 |
 |
 |
| |
On-treatment responses at weeks 4 and 12 can be used to predict sustained virologic response rates in HIV HCV co-infected patients treated with peginterferon alfa-2a (40KD) (PEGASYS) and ribavirin (COPEGUS)
|
| |
| |
Reported by Jules Levin
15th CROI, Feb 3-6, 2008, Boston
Maribel Rodriguez-Torres1, Juergen Rockstroh*2, Jean Depamphilis3, Giampiero Carosi4, Douglas T. Dieterich5, and Francesca Torriani6
Fundaci-n de Investigaci-n de Diego, Santurce, PR, USA1; University of Bonn, Bonn, Germany2; Roche, Nutley, NJ, USA3; Universita Degli Studi di Brescia, Brescia, Italy4; Mount Sinai School of Medicine, New York, NY, USA5; University of California, San Diego, CA, USA6
The rate of SVR was highest in patients who had cleared HCV RNA at week 4 or 12 of treatment:
_ The rate of SVR was highest in patients achieving an RVR (82% in those infected with HCV genotype 1 and 94% in those infected with HCV genotype 2 or 3).
_ Among patients with a cEVR, the SVR rate was 63% in those infected with genotype 1 and 70% in those infected with HCV genotype 2 or 3.
INTRODUCTION
Chronic hepatitis C is more difficult to treat in patients with HIV-HCV co infection than HCV mono-infection (1). In the APRICOT study, a sustained virologic response (SVR) was obtained in 29% of co-infected patients with HCV genotype 1 infection and 62% of those with HCV genotype 2 or 3 infection after 48 weeks of treatment with peginterferon alfa-2a (40KD) 180 _g/week plus ribavirin 800 mg/day (2).
It is now clear that the virologic response at weeks 4 and 12 of combination therapy can be used to guide the management of patients with HCV mono-infection (3). Approximately 90% of genotype 1-infected patients with a rapid virologic response (RVR) at week 4 and approximately 70% of those with a complete early virologic response (cEVR) at week 12 ultimately achieve an SVR (4).
The objective of this analysis is to evaluate the predictive value for SVR of the virologic response at both week 4 and week 12 of treatment in HIV-HCV (genotype 1, 2 or 3) co-infected patients in APRICOT.
AUTHOR CONCLUSIONS
The predictive value for SVR of the virologic response at week 4 and week 12 of combination therapy is similar in HIV-HCV co-infected patients to that in HCV mono-infected patients.
Whilst RVR at week 4 is the strongest predictor of SVR, patients with a cEVR at week 12 also achieve high SVR rates. Week 4 and 12 responses can be used to guide treatment decisions in HIV-HCV co-infected patients.
Prospective studies are needed to show whether these responses can be used together with other baseline predictors to guide treatment and optimize outcomes in co-infected patients, for example with abbreviated (i.e., 24-week) regimens for those with an RVR or extended (i.e., 72-week) regimens in those with a slow response, as has been demonstrated in HCV mono-infected patients (5,6).
METHODS
Patients
--Adult patients eligible for APRICOT were required to have HIV-1 infection confirmed by the presence of anti-HIV antibodies or HIV RNA in serum.
--Patients with CD4+ counts ÅÜ200 cells/mm3 were eligible, regardless of HIV RNA level; those with CD4+ counts between 100 and 199 cells/mm3 were eligible if their serum HIV RNA level was <5000 copies/mL.
--Patients were not required to be on antiretroviral therapy (ART) at the time of enrollment. If taking ART, they were required to have been on a stable regimen for at least 6 weeks. If not on ART, they were required to have been off treatment for the previous 8 weeks.
--With respect to their HCV disease status, patients eligible for APRICOT were required to have quantifiable serum HCV RNA (ÅÜ600 IU/mL), elevated alanine aminotransferase levels in serum, a biopsy result consistent with the diagnosis of chronic hepatitis C, and compensated liver disease.
--The complete inclusion and exclusion criteria and study design of APRICOT have been described elsewhere (2).
Treatment
--Patients included in this analysis were those who were randomly assigned to 48 weeks of treatment with peginterferon alfa-2a (40KD) 180 _g/week plus ribavirin 800 mg/day.
Outcomes
--The on-treatment virologic responses at weeks 4 and 12 were divided into four mutually exclusive categories (Table 1).
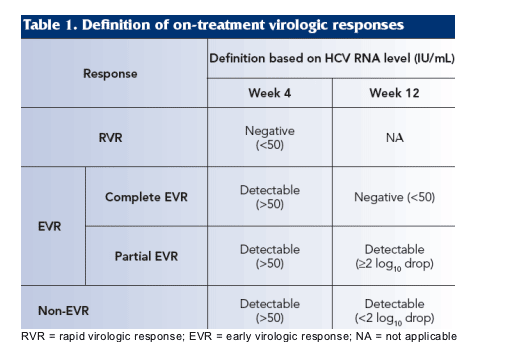
--SVR was defined as undetectable HCV RNA (<50 IU/mL) at the end of a 24 week untreated follow-up period (week 72).
--Rates of SVR were determined as a function of the on-treatment virologic response to therapy at weeks 4 and 12.
RESULTS
A total of 271 of 289 patients included in the intent-to-treat analysis were infected with HCV genotype 1, 2 or 3.
Overall, 21% (60/289) of patients achieved an RVR; 26% (74/289) of patients had a cEVR and 21% (61/289) of patients had a partial EVR (pEVR).
