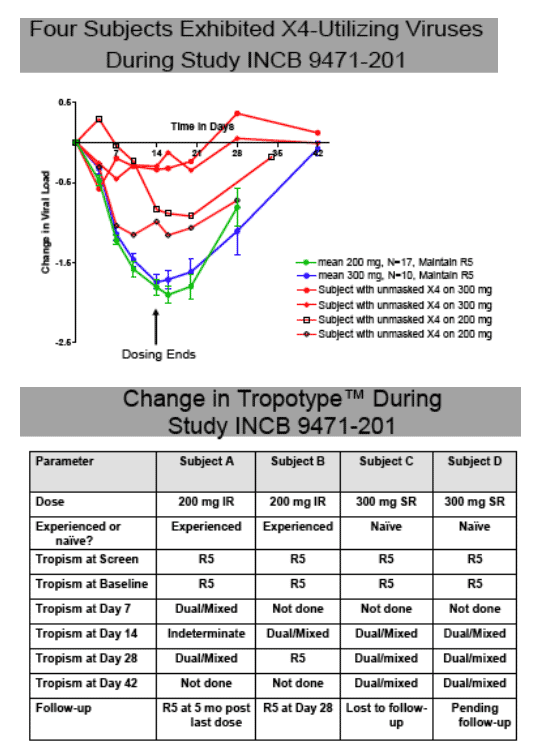
_ A minority (4 of 40) subjects showed an apparent change in tropotype during the study.
_ Clonal analysis of 2 of these subjects suggests that emerging dual/mixed virus reflected pre-existing X4-utilizing virus that was present at a low level.
_ Clones with reduced maximal suppression by CCR5 antagonists carried V3 loop substitutions previously described.
_ Bulk sequencing of the V3-gp41 region confirmed the association of basic residues at V3 loop position 11 and 25 in samples showing CXCR4-utilizing virus, and suggests that a neutral Gln or Asn at position 25 may also confer some CXCR4-utilizing effect.
Background: Study INCB 9471-201 was a 14-day monotherapy study of the once daily CCR5 antagonist INCB9471 in naive or therapyexperienced subjects who were R5-tropic by Trofile™ assay at Screen. Forty-nine subjects enrolled and received placebo (N=9), 100 mg (N=9), 200 mg (N=19) or 300 mg (N=12) of INCB9471.
Methods: Trofile assay, susceptibility (PhenoSense Entry Assay) and ENV genotype (HIV-1 GeneTanker C2V3-gp41HR1 domain Select assay)
were determined at Screening, Baseline, Day 14 and Day 28; clonal sequencing was determined by GeneSeq HIV ENV assay.
Results:
Virus present at Screening and Day 28 was highly susceptible to inhibition by INCB9471 (Screening IC50 = 12.1 ± 1.5 nM, N=49, Day 28 IC50 = 8.0 ± 1.5 nM, N=32), nearly all viral samples displayed ≥90% maximal inhibition by INCB 9471 or maraviroc.
Plasma derived viral RNA genotypes were obtained and compared to V3 polarity 11/25 basic prediction rules; modified prediction analysis, MuTanker™ was
also performed.
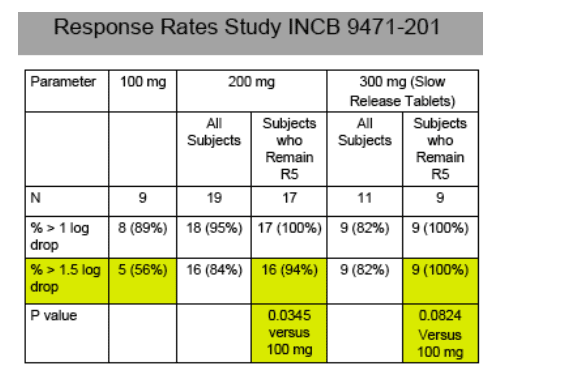
Thirty of 39 subjects receiving 14 days of INCB9471 treatment achieved viral load declines > 1.5 log10 relative to baseline: representing 5 of 9, 16 of 19, and 9 of 11 subjects receiving 100 mg, 200 mg or 300 mg INCB9471, respectively.
Circulating virus remained R5 tropic in 35 of 39 INCB9471-treated subjects. Dual/mixed tropic virus was observed on Day 14 and/or Day 28 for 2 subjects each, who received 200 mg or 300 mg INCB9471. These subjects exhibited attenuated viral load responses; 3 of 4 had prior therapy experience.
