 |
 |
 |
| |
Steady State Pharmacokinetics and Safety of Atazanavir (ATV) After Treatment with ATV 300 mg/Ritonavir 100 mg (ATV/r) QD + ZDV/3TC
During the 3rd Trimester of Pregnancy in HIV+ Women
|
| |
| |
Reported by Jules Levin
15th CROI, Feb 3-6, 2008, Boston
Timothy Eley1, Eric Vandeloise2, Michael Child1, Francesca Conradie3, Carmen Zorrilla4, Deirdre Josipovic5, Yonghua Wang1, Shanmei Liao1, Rong Yang6, Heather Sevinsky1,Marina Mathew6, Stefanie Marais6, Donnie McGrath6, Richard Bertz1, and The Atazanavir 182 Pregnancy Study Group
Bristol-Myers Squibb Research and Development, Princeton, NJ, USA1; Bristol-Myers Squibb Research and Development, Braine-l'Alleud, Belgium2; HIV Clinical Trial Unit, Westdene, RSA3; UPR School of Medicine, San Juan, PR4;
Perinatal Research Unit, University of the Witwatersrand, Soweto, RSA5; and Bristol-Myers Squibb Research and Development, Wallingford, CT, USA6
AUTHOR CONCLUSIONS
Relative to data in non-pregnant historical controls, the AUC and Cmin of ATV after treatment with ATV/r 300/100 mg QD in 3rd trimester were reduced by 40% and 21%, respectively.
Postpartum PK data suggest that the PK of ATV up to 4 weeks postpartum remains different from the non-pregnant state. Subsequent post-partum PK will be evaluated at ~8 weeks after delivery.
Based on the results of the interim analysis of ATV/r 300/100, the predefined Cmin criterion was met but the AUC criterion was not. Increasing the ATV dose to an ATV/r 400/100 mg QD regimen is indicated. A cohort of 20 is planned to evaluate this dosing regimen.
The study drugs were well tolerated by the subjects with no discontinuations due to drug-related AEs. There were no unexpected safety events that were related to ATV/r.
There was no evidence of pathologic neonatal jaundice and no infant required medical intervention for jaundice. This is consistent with the findings of previous studies of ATV in pregnancy by Ripamonti, et al,[3] and Natha, et al,[4] in cohorts of 17 and 26 infants, respectively.
All 10 infants had negative HIV DNA PCR testing at birth. All follow-up testing was also negative.
Background
Plasma concentrations of some protease inhibitors are reduced in the 3rd trimester (3TRI) of pregnancy relative to non-pregnant controls, presumably due to metabolic and physiologic changes associated with pregnancy. There are limited data on the pharmacokinetics (PK) and safety of ATV in pregnancy.
Methods
BMS AI424182 is an ongoing, prospective study in HIV+ pregnant women to determine dosing of ATV/r that achieves predefined AUC and Cmin targets in the 3TRI. A planned interim analysis was conducted when 12 subjects completed the 3TRI (weeks 28-36) intensive PK. Subjects ≥ 12 weeks gestation received ATV/r 300/100 mg once daily (QD) and ZDV/3TC 300/150 mg twice daily for 2 weeks prior to intensive PK sampling in the 3TRI and at 4 weeks post-partum (PP). Protocol-defined exposures not requiring a dose adjustment were geometric mean AUC_ ≥ 30 _g-h/mL and 10 of 12 Cmin ≥ 150 ng/mL. Safety assessments on mothers and infants included clinical examination and laboratory testing at each study visit. ATV and ritonavir were analyzed by LC/MS/MS. Descriptive statistics for 3TRI and PP PK parameters were calculated and compared with historical data in HIV+ subjects as a reference; all available safety data are reported.
