 |
 |
 |
| |
Data on 52-Week Safety and Efficacy of Tesamorelin, a Growth Hormone-releasing Factor Analogue, in HIV-infected Patients with Abdominal Fat Accumulation
|
| |
| |
Reported by Jules Levin
15th CROI, Feb 3-6, 2008, Boston
J Falutz1, S Allas2, J C Mamputu2, D Kotler3, M Somero4, D Berger5, S Brown6, G Richmond7, J Fessel8, and Steven Grinspoon*9
1Montreal Gen Hosp, McGill Univ Hlth Ctr,Canada; 2Theratechnologies Inc, Montreal, Canada; 3St Luke`s Roosevelt Hosp Ctr, Columbia Univ Coll of Physicians and Surgeons, New York, NY, US; 4Palm Springs, CA, US; 5Northstar Hlth Care, Chicago, IL, US; 6AIDS Res Alliance, West Hollywood, CA, US; 7Broward Gen Med Ctr, Fort Lauderdale, FL, US; 8Kaiser Fndn Res Inst, CA, US; and 9Massachusetts Gen Hosp, Harvard Med Sch, Boston, US
AUTHOR CONCLUSIONS
Administration of 2 mg tesamorelin to HIV patients with lipohypertrophy for 52 weeks was overall well tolerated without significant effect on glucose parameters, and resulted in improved body composition, including maintenance of VAT loss and preservation of subcutaneous adipose tissue and limb fat. In addition, treatment with tesamorelin was associated with a clinically significant decrease in triglycerides over 52 weeks. Overall, these results suggest that tesamorelin may be beneficial for the treatment of HIV patients with lipohypertrophy.
ABSTRACT
Background: Many HIV patients on ART show increased visceral adipose tissue (VAT), a known cardiovascular risk factor. Previous data showed that administration of tesamorelin (TH9507) to HIV patients with lipohypertrophy led to significant improvements in VAT, lipids and patient-reported outcomes at week 26 and sustained VAT loss at week 52 with continued treatment. Here we report additional data on safety and body composition.
Methods: Patients on ART with central fat accumulation were randomized to tesamorelin 2 mg (n = 273) or placebo (n = 137) subcutaneously daily for 26 weeks. At week 26, patients who first received tesamorelin were re-randomized to 2 mg tesamorelin (n = 154) or placebo (n = 50) for an additional 26-week treatment, whereas patients on placebo were switched to tesamorelin (n = 111). At week 52, end-points were assessment of safety, including glucose parameters, and duration of effects on VAT by computed tomography (CT) scan. All efficacy analyses were performed on the intent-to-treat population, whereas safety evaluation was performed on the safety population.
