 |
 |
 |
| |
A Study of Telaprevir (TVR) with Peginterferon Alfa-2a (P) and Ribavirin (R) in Subjects with Well-Documented Prior PR Null Response, Non-Response
or Relapse: Preliminary Results
|
| |
| |
Reported by Jules Levin
43rd EASL, Milan, Italy, April 23-27, 2008
Fred Poordad,1 Mitchell Shiffman,2 Kenneth Sherman,3 Jill Smith,4 Min Yao,5 Shelley George,5 Nathalie Adda,5 John McHutchison6 for the VX06-950-107 Study Team
1Cedars-Sinai Medical Center, Los Angeles, CA, USA; 2McGuire DVAMC, Fairfax, VA, USA; 3University of Cincinnati, Cincinnati, OH, USA; 4Hershey Medical Center, Hershey, PA, USA; 5Vertex Pharmaceuticals Inc., Cambridge,
MA, USA; 6Duke Clinical Research Institute and Duke University, Durham, NC, USA
Introduction
· The majority of hepatitis C virus (HCV) genotype 1 patients do not achieve sustained virological response (SVR) after a 48-week regimen of pegylated interferon alfa-2a/2b (P) and ribavirin (R).1-3
· Retreatment of prior PR non-responders (null and partial responders) and relapsers with PR achieves SVR rates of less than 10% and 30%, respectively.4-7
· Telaprevir (VX-950, TVR) is a specifically-targeted antiviral therapy for HCV (STAT-C) that potently and selectively inhibits the HCV NS3·4A protease.8
· The results in the Phase 2 treatment-na•ve studies of TVR support evaluation of a TVR-based regimen in patients who failed prior PR treatment.
· The ongoing Study 107 was designed to:
- Provide access to TVR for patients who participated in the control arms (PR) of the Phase 2 TVR randomized studies and who failed to achieve SVR
- Provide a unique opportunity to compare in individual patients the antivira response of TVR/PR to that of their original response to PR
- Determine the antiviral responsiveness to TVR/PR in well-characterized null and partial responders and relapsers to PR.
· We report here preliminary Week 4, Week 8 and Week 12 results.
Methods
Preliminary Analysis
· Preliminary analysis included all enrolled patients who received at least one dose of study regimen and who completed at least the Week 4 assessment.
· HCV RNA levels were measured using the HCV RNA Taqman assay Version 2.0 (Roche Molecular Systems Inc., Branchburg, NJ, USA):
- The lower limit of quantification (LLOQ) was <25 IU/mL
- The lower limit of detection (LOD) was <10 IU/mL (below limit of quantification and without detectable signal for HCV RNA).
· Safety assessments were recorded throughout the study.
· Descriptive statistics were used for this preliminary analysis.
Patient population
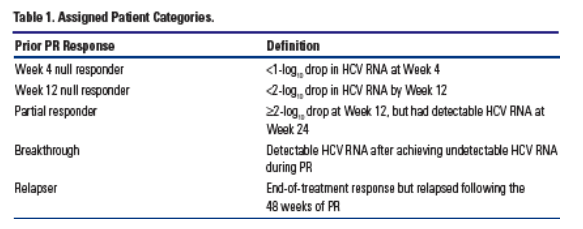
Patients who participated in the control arm (PR) of the Phase 2 TVR randomized studies and who failed to achieve SVR were included and designated as indicated in Table 1.
Study Design
· Open-label treatment of patients from control arms of PROVE1, PROVE2 and PROVE3 (investigation of HCV PROtease inhibition for Viral Evaluation) who met either on-treatment criteria for non-response, or who relapsed after completion of 48-week PR.
· Patients who discontinued because of adverse events (AEs) in the PROVE studies were not eligible for inclusion.
· Standard entry criteria as for other PR studies were applied.
· Study dosing consisted of 12 weeks TVR plus PR, followed by 12 weeks of PR (T12/PR24) at the following doses:
- TVR 750 mg every 8 hours
- P 180 _g subcutaneously, weekly
- R 1000 mg or 1200 mg daily.
· Stopping rule criteria at Week 4 and Week 12 were defined as HCV RNA >25 IU/mL.