Results
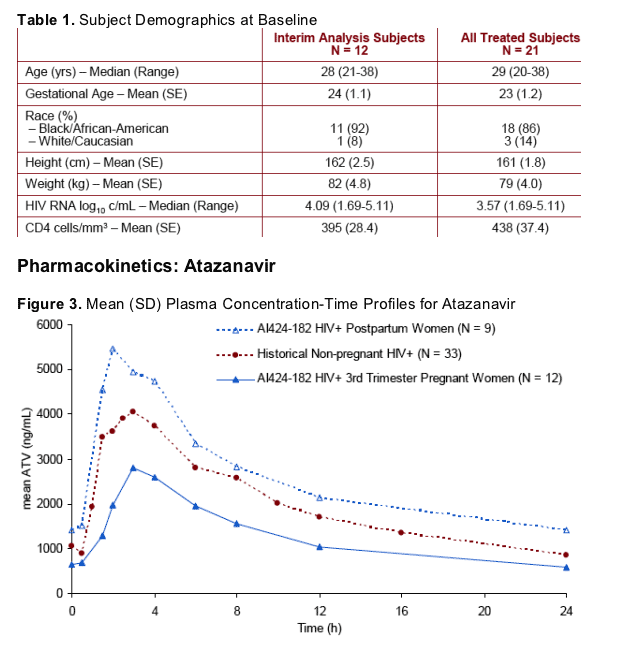
ATV geometric mean [GM (%CV)] AUC was 26.6 (43) _g-h/mL for the 3TRI intensive PK (n=12); all ATV Cmin were ≥150ng/mL; Cmin GM (%CV) was 514 (48) ng/mL. 3TRI ATV AUC and Cmin were approximately 40 and 21% lower, respectively, than historical data in HIV+ subjects [44 (51) _g-h/mL and 654 (76) ng/mL, respectively (n=33)].
PP (n=9) GM (%CV) AUC [57 (41) _g-h/mL] and Cmin [1192 (68) ng/mL] appear higher than historical data.
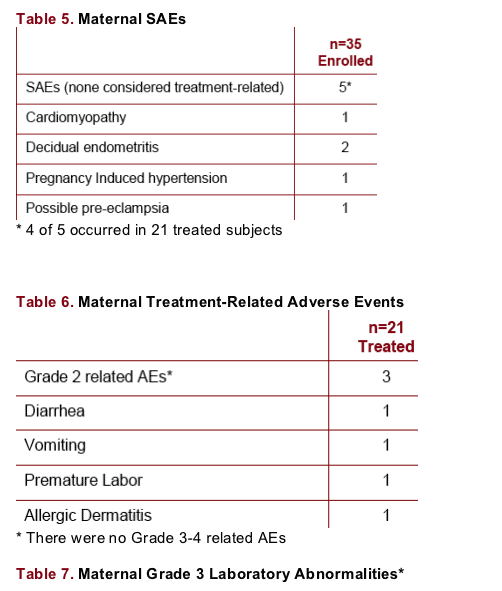
SAEs unrelated to study drug occurred in 4/21 treated subjects. Grade 3 bilirubin occurred in 5/18 subjects with lab results. All 10 subjects reaching delivery achieved HIV RNA < 50 copies/mL; all infants tested HIV negative. All infant bilirubins were within normal limits through day 14; there was 1 Grade 3 (4.5 mg/dL) at day 15.
Conclusions
ATV/r 300/100 QD did not meet the protocol-defined AUC criterion. Although the Cmin criterion was met, these exposures may not be optimal for ARV experienced patients. A protocol-specified dose increase to ATV/r 400/100 QD in the 3TRI will be evaluated in a subsequent cohort. ATV exposures appear higher than historical data at 4 weeks PP in HIV+ women likely due to ongoing physiologic changes. Study drugs were well tolerated. No unexpected safety events related to ATV/r occurred.
BACKGROUND
Atazanavir (ATV), a potent, once-daily (QD) HIV-1 protease inhibitor (PI), has demonstrated clinical efficacy comparable to standard of care in antiretroviral naive patients, and when boosted with ritonavir (r), to twice-daily lopinavir/r (LPV/r) in treatment-experienced patients.
Plasma concentrations of some PIs are reduced in the 3rd trimester of pregnancy relative to non-pregnant controls, presumably due to metabolic and physiologic changes associated with pregnancy.[1]
Due to reduced concentrations observed in the third trimester with LPV/r at 400/100 mg twice daily, a dose increase of LPV/r to 533/133 mg twice daily in the third trimester produced exposures similar to non-pregnant historical controls.[2]
There are limited data on the pharmacokinetics (PK) and safety of ATV in pregnancy. Ripamonti, et al, reported ATV plasma concentrations in 17 women in the third trimester that were similar to post-partum.[3]
BMS AI424182 is an ongoing, prospective study in HIV+ pregnant women to determine dosing of ATV/r that achieves predefined AUC and Cmin targets in the third trimester.
The results presented here are from a planned interim analysis that was conducted when 12 subjects completed the third trimester (weeks 28-36) intensive PK. At the time of the interim analysis, post-partum data from 9 mothers and 10 infants were also available.