Clonal analysis (Trofile, sequencing, susceptibility) has been completed on 2 of these subjects. In subject A, receiving 200 mg, dual tropic phenotype was observed in ~13% of clones examined at Baseline; corresponding V3 loop sequences revealed lysine at position 25 relative to other viruses in the swarm with glutamic acid. Circulating virus was again R5 tropic subsequent to study completion. In subject B, receiving 200 mg, circulating virus was dual/mixed at Day 14, returning to R5 tropic at Day 28. At Day 14, the CXCR4-using viruses formed a monophyletic group, and were distinct from R5 viruses, suggesting that
the dual virus present at Day 14 likely arose via outgrowth from an unidentified ancestral virus present at Baseline.
Conclusions: As shown with other CCR5 antagonists, the data from a 14-day
monotherapy study with once-daily INCB9471 suggests that emergence of CXCR4-using viral variants in a minority of the treated patients reflects outgrowth from pretreatment viral reservoirs.
BACKGROUND
· INCB9471 is a selective CCR5 Antagonist
- Long half life of ~60 hours allows once daily dosing
- Protein binding adjusted 90% inhibitory concentration of 50-60 nM
· Study INCB 9471-201 was a 14 day monotherapy study in treatment naive and
treatment experienced subjects
- 3 doses examined:
· 100 mg of an immediate release (IR) formulation tablet
· 200 mg of an immediate release (IR) formulation tablet
· 300 mg of a prototype slow release (SR) formulation tablet
- Viral load was determined at every visit
- Co-receptor tropism was assessed at Screening, Baseline, Day 14 and Day 28 on samples with viral load of at least 1000 copies/mL
- In vitro susceptibility to INCB 9471 and maraviroc was determined at Baseline and Follow-up (Day 28) visits using the PhenoSense Entry Assay of Monogram
BioSciences
- ENV sequence (including the C2-V3-gp41 region) was determined at Baseline and Follow-up (Day 28) visits using the RTT C2V3/gp41 Assay of Research Think Tank, Inc.
- CCR5 genotype was determined at Baseline using the Gen-Hum-CCR5 GeneTanker Assay of Research Think Tank, Inc.
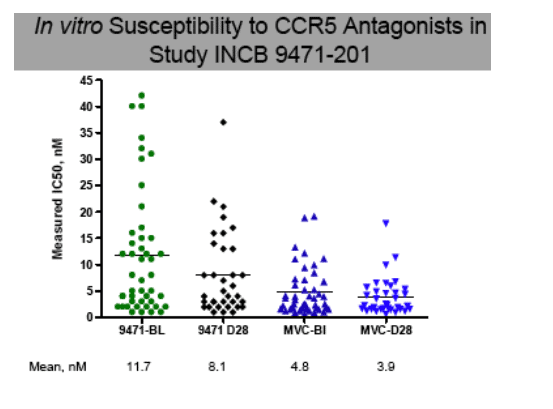
As anticipated for subjects previously naive to CCR5 antagonists, and harboring CCR5 virus exclusively (within the sensitivity constraints of the Trofile™ assay to detect minor populations), recombinant virus prepared from the plasma virus from the Baseline samples from Study INCB 9471-201 was very sensitive to inhibition by INCB 9471 or maraviroc. For all subjects combined, the mean ± SEM for the IC50 was 11.7 ± 1.8 nM, N=46 for INCB 9471, and 4.8 ± 0.7 nM for maraviroc. This difference in in vitro potency is similar to that measured for peripheral blood mononuclear cells, where INCB 9471 has a 90% inhibitory
concentration value of 10.2 nM and maraviroc has a 90% inhibitory concentration value of 6.4 nM. At least 90% inhibition of virus replication was demonstrated with INCB 9471 and maraviroc for all Screening and Baseline samples. The recombinant viruses that were least sensitive to inhibition by INCB 9471 (ie, had IC50 values of 30 nM or higher, see Figure 6) were also least sensitive to maraviroc, although the IC50 values of these recombinant viruses were less than 4-fold different from the geometric mean. The viral load in two of these three corresponding subjects achieved declines relative to baseline of 2.0 or
more logs, the third subject received placebo. In general, there was little change
in the sensitivity of patient virus over the course of the study; Day 28 IC50 values
were slightly lower for both maraviroc and INCB 9471. At least 90% maximal
suppression by maraviroc or INCB 9471 was shown by all by one Day 28 virus
sample (had 85% maximal inhibition); although clonal analysis has identified
viruses with attenuated sensitivity (see Subject B data to the right).