RESULTS
Patient Disposition and Baseline Characteristics
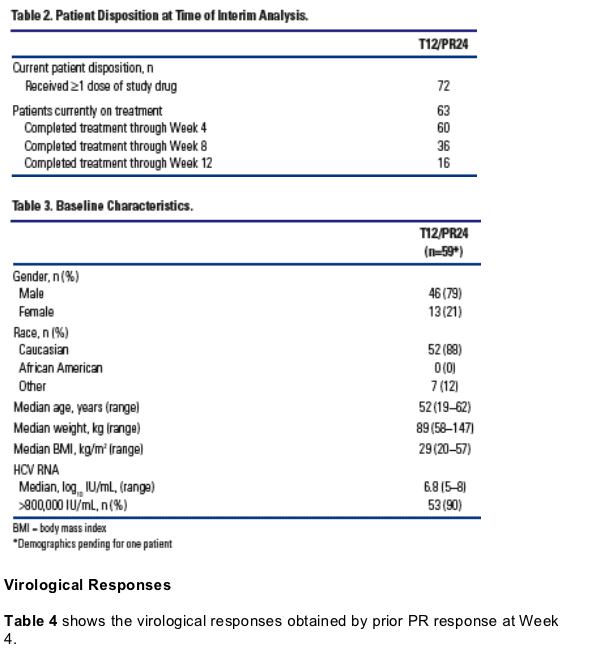
A total of 72 patients received at least one dose of study drug (Table 2).
Premature discontinuations during the first 12 weeks were:
- Six patients due to Week 4 stopping rule
- Two patients due to breakthrough, both at Week 2
- Two patients due to AE (one patient discontinued due to stopping rule).
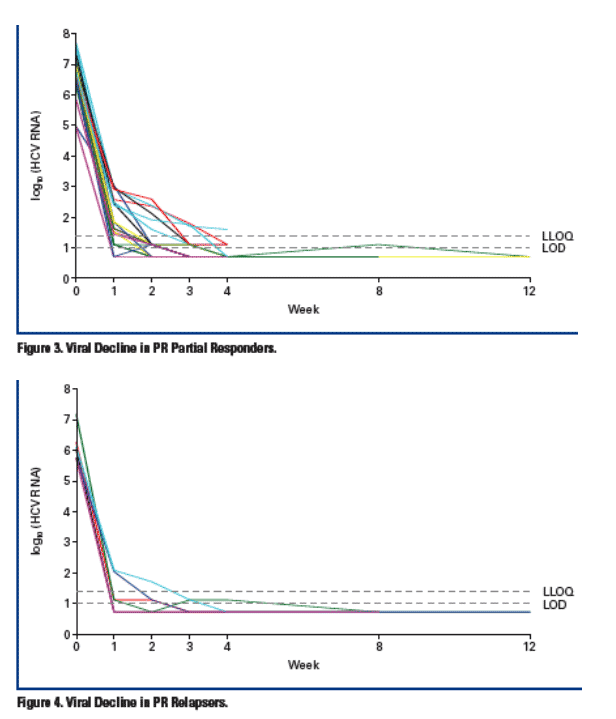
This preliminary analysis is based on the patients who reached at least Week 4 assessment (Table 3).
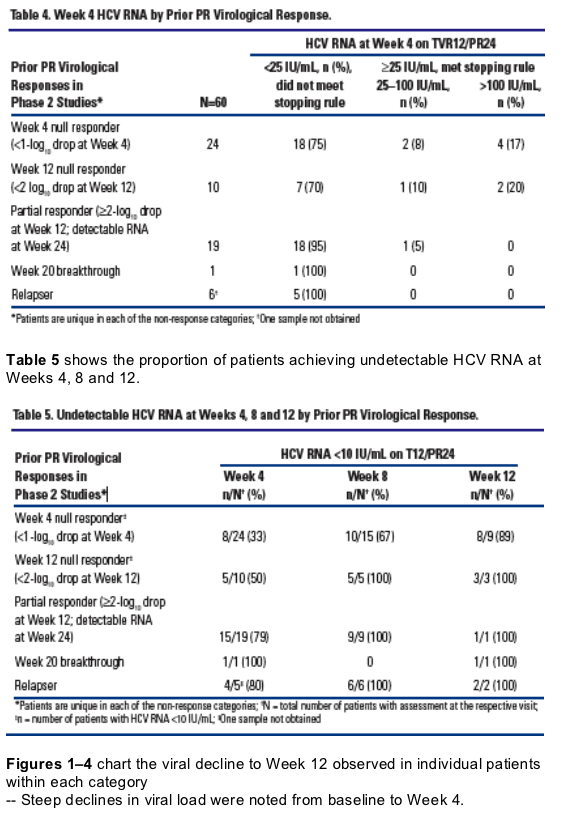
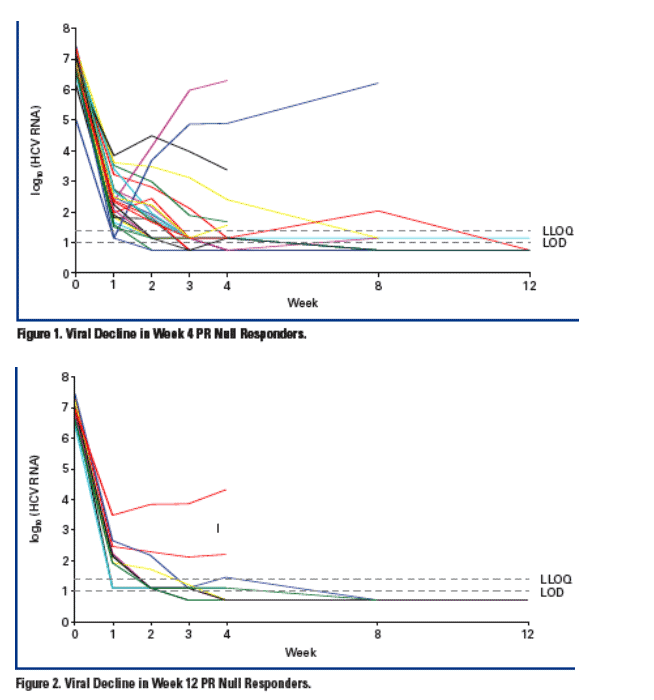
Two patients (2/60, 3%) have developed viral breakthrough.* In each case, viral breakthrough occurred at Week 2
- Both patients were previously Week 4 null responders to PR
- Results of population sequencing for these patients revealed V36M/R155K
variants at the time of breakthrough.
To date, no viral breakthrough was observed in the patients who did not meet the Week 4 stopping rule and continued on treatment.
*Viral breakthrough on TVR/PR was defined as:
· An increase of >1-log10 above the HCV RNA nadir
· An increase in HCV RNA to >100 IU/mL in patients with previously undetectable HCV RNA
Safety and Tolerability
The types of adverse events encountered were those that are expected with PR, and are consistent with those previously reported in patients treated with TVR based regimens in the PROVE studies.
Two patients discontinued treatment due to AEs (one due to pleuritis/costochondritis, and one due to generalized rash).
Conclusions
In this interim analysis of telaprevir-based therapy in HCV genotype 1 patients with well-characterized null response, partial response or relapse to prior pegylated interferon and ribavirin therapy:
