 |
 |
 |
| |
Pharmacokinetics and Potent Anti-HIV-1 Activity of Amdoxovir Plus Zidovudine in a Randomized Double-blind Placebo-controlled Study
|
| |
| |
Reported by Jules Levin
15th CROI< Feb 3-6, 2008, Boston
Robert Murphy1, Carlos Zala2, Claudia Ochoa3, Phillip Tharnish4, Judy Mathew4, Emilie Fromentin5, Ghazia Asif5, Selwyn J. Hurwitz5, Nancy M. Kivel4, Raymond F. Schinazi5
1Univ Pierre et Marie Curie-Paris VI, Paris, France, 2Hosp Privado Modelo, Buenos Aires, Argentina, 3ACLIRES-Argentina, Buenos Aires, Argentina, 4RFS Pharma, LLC, Tucker, GA, US, 5Emory Univ Sch of Med/VA Med Ctr, Atlanta, GA
AUTHOR RESULTS & CONCLUSIONS
· DAPD and DAPD/ZDV were safe, effective and well tolerated.
· DAPD/ZDV 200 mg achieved a 2 log10 copies/mL reduction in HIV-1 RNA.
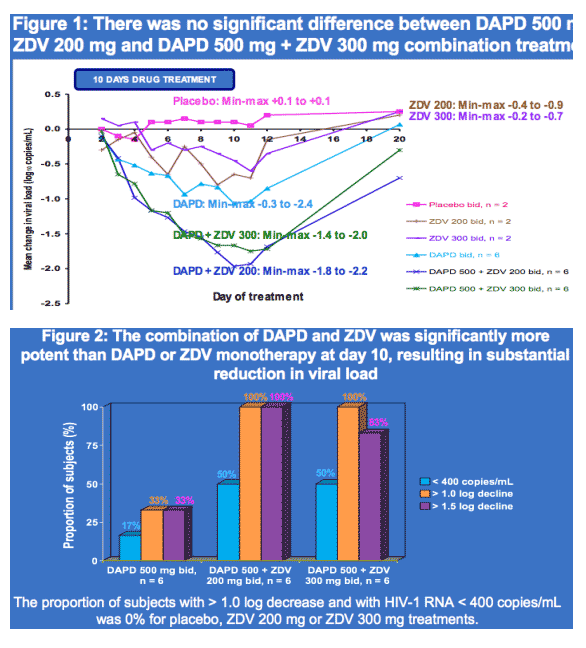
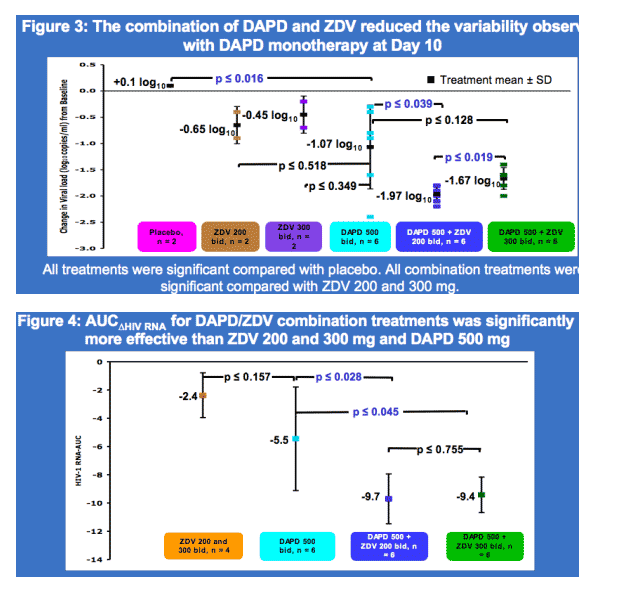
· There was no major difference in VL or AUC reduction when DAPD was combined with either ZDV at 200 or 300 mg bid, supporting the in silico findings.