The distribution of SVR rates by the on-treatment virologic response are presented in Figure 1 for patients infected with HCV genotype 1 and in Figure 2 for patients infected with HCV genotype 2 or 3.
Figure 1. Virologic responses at week 4 and 12 in HIV-HCV (genotype 1) co-infected patients treated for 48 weeks withpeginterferon alfa-2a 180 _g/week plus ribavirin 800 mg/day in APRICOT.
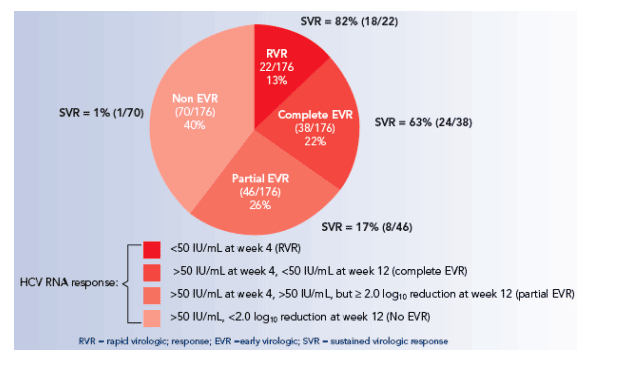
The rate of SVR was highest in patients who had cleared HCV RNA at week 4 or 12 of treatment:
_ The rate of SVR was highest in patients achieving an RVR (82% in those infected with HCV genotype 1 and 94% in those infected with HCV genotype 2 or 3).
_ Among patients with a cEVR, the SVR rate was 63% in those infected with genotype 1 and 70% in those infected with HCV genotype 2 or 3.
Figure 2. Virologic responses at week 4 and 12 in HIV-HCV (genotype 2 or 3) co-infected patients treated for 48 weeks with peginterferon alfa-2a 180 _g/week plus ribavirin 800 mg/day in APRICOT.
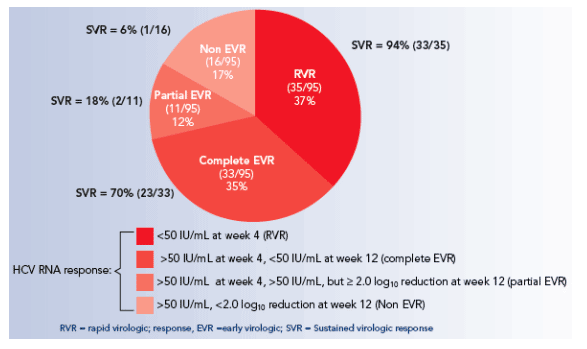
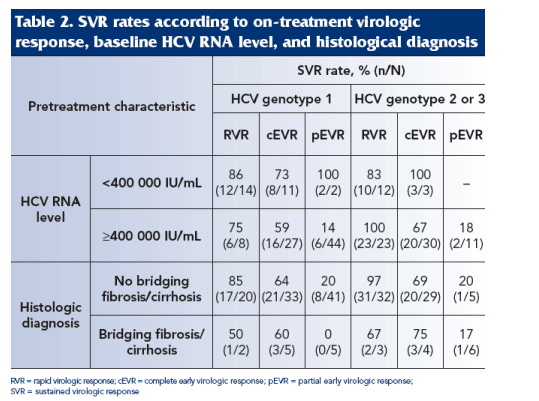
The rate of SVR was lowest in those with detectable HCV RNA atweek 12 (i.e., in those with a pEVR or non-RVR; Figures 1 and 2).
Among patients with an RVR or cEVR, the rate of SVR was highest in those with a low pre-treatment HCV RNA level (<400 000 IU/mL) and in those without advanced fibrosis (Table 2).
REFERENCES
1. Alberti A, Clumeck N, Collins S, Gerlich W, Lundgren J, Palu G, et al. Short statement of the first European Consensus Conference on the treatment of chronic hepatitis B and C in HIV co-infected patients. J Hepatol 2005; 42(5):615 624.
2. Torriani FJ, Rodriguez-Torres M, Rockstroh JK, Lissen E, Gonzalez-Garcia J, Lazzarin A, et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection in HIV-infected patients.N Engl J Med 2004; 351(5):438-450.
3. Marcellin P, Heathcote EJ, Craxi A. Which patients with genotype 1 chronic hepatitis C can benefit from prolonged treatment with the ÅeaccordionÅf regimen? J Hepatol 2007; 47(4):580-587.
4. Marcellin P, Jensen D, Hadziyannis S, Ferenci P. Differentiation of early virological response (EVR) into RVR, complete EVR (cEVR) and partial EVR (pEVR) allows for a more precise prediction of SVR in HCV genotype 1 patients treated with peginterferon alfa-2a (40KD) (Pegasys) and ribavirin (Copegus) [abstract no. 1308]. Hepatology 2007; 46(4 Suppl 1):818A.
5. Ferenci P, Laferl H, Scherzer T-M, Brunner H, Maieron A, Gschwantler M, et al. Response-guided therapy in a prospective trial of peginterferon alfa-2a (40KD)/ribavirin treatment in patients with genotypes 1 or 4 [abstract 1301]. Hepatology 2007; (46 Suppl 1):814-5A.
6. Sanchez-Tapias JM, Diago M, Escartin P, Enriquez J, Romero-Gomez M, Barcena R, et al. Peginterferon-alfa2a plus ribavirin for 48 versus 72 weeks in patients with detectable hepatitis C virus RNA at week 4 of treatment. Gastroenterology 2006; 131(2):451-460.
|
| |
|
 |
 |
|
|