OBJECTIVES
Primary
To determine which dosing regimen of ATV/r produces adequate drug exposure during pregnancy compared to drug exposure in historical data in HIV-infected subjects
Secondary
To assess the safety in the neonate of maternally administered ATV/r
To assess the safety and tolerability of an ATV/r-based HAART regimen during pregnancy.
To compare ATV/r drug exposure during the 3rd trimester of pregnancy to drug exposure in the same women in the study cohort during the postpartum period
METHODS
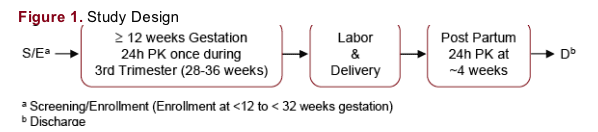
Study Design
Multi-center, open-label, prospective, single-arm study (Figure 1)
Subjects received ATV/r 300/100 mg once daily (QD) and ZDV/3TC 300/150 mg twice daily for at least 2 weeks prior to intensive PK sampling in the third trimester and 4 weeks post-partum
Key Inclusion Criteria
HIV-1 infected, pregnant women with a CD4 count of ≥ 200 cells/mm3 at ≥ 12 weeks to < 32 weeks gestation
Singleton pregnancy
Agreement to formula feed throughout study
ARV history of the following:
ARV-naive with HIV RNA > 400 copies/mL
Currently on HAART and switching to the study regimen for a reason other than virologic failure of a PI-based regimen if HIV RNA < 50 copies/mL
HAART for 90 days or less with HIV RNA > 50 copies/mL but ≥ 1 log drop in HIV RNA within 90 days of screening
ATV-based HAART for ≥ 3 weeks not allowed except for prior MTCT prevention with documented < 50 copies/mL at the time of discontinuation of ATV
Limited prior use of FTC or 3TC allowed
Prior use of ddI, ddC, d4T, ZDV, TDF, ABC or NNRTIs allowed
Pharmacokinetics
Blood samples collected following ≥ 2 weeks of adherent dosing
- Up to 24 hours post-dose for ATV/r from mother in 3rd trimester and ~ 4 weeks post-partum
- Single blood collection from mother and neonate (umbilical cord) at Labor and Delivery
PK parameters derived by non compartmental PK analysis: Cmax, Tmax, Cmin, AUC(TAU), T-Half
ATV and ritonavir analyzed by LC/MS/MS
- ATV standard curve range (10-10,000 ng/mL)
- Ritonavir standard curve range (5-5,000 ng/mL)
Statistics
-- Descriptive statistics for third trimester and post-partum parameters were calculated
-- Ratios of adjusted geometric means, point estimates and 90% confidence intervals for ATV Cmax, AUC(TAU) and Cmin in the third trimester relative to postpartum data were calculated with postpartum data as a reference
--Ratios of geometric means, point estimates and 90% confidence intervals for ATV Cmax, AUC(TAU) and Cmin were calculated for third trimester data relative to pooled historical HIV+ data and for postpartum data relative to historical HIV+ data
Safety
All safety assessments on mothers and infants including clinical examination and laboratory testing at each study visit at the time of the interim datalock are reported
Incidence of AEs were tabulated and reviewed for potential significance and clinical importance
Interim Analysis
An interim analysis was planned once the PK data from the third trimester in 12 subjects were available.