· DAPD/ZDV VL decline was more than additive compared to DAPD or ZDV alone, suggesting synergy.
· DAPD/ZDV combination markedly decreased VL variability compared with DAPD alone.
· There were no clinically significant changes from Baseline in any of the biochemistry, hematological, or urinalysis parameters studied.
· There were no significant differences in mean change in CD4+ cell count from Baseline at Day 10 between any of the treatment groups.
· There were no clinically relevant changes in hemoglobin or mean corpuscular volume (MCV) from Baseline between treatment groups.
· There were no changes in Baseline mutations, or development of new mutations at Day 10.
· There were no apparent pharmacokinetic drug interactions between DAPD/DXG and ZDV.
· Longer studies with co-formulated DAPD/ZDV are warranted to confirm and extend the results of the current study.
ABSTRACT
Background: Amdoxovir (DAPD) is a NRTI that is synergistic in human lymphocytes with zidovudine (ZDV), and in combination prevents selection of TAMs and K65R mutations. ZDV, the first NRTI developed for HIV, is active, but has well characterized hematologic toxicities. In silico, ZDV (200 mg bid) produces reduced ZDV-monophosphate levels, associated with toxicity, while maintaining ZDV-triphosphate levels necessary for antiviral effect. The objective was to evaluate the safety, pharmacokinetics (PK) and anti-HIV activity of DAPD with and without ZDV at two doses, as a prelude to advanced phase 2 studies.
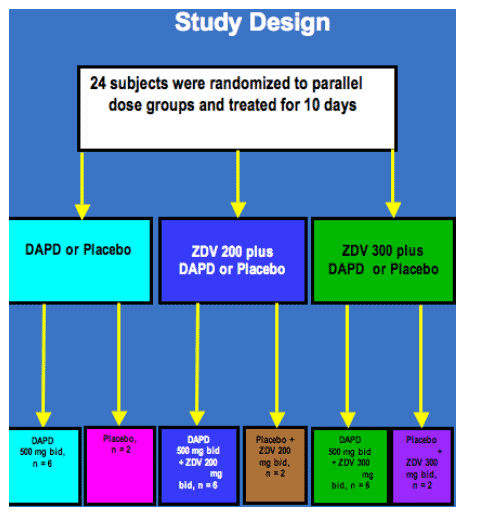
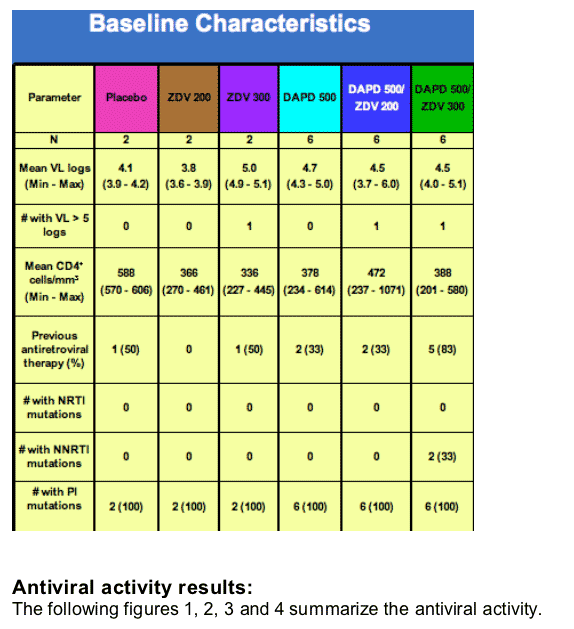
Methods: Twenty-four subjects, not receiving antiretroviral therapy and with plasma HIV-1 RNA (viral load, VL) ≥ 5,000 copies/ml, were randomized to DAPD 500 mg bid, DAPD 500 mg plus ZDV 200 or plus ZDV 300 mg bid for 10 days. In each arm, subjects were randomized 3:1 to DAPD or placebo. VL was determined daily, and PK sampling was performed on Days 1 and 10. Analyses were performed using an intent-to-treat approach. The mean change in VL log10 from baseline at Day 10 and cumulative change in VL log10 from baseline to day 10 (AUCVL) were determined. Plasma DAPD, b-D-dioxolane guanosine (DXG) metabolite, and ZDV levels were measured by LC-MS/MS. Safety was evaluated by proportion of ≥ Grade 3 adverse events per treatment, using ACTG toxicity grading scale. ANOVA was performed to relate AUCDXG and AUCZDV with AUCVL.
