 |
 |
 |
| |
Multicenter, Retrospective Study of Incidence and Predictors of Adverse Events Associated With Nevirapine Use in a Cohort of Pregnant and Nonpregnant Women Treated at 3 Urban HIV Clinics in the United States
|
| |
| |
Reported by Jules Levin
15th CROI, Feb 3-6, 2008, Boston
E. Aaron,1 M. C. Kempf,2 S. Criniti,1 E. Tedaldi,3 A. Warriner,4 R. Kumar,5 E. Gracely,6 L. H. Bachmann2,4,7
1Division of Infectious Diseases and HIVMedicine, Department ofMedicine, Drexel University College ofMedicine, Philadelphia, PA; 2Department of Epidemiology, University of Alabama at Birmingham, Birmingham, AL;
3Temple University School ofMedicine, Philadelphia, PA; 4Department ofMedicine, University of Alabama at Birmingham, Birmingham, AL; 5Department of InternalMedicine,Mercer University,Macon, GA;
6Department of Family, Community, and PreventiveMedicine, Drexel University College ofMedicine, Philadelphia, PA; 7Birmingham Veterans AdministrationMedical Center, Birmingham, AL
BACKGROUND
· Despite significant advances in HIV therapy, toxicities associated with antiretroviral therapy (ART) have become a limiting factor in the treatment of persons living with HIV.
· Adverse events characterized by liver and skin toxicities that usually occur within the first 18 weeks of treatment have been associated with the use of nevirapine (NVP).
· Some investigators have reported these toxicities more frequently in women with higher baseline CD4 counts (>250 cells/mm3),1 in those who are pregnant,2 and in the presence of pre-existing liver disease (ie, hepatitis C virus [HCV]).3,4
· However, there are conflicting data regarding pregnancy and risk of liver or skin toxicity. Although deaths due to hepatic failure have been reported in HIV-infected pregnant women receiving NVP as part of an ART regimen, it has not been well studied whether pregnancy increases the risk of liver enzyme elevations (LEE) or rash in women receiving NVP or other ART.5
OBJECTIVE
· To assess the prevalence and predictors of developing LEE and rash associated with NVP versus non-NVP use in pregnant and nonpregnant women.
AUTHOR CONCLUSIONS
· Women on NVP-based regimens did not have an increased risk for developing grade ≥2 LEE compared with women on non-NVP-based regimens.
· Women on NVP with nadir or baseline CD4 >250 cells/mm3 had an increased risk of LEE compared with women on non-NVP regimens with CD4 >250 cells/mm3.
· Women on NVP with a nadir or baseline CD4 >250 cells/mm3 had a significantly increased rate of rash compared with women on non-NVP regimens with CD4 >250 cells/mm3 in both pregnant and nonpregnant women.
· NVP was predictive of rash even when controlling for CD4 count and pregnancy.
· Pregnancy is not an independent risk factor for developing LEE or rash.
· HCV coinfection was a significant predictor of developing grade ≥2 LEE independent of ART regimen.
· The majority of liver and skin adverse events in both regimen groups occurred within 6 weeks of initiating ART.
AUTHOR DISCUSSION
· This large cohort study provides additional information for clinicians in assessing the risk of NVP-induced liver and skin toxicity.
· The clinical implications of our analysis are that safety will be improved by following current treatment guidelines regarding CD4 counts at the time that NVP is initiated.
· Nadir CD4 count may be as important as baseline CD4 counts in the decision making process; however, this has not been validated in a prospective study.
· In our analysis, pregnancy was not found to be an independent risk factor for developing hepatic or skin toxicity.
· HCV-coinfected patients should undergo close monitoring for liver enzymes.6
· Empiric use of NVP in the absence of resources for close monitoring of CD4 count and liver enzymes calls for careful consideration of the risks and benefits of NVP therapy in such settings.
METHODS
· Three university-based clinics with large prenatal patient populations: Drexel University College of Medicine, Philadelphia, PA; Temple University, Philadelphia, PA; and University of Alabama at Birmingham, Birmingham, AL.
· Women aged 18-55 who initiated an ART regimen and had at least 1 visit in 12 weeks between January 1999 and August 2005.
· Scoring criteria from the US National Institute of Allergy and Infectious Disease, Division of Acquired Immunodeficiency Syndrome (DAIDS) were used. LEE: DAIDS grade ≥2 that was worse than baseline, occurring within 18 weeks of ART initiation.