- At Week 4, 83% achieved HCV RNA <25 IU/mL
- All of the patients who continued beyond Week 4 maintained their viral responses by Week 8 and Week 12
- Viral breakthrough was infrequent (3%) and was identified very early in treatment
- The safety assessment is consistent with that previously described in the PROVE trials.
The preliminary findings suggest that prior pegylated interferon non-responders, including true null responders, can achieve and maintain on-treatment response with the addition of telaprevir.
Based on this current analysis that demonstrates relapsers and partial responders have a Week 4 response that is similar to that observed in treatment na•ve patients, the protocol is being amended such that these patients will receive the response-driven regimen that is being utilized in the Phase 3 ADVANCE study (12 weeks of telaprevir, in combination with 24 or 48 weeks of PR depending on early viral response). Null responders will receive 48 weeks of PR.
Continuing follow-up of these patients will be required to evaluate the clinical significance of these promising early findings.
References
1. Manns MP, et al. Lancet 2001;358:958-65.
2. Fried MW, et al. N Engl J Med 2002;347:975-82.
3. Hadziyannis SJ, et al. Ann Intern Med 2004;140:346-55.
4. Shiffman ML. Hepatology 2002;36:S128-34.
5. Shiffman ML, et al. Gastroenterology 2004;126:1015-23.
6. Ahmed F, et al. Gastroenterol Clin North Am 2004;33:513-26.
7. Poynard T, et al. 43rd EASL 2008, Milan, Italy, April 23-27 2008.
8. Lin K, et al. Antimicrob Agents Chemother 2004;40:4784-92.
|
| |
|
 |
 |
|
|