Results: Twenty-four subjects, with mean plasma HIV-1 RNA (viral load, VL) 4.5 log10 copies/mL and mean CD4+ 417 cells/ml were randomized and completed the study. On day 10, mean VL log10 change was +0.10 with placebo, -0.65 with ZDV 200, -0.45 with ZDV 300, -1.07 ± 0.80 with DAPD, -1.97 ± 0.16 with DAPD/ZDV 200, and -1.67 ± 0.21 with DAPD/ZDV 300. DAPD/ZDV 200 was significantly more potent than DAPD (p < 0.04), suggesting synergy, and there was markedly decreased VL variability with DAPD/AZT compared with DAPD alone. VL decline was significantly improved with DAPD/ZDV 200 and 300 mg compared with ZDV monotherapy (p ≦ 0.0001). Adverse events were mild to moderate and transient. As anticipated, no statistically significant drug-drug interactions were noted. Preliminary ANOVA of the pooled data identified AUCDXG, but not AUCZDV, as a significant covariate of AUCVL (p < 0.0008).
Conclusions: DAPD and DAPD/ZDV were well tolerated, and the combination produced about a 2 log VL reduction after 10 days. The lack of correlation between variations in plasma AUCZDV with VL decline supports in silico findings. Thus, co-formulated DAPD/ZDV warrants further development for second line therapy
STUDY RATIONALE
GOAL: Develop a DAPD/ZDV co-formulation with reduced resistance and improved safety and efficacy profile.
· DAPD has increased sensitivity to M184V/I mutations and is active against certain TAMs.1
M184V/I is selected by majority of first line therapies and is a likely mutation to be harbored in treatment-experienced subjects.
· ZDV has anti-K65R activity and therefore, could be incorporated as a 'resistance repellent' for the K65R mutations that may result from prolonged treatment with DAPD as well as tenofovir.2, 3
· In human peripheral blood mononuclear cells, the combination of DAPD and ZDV completely prevented the development of DAPD or ZDV-associated resistance.4
In silico, ZDV 200 mg bid compared with ZDV 300 mg bid produced decreased ZDV-MP levels, associated with toxicity, without reducing ZDV-TP levels, necessary for antiviral
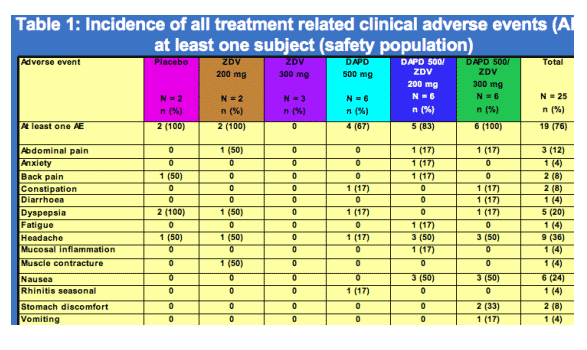
Safety results:
Study drugs were well tolerated. All treatment emergent adverse events (table 1) were mild to moderate and transient. No Grade 3 or 4 laboratory abnormalities developed, and there were no increases in grade from Baseline. No laboratory abnormalities or adverse events led to study drug modification or discontinuation. There were no deaths or serious adverse events.