Targeted exposures and a decision tree for the interim analysis is presented in Figure 2

RESULTS
Demography
At the time of the interim analysis,
- Thirty-five (35) expectant mothers were enrolled; twenty-one (21) were treated; Twelve (12) underwent third trimester intensive PK
- One (1) mother discontinued due to premature delivery
- Nine (9) had completed the study
- Eleven (11) were still on treatment
- There were ten (10) live-born infants
3rd Trimester Interim PK
The geometric mean ATV AUC(TAU) observed in the third trimester was 26.6 _g-h/mL and fell below the protocol-defined minimum AUC(TAU) of 30 _g-h/mL
All individual ATV Cmin values, however, were above the protocol-defined minimum Cmin of 150 ng/mL; the lowest observed Cmin in the third trimester was 196 ng/mL
Based on the predefined PK criteria for the interim analysis, these exposures will trigger a protocol-defined dose increase and evaluation of ATV/r 400/100 once daily with ZDV/3TC twice daily in the third trimester
Third trimester ATV exposures at 300/100 were comparable with those observed by Ripamonti et al[3]
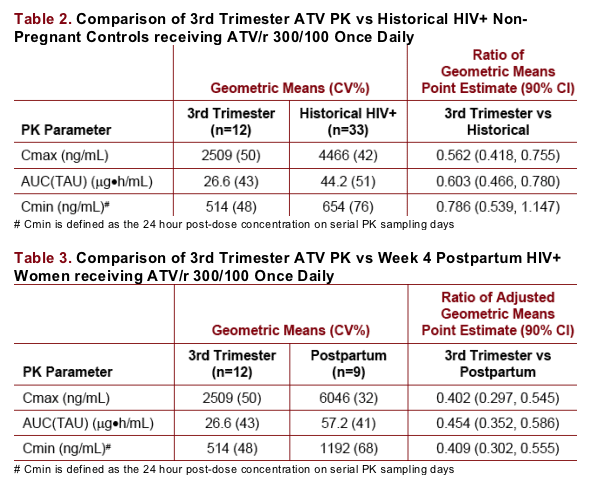
ATV AUC(TAU), Cmax, and Cmin were lower in the third trimester of pregnancy relative to historical data from HIV-infected subjects (Table 2) and week 4 postpartum (Table 3).
The finding of notable difference between 3rd trimester and postpartum data is in contrast to Ripamonti et al, who reported a mean time of postpartum PK as 10 weeks (71 days) after delivery[3]
Postpartum ATV AUC(TAU) and Cmin were approximately 30% and 82% higher (90% CI: 0%-69% and 17%-183%, respectively), relative to historical data from HIV-infected subjects, suggesting the PK of ATV had not returned to baseline by 4 weeks postpartum
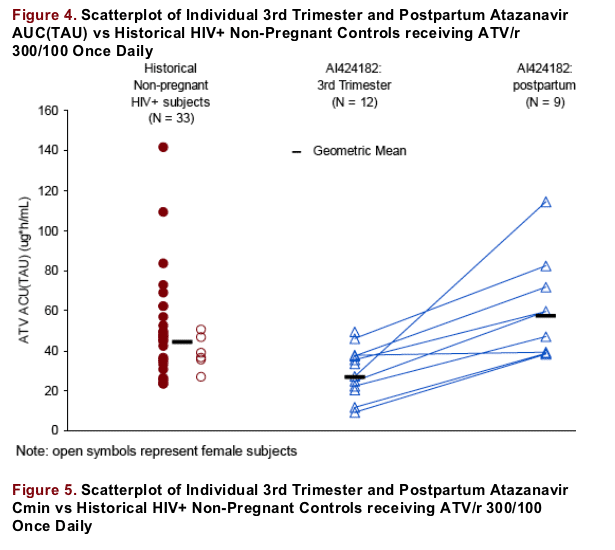
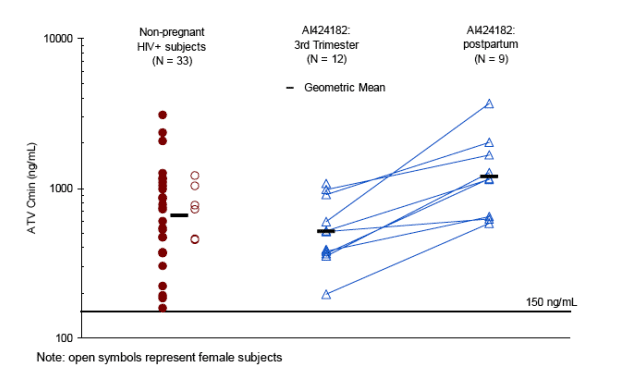
The effect of pregnancy was relatively consistent across subjects: the subjects with the highest AUCs in the third trimester tended to have the highest AUCs post-partum (Figure 4, connected symbols). Similar trends were observed
with ATV Cmin (Figure 5)
Note that all ATV Cmins were > 150 ng/mL (~10-fold the protein binding adjusted EC90 for ATV vs wild-type HIV)
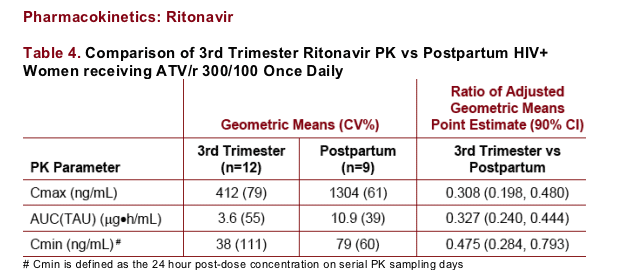
Ritonavir plasma concentrations are reduced in the third trimester of pregnancy relative to postpartum
Ritonavir plasma concentrations were comparable between postpartum and historical data in HIV-infected subjects (data not shown)
Safety: Mothers
There were no deaths or AEs leading to discontinuation. There were no on-study CDC Class C AIDS events.
