 |
 |
 |
| |
Therapeutic Drug Monitoring of Lopinavir and Saquinavir in Thai HIV-infected Children
|
| |
| |
Reported by Jules Levin
15th CROI, Feb 3-6, 2008, Boston
Jasper van der Lugt1, Torsak Bunupuradah1, Pope Kosalaraksa2, Thanyawee Puthanakit1, Chulapan Engchanil2, Waraporn Sakornjun1, Meena Gorowara1,
Kiat Ruxrungtham1,3, David Burger4, Jintanat Ananworanich1,5
1The HIV Netherlands Australia Thailand Research Collaboration (HIV-NAT), Bangkok, Thailand; 2Khon Kaen University, Khon Kaen, Thailand; 3Chulalongkorn University, Bangkok, Thailand;
4Radboud University Nijmegen Medical Center, Nijmegen, The Netherlands; 5South East Asia Research Collaboration with University of Hawaii (SEARCH), Bangkok, Thailand
Author Discussion
· Adequate plasma levels for SQV and LPV were found for almost all patients at each time point
· Despite the dose reduction over time for SQV, the Cmin levels remained well above the target level
· Growth and development likely contributed to the high inter- and intra-variability among the patients
· No correlation between plasma levels and lipid elevations was noted for either drug. This is in contrast with our previous reports that show LPV Cmin above 5.5 mg/L and maximum plasma concentration (Cmax) above 15 mg/L correlated with
hypercholesterolemia3
· Consistent with our previous reports, Cmin above 1.0 mg/L for LPV correlated with viral load suppression3, while there was no correlation for SQV
Author Conclusion
· High drug levels of SQV and LPV were consistently observed in children treated with standard dosages of both drugs
· Dose reduction of SQV did not influence the minimum steady-state SQV plasma concentration
· Lower-than-manufacturer-recommended dosages of SQV and LPV may be preferred in Thai children
Background
Therapeutic Drug Monitoring (TDM):
· The processes of growth and development have a significant impact on drug metabolism
· Low drug concentrations may lead to virological failure while high drug concentrations may lead to toxicity
· TDM measures drug concentrations at a particular time point
· The use of TDM makes it possible to optimize plasma drug concentrations of antiretroviral drugs in HIV-infected children
· Inter- and intra-variability are important parameters for interpreting TDM results
Thai Setting:
· In the Thai setting, second-line antiretroviral therapy options are limited for children
· The combination of saquinavir (SQV) and lopinavir (LPV) is a potent second-line treatment regimen in this population1 (see poster 586 CROI 2008)
· High plasma SQV levels have been observed in Thai adults and children using standard dosages of SQV1,2
· The data on appropriate dosages to maintain plasma levels above the minimum inhibitory concentration (IC50) for wild-type HIV viruses for SQV and LPV/ritonavir (LPV/r) in Thai children are unknown
Objective
· To evaluate the plasma concentrations of SQV and LPV over a 96-week period
· To study the variability between and within the patients
Study Sites
· HIV-NAT in Bangkok (n = 20)
· Khon Kaen University in Khon Kaen, Northeast Thailand (n = 30)
Methods
· Fifty HIV-infected children under the age of 16 years who were PI-naive and failing nonnucleoside reverse transcription inhibitor (NNRTI)-based treatment were enrolled in this study
· Patients were assigned to:
- LPV/r dosed at 230/57.5 mg/m2 twice daily (BID), provided as capsules (LPV/r 133/33 mg per capsule) and supplemented with oral solution containing LPV/r
80/20 mg/mL
- SQV dosed at 50 mg/kg BID, provided as 200 mg SQV mesylate capsules