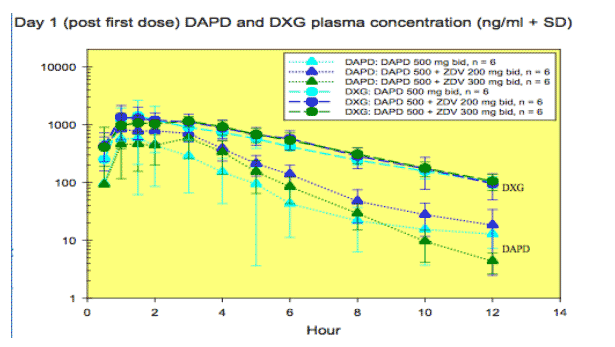
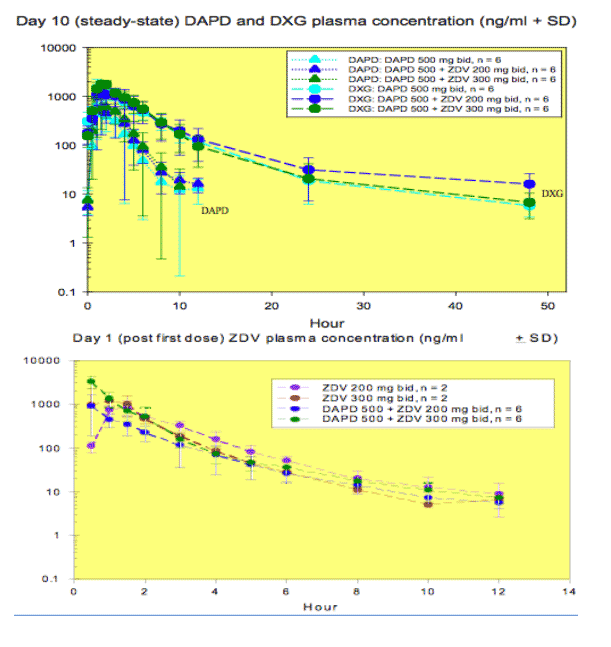
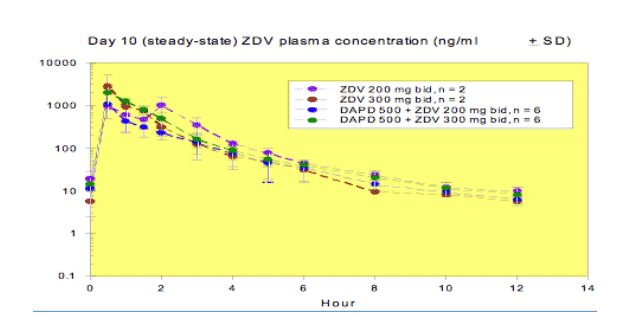
Pharmacokinetic results:
Preliminary data indicated that DAPD is rapidly cleared from plasma and its metabolite, DXG, was adequately delivered to the plasma in the presence or absence of ZDV. A biphasic decline was evident for DXG after 24 hr following the last dose on Day 10. ZDV plasma concentrations on Day 10 were similar to those on Day 1, as expected. The non-compartmental PK parameters for DAPD/DXG and ZDV did not appear to be affected by the co-administration of ZDV 200 or 300 mg or DAPD, respectively.
REFERENCES
1. Mewshaw et al. J AIDS 29:11-20, 2002
2. White et al. Antimicrob Agents Chemother 46: 3437-46, 2002
3. Parikh et al. Antimicrob Agents Chemother 49: 1139-1144, 2005
4. Rapp et al. XVI International HIV Drug Resistance Workshop, Barbados, West Indies, June 12-16, 2007
5. Hurwitz et al. Submitted to Antimicrob Agents Chemother, January 14, 2008
Supported in part by NIH, VA and Emory Center for AIDS research. We acknowledge all subjects who participated in this study for their time and dedication.
COI: Dr. R. F. Schinazi is the Founder of RFS Pharma, LLC and the inventor of DAPD and may receive royalties from the future sales of DAPD.
|
| |
|
 |
 |
|
|