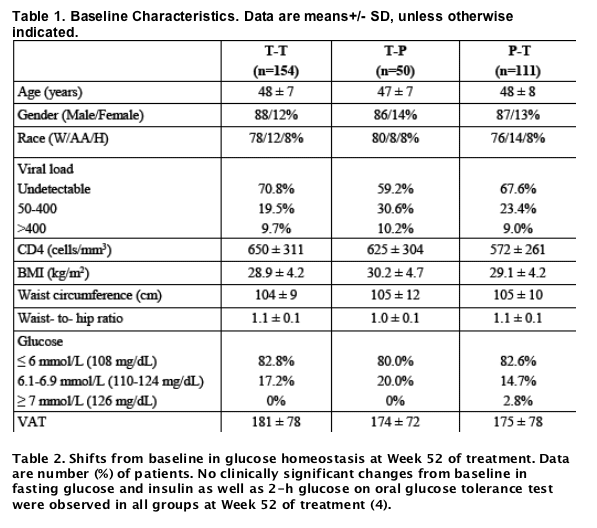
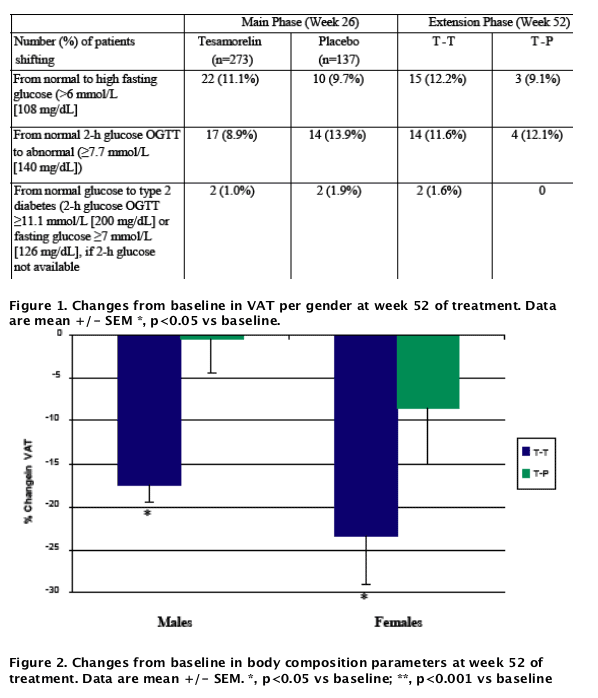
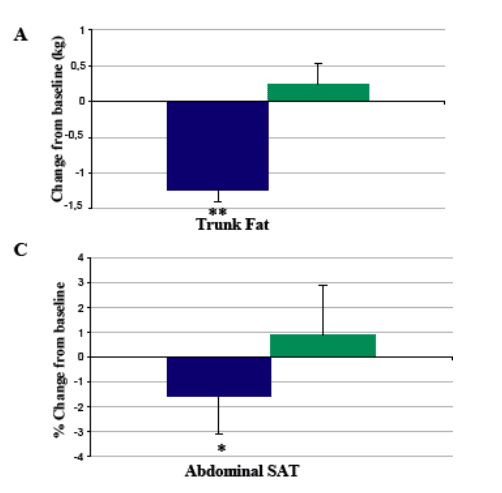
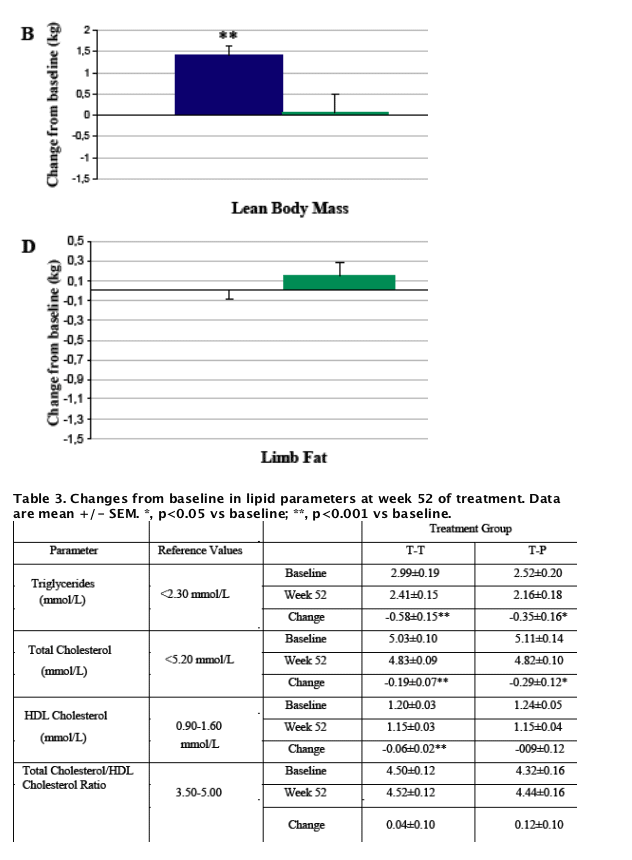
Results: Study population included 275 males and 40 females. Mean age was 48±7 (SD) years, waist circumference 104±10 cm. There were no clinical differences in shifts from normal to high fasting glucose between patients treated with tesamorelin for 52 weeks (12.2%) vs 26 weeks (11.1%), while the percentage in the placebo group was 10% at week 26. The percentage of patients who went from normal 2-hour oral glucose tolerance test (OGTT) to abnormal was 11.6% among patients treated with tesamorelin for 52 weeks vs 12.1% among patients who stopped treatment at week 26. At week 52, changes from baseline in VAT were significant in both males and females in the tesamorelin group (-17.4 and -23.4%, respectively). As observed with VAT, the decrease from baseline in trunk fat at week 26 was sustained at week 52 of treatment (-1.2±2.2 kg, p = 0.51 vs week 26), whereas no further changes were observed in SAT (-7.8±39.0 cm2, p = 0.22 vs week 26) and limb fat (+0.0±1.1 kg, p = 0.27 vs week 26). Finally, the increase from baseline in lean body mass at week 26 was sustained at week 52 of treatment (+1.4±2.9 kg, p = 0.63 vs week 26).
Conclusions: Treatment with 2 mg tesamorelin for 52 weeks was overall well tolerated, without clinical effect on glucose parameters, and resulted in improved body composition, including maintenance of VAT loss and preservation of subcutaneous adipose tissue and limb fat. These results suggest that tesamorelin may be beneficial for the treatment of HIV patients with lipohypertrophy.
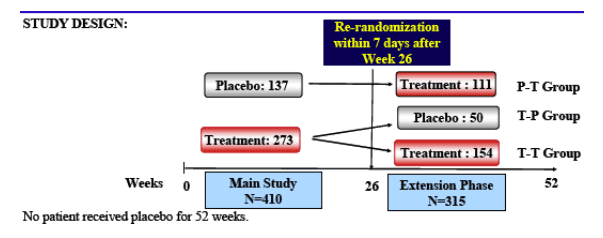
ENDPOINTS
Week 26:
Primary endpoint: % change in VAT from baseline by computed tomography (CT) scan at L4-L5.
Secondary endpoints: triglycerides, total cholesterol/HDL cholesterol ratio, IGF-1 levels, patient outcomes related to body image, safety (glucose, insulin, adverse events and others)
Week 52: Primary endpoint: safety (glucose, insulin, adverse events and others)
Other endpoints: duration of effect on VAT following a 26-week treatment with 2 mg tesamorelin; 52-week efficacy data on VAT.
RESULTS
|
| |
|
 |
 |
|
|