Monogram Biosciences, Inc. conducted a clonal tropism and sequencing analysis for Subjects A and B. Circulating virus was tested from the plasma of patients using the first generation Trofile™ assay for co-receptor tropism. The entire HIV envelope (ENV) coding sequence was amplified from plasma samples by RT/PCR and was ligated into a pCXAS expression vector to create an envelope expression vector library. Virus particles carrying ENV glycoproteins derived from the plasma virus were produced by transfecting HEK293 cells with the purified ENV expression vector library and an HIV-1 genomic vector lacking the ENV region and containing a firefly luciferase gene. The ability of the resultant pseudoviruses to infect U87 cells expressing CD4 receptor and either the CCR5
or CXCR4 co-receptor was assessed by measuring luciferase relative light units (RLU). Tropism was confirmed by the inhibition of viral replication in each cell type using a specific co-receptor inhibitor. For the clonal analysis, 48 individual ENV clones of plasma virus from each of three time points for Subject A and Subject B were prescreened for viability and tropism in single-well cultures of CCR5 or CXCR4- expressing cells. This preliminary analysis is summarized as follows:
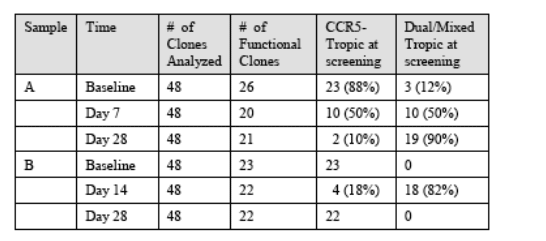
As shown in the table, dual/mixed tropic virus was present at a low level even in the Baseline sample from subject A; Only CCR5-tropic virus was detected in Baseline clones examined from the plasma virus population of Subject B. Twelve (12) viable clones were selected for further examination to confirm the tropotype assignment, to determine the phenotypic susceptibility of resultant virus to inhibition by INCB009471 and by maraviroc, and to sequence the entire ENV gene for phylogenetic analysis.
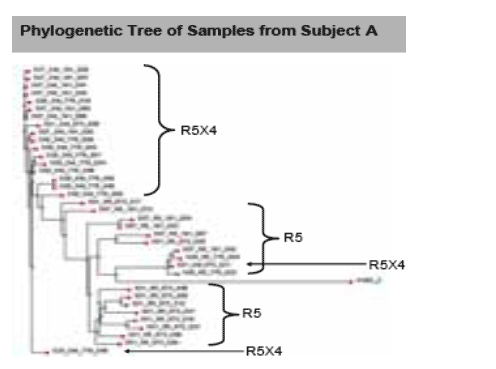
The tree for Subject A shows a separation in the clustering of R5 and DM viruses. The R5 viruses for this subject show two sub-branches as well. A bootstrap analysis using the paupsearch program for these sequences indicates that the R5 branch is present in 79% of the 1000 bootstrap trials. Of 12 clones analyzed corresponding to Baseline (Day 1), two clones had the dual/mixed tropotype™. One clone (021) clustered with R5 viruses, clone 036 was clustered in the middle of the group of dual/mixed encoding ENV sequences present at Day 7 and at Day 28. Therefore, based on sequence alignments, and tropism testing, it can be concluded that the dual/mixed virus present in Subject A at Day 7 and Day 28 arose from virus typified by the Day 1 clone 036.
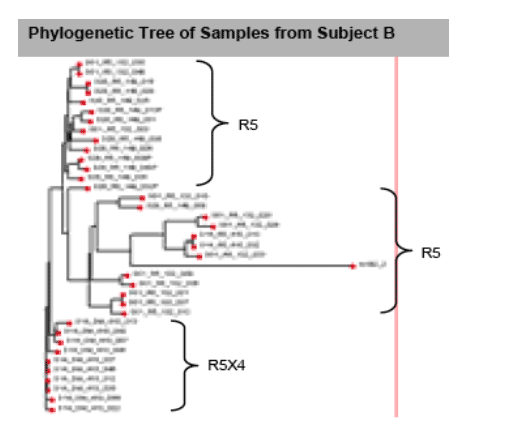
The tree for Subject B shows that all of the R5 sequences cluster together whether they are derived from Day 1, Day 14 or Day 28, and separate into two subbranches. The DM sequences from Day 14 also cluster together as a single branch. A bootstrap analysis using the paupsearch program indicates that this DM branch is present in 82% of the 1000 bootstrap trials generated for these sequences (data not shown). This indicates that the ENV sequences
corresponding to the dual/mixed tropotype™ are clustered together in a group that is phylogenetically distinct from R5-tropic viruses. Day 1 R5 viral sequences comprise a diverse group, with only 6 of 12 sequences showing the closest match to the Day 1 R5 consensus sequence. However, all closest matches are to R5 sequences, either from Day 14 or Day 28. Three of 12 Day 1 clones show some similarities to the Day 14 dual/mixed sequences, though the latter are not the closest match to the Day 1 sequences. Based on the limited sequences conducted, the data are not inconsistent with the dual/mixed tropotype™ having arisen from an unidentified ancestor virus population, and not from any identified R5 tropic virus. This conclusion is similar to that advanced by Westby et al., in describing the clonal analysis of viruses from the maraviroc monotherapy trial, in which a remarkably similar phylogenetic distribution of R5 versus dual/mixed viruses was observed to the data shown for Subject B with INCB 9471.