· Rash: DAIDS grade ≥2 skin reaction, occurring within 18 weeks of ART initiation in patients without baseline rash.
· Patients on NVP versus non-NVP regimens were compared using chi-square test, unpaired t test, orMann-Whitney U test. Logistic regression was used to control for confounding.
· Analyses were stratified by baseline and nadir CD4 cell count (≦250 cells/mm3 vs >250 cells/mm3) and pregnancy status.
RESULTS
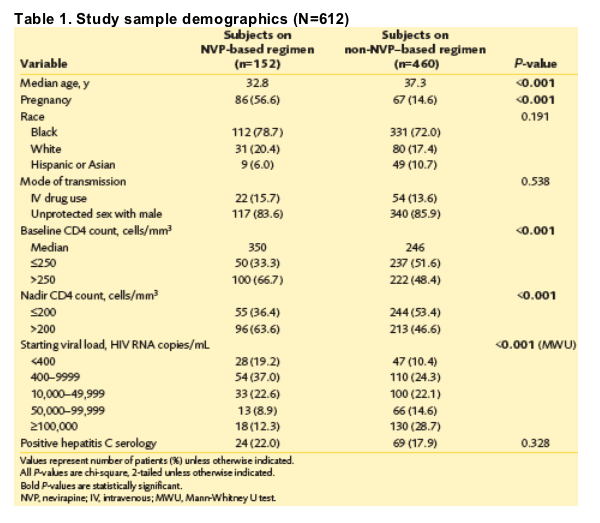
· Of the 651 patient charts reviewed, 39 were patients excluded after becoming pregnant following the start of their ART, leaving 612 subjects for analysis. Of those 612 subjects, 152 (24.8%) initiated NVP-containing ART regimens, with 86 pregnant (56.6%), and 460 initiated non-NVP-containing regimens, with 67 pregnant (14.6%) (Table 1).
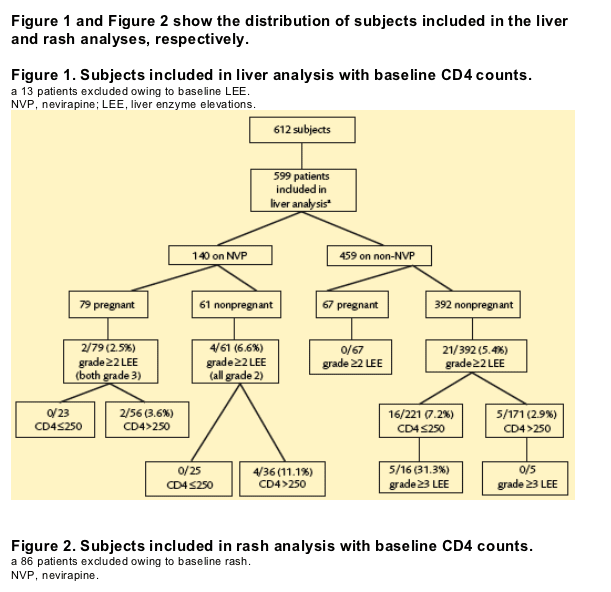
Liver enzyme elevations
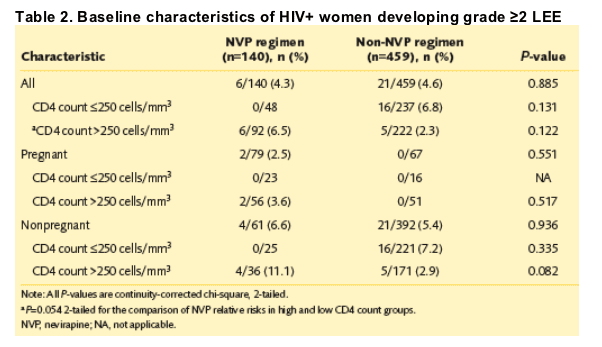
· There were no significant differences between the NVP and non-NVP groups for the rate of developing grade ≥2 LEE (P=0.885) or grade 3 LEE (P=0.668). Two patients on NVP developed grade 3 LEE; both were pregnant and had CD4 >250 cells/mm3. All grade ≥3 LEE (n=5) occurred in nonpregnant women on a non-NVP regimen with CD4 ≦250 cells/mm3.
· For nadir CD4 >250 cells/mm3, there was a trend towards a higher rate of LEE in NVP compared with non-NVP patients (P=0.054). In the group with nadir CD4 ≦250 cells/mm3, the pattern was reversed; non-NVP patients had a higher rate of LEE than those on NVP (P=0.21) (Table 2).