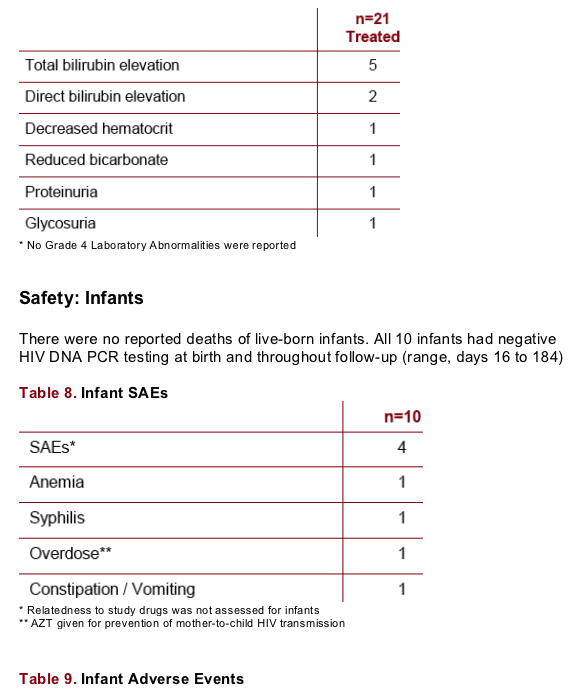
Safety: Infants
There were no reported deaths of live-born infants. All 10 infants had negative HIV DNA PCR testing at birth and throughout follow-up (range, days 16 to 184)

No infant had a ≥ Grade 1 total bilirubin in the age Day 1 to Day 14 period. One infant had a Grade 3 total bilirubin (4.5 mg/dL at age 15 days). Other Grade 3 to 4 laboratory abnormalities were hyperkalemia (2/9 subjects)
Most infants had increasing bilirubin through the first 3 days of life, with resolution to normal levels thereafter (Figure 6)
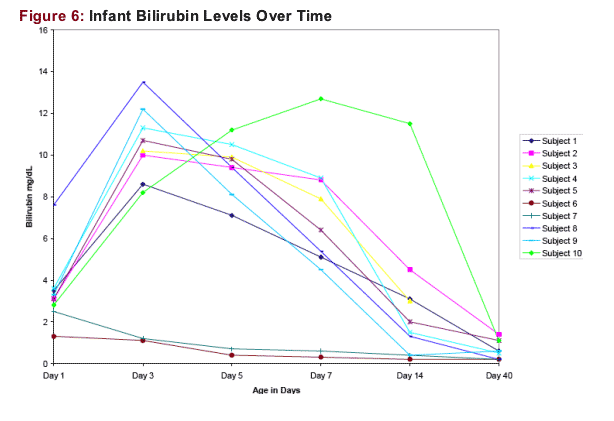
Data for Day 3,5 and 7 are +/- 1 day
Data for Day 14 are +/- 2 days
Data for Day 40 are +/- 4 days
REFERENCES
[1] M Mirochnick, E Capparelli, Pharmacokinetics of antiretrovirals in pregnant women. Clin Pharmacokinet. 2004; 43(15):1071-87.
[2] M Mirochnick, A Stek, E Capparelli, et al. Adequate lopinavir exposure achieved with a higher dose during the third trimester of
pregnancy (poster 710). 13th CROI, Denver, CO, USA 2006.
[3] D Ripamonti, D Cattaneo, F Maggiolo, et al. Atazanavir plus low-dose ritonavir in pregnancy: pharmacokinetics and placental
transfer. AIDS. 2007 Nov 30; 21(18): 2409-15.
[4] M Natha, P Hay, G Taylor, et al. Atazanavir use in pregnancy: a report of 33 cases (poster 750). 14th CROI, Los Angeles, CA,
USA, 2007.
|
| |
|
 |
 |
|
|