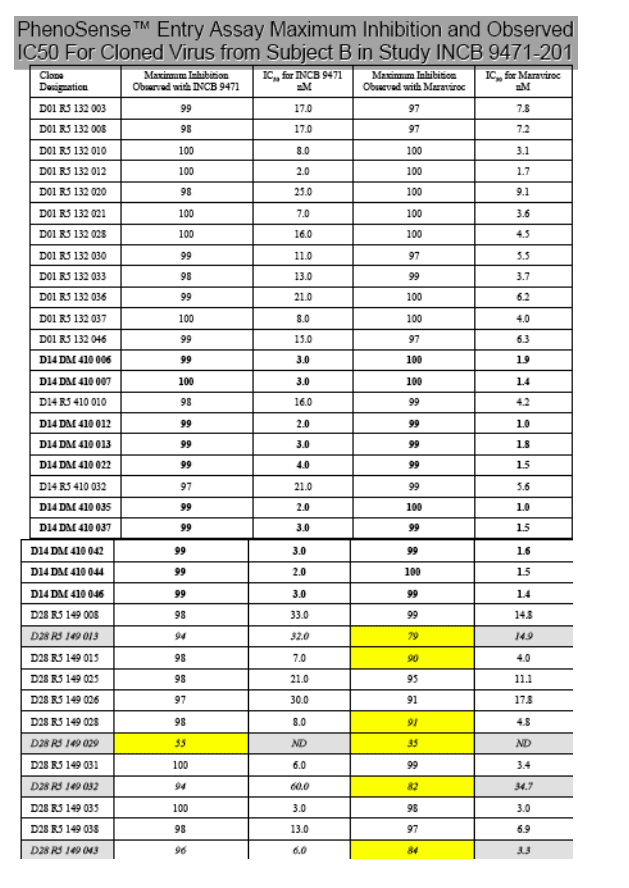
When individual cloned viruses from Subject A and B were assessed using PhenoSense Entry assay, several Day 28 R5 clones in Subject B exhibited partial inhibition by maraviroc. One of 6 clones with highly attenuated sensitivity to maraviroc also showed a highly attenuated maximal response to INCB009471. In the table, dual/mixed clones are bolded; clones with sub-maximal response are shaded and italicized. All of the clones with attenuated sensitivity to maraviroc showed a substitution of V (valine) for I (isoleucine) at position 26, a mutation previously described by Westby et al., 2006 (J. Virol 81:2359) as associated with reductions in susceptibility to maraviroc. Clone 029 from subject B shows only 35% inhibition with the maximal concentration tested of maraviroc, and only 55% maximal inhibition with INCB 9471. Analysis of the specific sequence of this clone revealed an additional substitution of T (threonine) for A (alanine) at position 19 of the V3 loop, a mutation also previously described by Westby et al. The overall in vitro susceptibility of the Day 28 mixture showed 99% maximal inhibition. The impact of specific clonal populations that show attenuated susceptibility to INCB 9471 on the overall antiviral response of the viral swarm in a subject on treatment awaits longer term virological follow-up.

GeneTanker plasma sequencing of the V3- Loop and MuTanker™translation analysis of Selected Subjects in Study INCB 9471-201.
V3 loop codons 11 and 25 are important in predicting R5 and CXCR4-using virus. R5-using virus has a preference for neutral amino acids Ser (S) or Gly (G) at position 11 (S- 11 and G- 11) and CXCR4-using virus preference is for the basic amino acid Arg (R). Codon selection for Lys (K) or Arg (R) at position 25 (K- 25 or R- 25) are also associated with CXCR4-using virus while acidic amino acids Glu (E) and Asp (D) predict R5-using virus (E- 25 or D- 25). At position 25, a mixture nucleotide codon (RMA) for Subject "A" at baseline predicted the amino acids (AAA) or Lys (K), (ACA) Thr (T), (GAA) Glu (E) and (GCA) Ala (A). Day
14 plasma genotype for subject "A" demonstrated a selective preference for K- 25 virus and a dual/mixed tropotype. In addition, mixture codons at position 25 predicting CXCR4-using virus were detected in Day 14 samples for subjects "B and C". The screen/ baseline results for both of these subjects indicates R5 virus. By Day 28 R5 virus population were again predominant in Subject "B". Subject "D" had R5 virus at screen and the Day 14 results demonstrated a codon selection preference for R-11 and a dual/mixed tropotype. At position 25, the neutral Asn (N) was observed at Day 14. Interestingly, Subject "E" and "H" also had a neutral Gln (Q) amino acid at codon 25 (Q- 25), but had the typical R5
determining amino acid residue Ser (s) at position 11. Subject "E" was the only subject that yielded the R5 tropotype but did not achieve a 1.5 log reduction in viral load from baseline in the 200 mg QD dose group.
|
| |
|
 |
 |
|
|