- Lamivudine, 4 mg/kg BID, was added in patients who had no prior exposure
· The study physician could change the dose based on his/her discretion
· Predose plasma concentrations (Cmin) were prospectively collected at weeks 12, 24, 36, 48, 60, 72, 84, and 96
· Patients with plasma concentrations below 0.1 mg/L for LPV and 0.02 mg/L for SQV were excluded from analysis as this indicated nonadherence
· Desired Cmin was defined as levels above the IC50 for each drug (>0.1 mg/L for SQV and >1.0 mg/L for LPV)
· Inter-variability was calculated as a coefficient of variance (CV) and the intrasubject variability was calculated as the standard deviation (SD) divided by the mean over the different time points
· A paired t-test was used to detect differences between time points in Cmin
Definitions:
Intervariability: representing the differences between the subject was defined as:
standard deviation X 100 = coefficient of variance
mean Cmin levels different subjects
Intravariability: representing the differences within the
subject was defined as:
standard deviation X 100 = coefficient of variance
mean Cmin levels same subject
Results
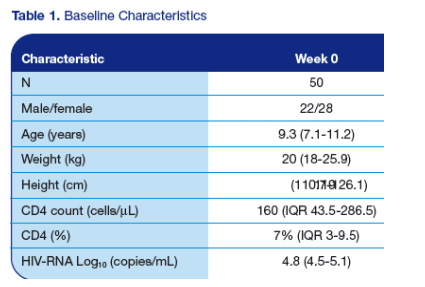
· For baseline characteristics see Table 1
· Samples from 42-48 patients were available for analysis at each time point. Missing values were lost to follow up or were levels below 0.1 mg/L (for LPV)
or 0.02 mg/L (for SQV)
· No correlation was found between the plasma drug concentration and an increase of any of the lipid parameters, as calculated by the linear regression model
· A correlation was found between the viral load and the cut off of 1.0 mg/L for LPV (p=0.02); for SQV, such a correlation was not found, either for 0.1 mg/L or for 0.28 mg/L as a cut off
· Table 2 depicts the mean (SD) Cmin levels for the different time points and the inter- and intrapatient variability
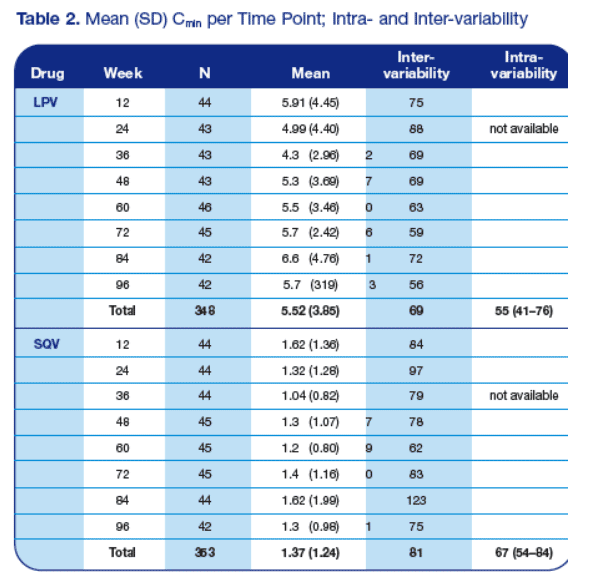
· For LPV, 19 out of the 348 samples had a plasma drug concentration below 1.0 mg/L (5.5%); for SQV, 13 out 353 had plasma drug concentrations below
0.1 mg/L. These values have shown to be the minimum value to suppress the wild-type HIV strain (www.HIVPharmacology.com)
· Cmin levels remained consistent throughout the study, despite drug dosages that were significantly reduced by the end of the study (p=0.55 and p=0.09 for LPV and SQV, respectively)
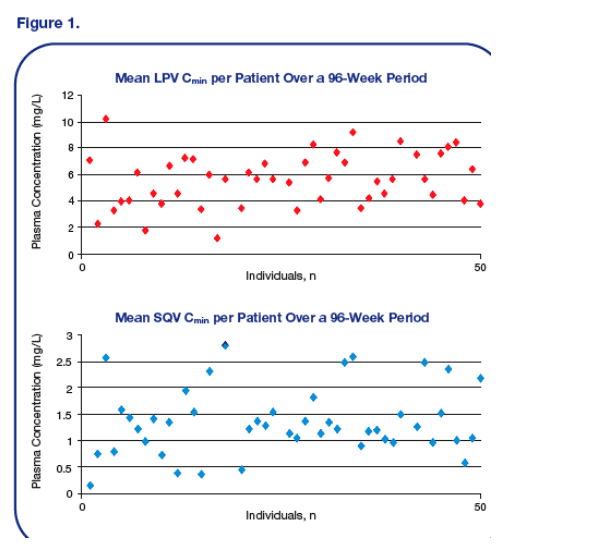
· Figure 1 depicts the mean Cmin at all time points for each individual patient
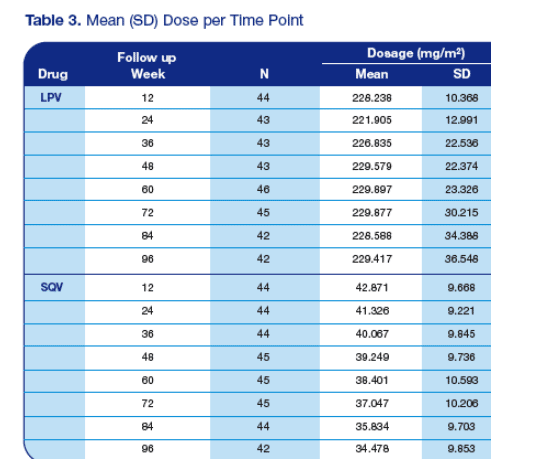
· Table 3 depicts the mean drug dosages per time point
· The SQV dose was reduced by approximately 8 mg per/kg whereas the LPV dose was not reduced
References
1. Ananworanich J, Hill A, Siangphoe U, Hirschel B for The Staccato Study Group. A prospective study of efficacy and safety of once daily saquinavir/ ritonavir plus 2 NRTIs in treatment naive Thai patients. Antiviral Therapy. 2005:10:761-7.
2. Autar RS, Boffito M, Hassink E, Wit F, Ananworanich J, Siangphoe U, Pozniak A, Cooper D, Phanuphak P, Lange J, Ruxrungtham K, and Burger D. The interindividual variability of once-daily ritonavir boosted saquinavir pharmacokinetics in Thai and United Kingdom patients. J Antimicrobial Chemother. 2005:56(5):908-13.
3. Ananworanich J, Kosalaraksa P, Hill A, Siangphoe U, Bergshoeff A, Pancharoen C, Engchanil C, Ruxrungtham K, Burger D and the HIV-NAT 017 Study Team. Pharmacokinetics and 24-week efficacy/safety of dual boosted saquinavir/lopinavir/ritonavir in nucleoside pre-treated children. Ped Infect Dis. 2005:24:874-9.
|
| |
|
 |
 |
|
|