· The majority of liver and skin adverse events (66.2%) occurred within 6 weeks of initiating ART.
· Among all 33 subjects who had any LEE grade ≥2 that was worse than baseline, 46% had symptoms (ie, nausea, vomiting, jaundice, and abdominal pain): 5/8 (62.5%) on NVP and 10/25 (40%) on non-NVP regimens (P=0.48).When stratified by CD4 counts, there were no LEE in the NVP group with CD4 ≦250 cells/mm3 and in the non-NVP group with CD4 ≦250 cells/mm3, and there were 18 with LEE of whom 9 (50%) had symptoms. In the group with CD4 >250 cells/mm3 and grade ≥2 LEE, 5/8 (62.5%) NVP patients and 1/7 (14%) non-NVP patients had symptoms (P=0.17 by continuity-corrected chi-square).
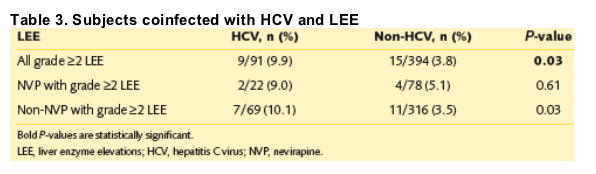
· Multivariate logistic regression showed that women were more likely to develop a grade ≥2 LEE if coinfected with HCV (odds ratio [OR], 2.65; 95% CI, 1.09-6.40). NVP, pregnancy, and CD4 count did not predict in the logistic regression. HCV-coinfected subjects on NVP-containing regimens were no more likely than subjects on non-NVP regimens to experience LEE (9.0% vs 10.1%, respectively; P=1.0). However, those patients on NVP were more likely to be tested for HCV (Table 3).
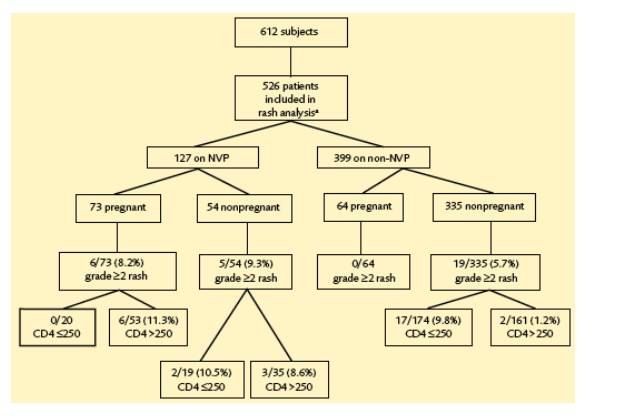
Rash
· Moderate to severe rashes (grade ≥2) were diagnosed in 30 (5.7%) women.
· There was a trend toward higher frequency among women who initiated NVP-based regimens: 11/127 (8.7%) versus 19/399 (4.8%) (P=0.099).
· Among women with baseline CD4 >250 cells/mm3, NVP was significantly associated with grade ≥2 rash (P=0.001) (Table 4). This pattern was observed in both pregnant and nonpregnant women with CD4 >250 (P=0.045 and P=0.057, respectively). In women with baseline CD4 ≦250 cells/mm3, fewer rashes occurred in the NVP group, although this difference was not significant (P=0.633).
· NVP treatment was associated with grade ≥2 rashes among pregnant women (P=0.054) but not among nonpregnant women (P=0.476). All grade ≥2 rashes among pregnant women occurred among women with baseline CD4 >250 cells/mm3 who were on a NVP-based treatment. In nonpregnant women with baseline CD4 ≦250 cells/mm3, the rate of grade ≥2 rashes was similar for both regimen groups (10.5% vs 9.8%) (Table 4).
· When stratified by CD4 count, there was a significant increase in skin toxicities in NVP-based regimens compared with non-NVP regimens with nadir CD4 >250 cells/mm3 (P< 0.001). The same significance was seen for pregnant and nonpregnant women, P=0.048 and P=0.003, respectively. NVP use did not predict skin toxicities in the low nadir CD4 group, either in pregnant or nonpregnant women.
· Multivariate logistic regression controlling for CD4 count and pregnancy showed that women were more likely to develop a grade ≥2 rash if they initiated a NVP-based regimen (OR, 2.78; 95% CI, 1.14-6.76).

|
| |
|
 |
 |
|
|