 |
 |
 |
| |
COLCHICINE VERSUS PEG-INTERFERON ALFA 2B LONG TERM THERAPY: RESULTS OF THE 4 YEAR COPILOT TRIAL
|
| |
| |
Reported by Jules Levin
43rd EASL Conference, April 23-27, 2008, Milan, Italy
N.H. Afdhal1, R. Levine2, R. Brown Jr3, B. Freilich4, M. O'Brien5, C. Brass6
1 Beth Israel Deaconess Medical Center, Boston,MA;
2 Syracuse University, Syracuse, NY;
3 Columbia College of Physicians and surgeons, New York, NY;
4 Liver - Pancreas Institute, Kansas City, MO;
5 Boston University, Boston, MA;
6 Schering Plough, Kennilworth, NJ, USA
Afdahl concluded: "There still could be consideration for Peg-Ifn alfa-2b as maintenance therapy in a subset of patients with cirrhosis and portal hypertension who failed eradication therapy.
The mechanism of action we believe is mediated by the effects of portal hypertension and this seems to be more profound in the early phases of treatment."
Maintenance therapy with PEG was associated with improved disease free survival almost exclusively in patients with portal hypertension and should be considered in those patients.
There are 3 large randomized prospective maintenance trials: HALT-C, COPILOT, and EPIC. HALT-C is a large trial by the NIH that look at patients with liver fibrosis and randomized them to a runin phase of peginterferon therapy and then a maintenance phase with peginterferon.
HCV patients who have advanced liver disease and have failed interferon anti-viral therapy may benefit from low dose interferon to prevent clinical disease progression. The COPILOT trial is a four year randomized study of colchicine (COLC) 0.6mg bid versus PEG-IFN alfa 2b (0.5mcg/kg weekly) with the primary endpoints of liver failure, death, transplant, variceal bleeding and HCC.
METHODS: IFN failures with Ishak stage 3 � 6 were randomized directly to receive either COLC or PEG and followed at 3 monthly intervals and screened for HCC by US every 6 months. All clinical endpoints were recorded and independently verified. Analysis was by Kaplan Meier for ITT and also per protocol, on drug, (PP) with patients censored at time of dropout. Dropouts were followed for end points and included in the ITT.
Inclusion/Exclusion Criteria
Failure of prior interferon based therapies.
No treatment for 3 months prior to entry
CTP score 7 or less.
Ishak Fibrosis Stage of 3 or more.
HBsAg and HIV negative.
PRIMARY ENDPOINTS
Death
Liver transplant
HCC
Variceal or Portal hypertension Bleeding
Liver failure�deined in this study as an increase in Childs Pugh Score (CTP) by 2 points WITH ascites, jaundice or encephalopathy
The final endpoint that was reached was the first endpoint reached by the patient.
SUMMARY of 2 Year Results
Peg-IFN alfa-2b was superior to Colchicine as maintenance therapy with 50% reduction in liver related event rate.
Peg-IFN alfa-2b efficacy superior in patients with cirrhosis and portal hypertension.
Excellent safety profile for Peg-IFN alfa-2b and colchicine.
Patients Baseline
83% had cirrhosis in both arms.
48% had Portal HTN in Colchicine arm and 42% in Peg-Ifn arm.
CPT 5 was the majority with 77% on colchicines and 78% on Peg-IFN and about 202% having CPT of 6 or 7.
Patient Disposition
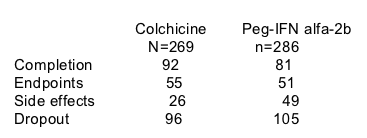
49% of patients failed to complete 4 years of active treatment, most patients continued with followup, the loss to followup was very low. The final overall loss in the study was under 10%.
Viral Studies
Quantification of HCV RNA was inadequate; overall non-significant change in HCV RNA in Peg-IFN group; overall 0.5 log increase in colchicines group.
Failure To Comply
Mean followup was 2 years. Most of the patients who dropped out did so between years 2-4.
PEG IFN Side Effects (causes for stopping therapy)
Lymphadenopathy (1)
Stroke (3), retinopathy (1)
Asthma (1)
Pulmonary hypertension (1)
Renal disease (3)
Pancreatitis (1)
Anemia (1)
Skin rash, hair loss (3).
AUTHOR SUMMARY
Peg-IFN alfa-2 is not superior to colchicines as maintenance therapy in ITT analysis.
Peg-IFN alfa-2b efficacy is superior to colchicines in patients with cirrhosis and ortal hypertension particularly in patients staying on treatment.
No difference in prevention of HCC between Peg-IFN alfa-2b and colchicines.
Side effects requiring stopping therapy increased between 2 and 4 years in Peg-IFN alfa-2b group.
AUTHOR CONCLUSIONS
Afdahl said: "There still could be consideration for Peg-Ifn alfa-2b as maintenance therapy in a subset of patients with cirrhosis and portal hypertension who failed eradication therapy.
The mechanism of action we believe is mediated by the effects of portal hypertension and this seems to be more profound in the early phases of treatment."
Program Abstract
RESULTS:
555 patients were randomized and received at least one dose of drug (COLC 269; PEG 286). The demographics were similar between groups with a mean age of 51 years, 70% male, 87% Caucasian, 78% had cirrhosis and 45% had portal hypertension.
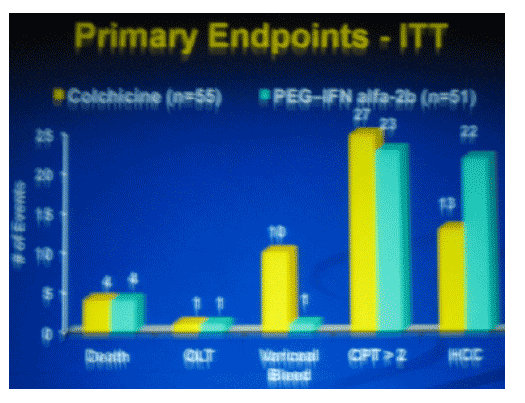
Clinical endpoints were seen in 110 patients (20%); 57 patients on COLC and 53 on PEG by ITT and 45 and 36 on PP analysis respectively.
HCC was a more common presentation in PEG (26) compared to COLC (12).
Complications of portal hypertension were more common in COLC (39) than PEG (26) groups with the major difference occurring in variceal bleeding.
In both ITT and PP analysis there was a benefit in favor of PEG on event free survival only in patients with portal hypertension at 2 and 4 years, which was more prominent in the PP (on drug) analysis (p = 0.027).
Overall, 49% of patients discontinued medication over 4 years secondary to failure to comply in 36% and side effects in 13%.
CONCLUSION: Maintenance therapy with PEG was associated with improved disease free survival almost exclusively in patients with portal hypertension and should be considered in those patients.
|
| |
|
 |
 |
|
|