 |
 |
 |
| |
HCV Protease Inhibitor Telaprevir, PROVE1 Study: final results of a phase 2 study with peginterferon plus ribaviron in treatment-naive patients with hepatitis C
|
| |
| |
Reported by Jules Levin
EASL- 43rd Annual Meeting of the European Association for The Study of the Liver
Milan, April 23-27, 2008
PROVE1: investigation of HCV PROtease Inhibition for Viral Evaluation
Randomized, controlled, Phase 2b clinical trial
--260 treatment-naive subjects with HCV genotype 1 infection
--37 US clinical centers
--Compares telaprevir (TVR)-based treatment arms of 12-, 24- and 48-week durations against a 48-week Peg-IFN/RBV control arm
--interim results were presented in 2007 (McHutchison JM et al EASL 2007; Jacobson I et al AASLD 2007)
Objectives
--Assess the SVR rate that could be achieved with TVR-based treatment
--determine the optimal duration for TVR-based treatment
--Assess the safety and tolerability of TVR-based treatment
4 Study arms:
PR48 - Peg-RBV (Control) 48 weeks (n=80)
T12/PR12 - TVR + Peg+RBV (n=20) 12 weeks treatment
T12/PR24 - (n=80), TVR + Peg-Ifn+RBV for 12 weeks followed by Peg/RBV 12 weeks
T12/PR48 - (n=80), TVR + Peg-IFN + RBV for 12 weeks, followed by Peg-RBV for 36 weeks
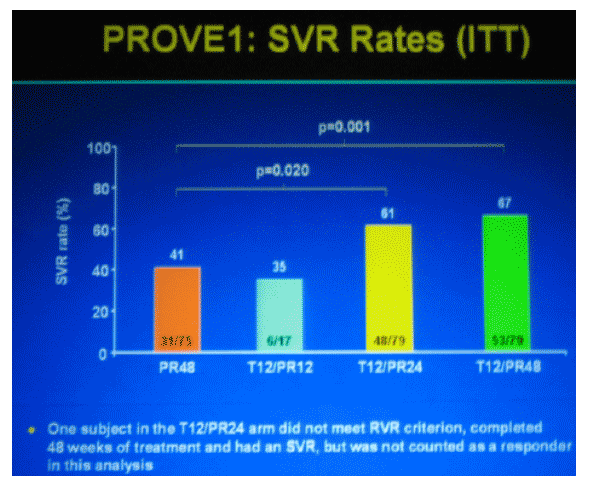
PRIMARY ENDPOINT
The prorton of subjects who achieve SVR, defined as undetectable HCV RNA (LOD 10 IU/mL, as measured by the TaqMan assay) 24 weeks after the end of dosing
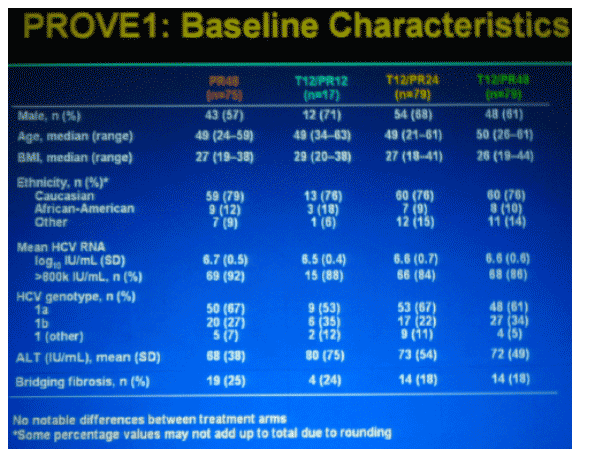
SUBJECTS
--treatment-naive
--infected with genotype 1
--subjects with cirrhosis were excluded
STATISTICAL ANALYSIS
--ITT analysis included all randomized subjects who received at least one dose of study drug


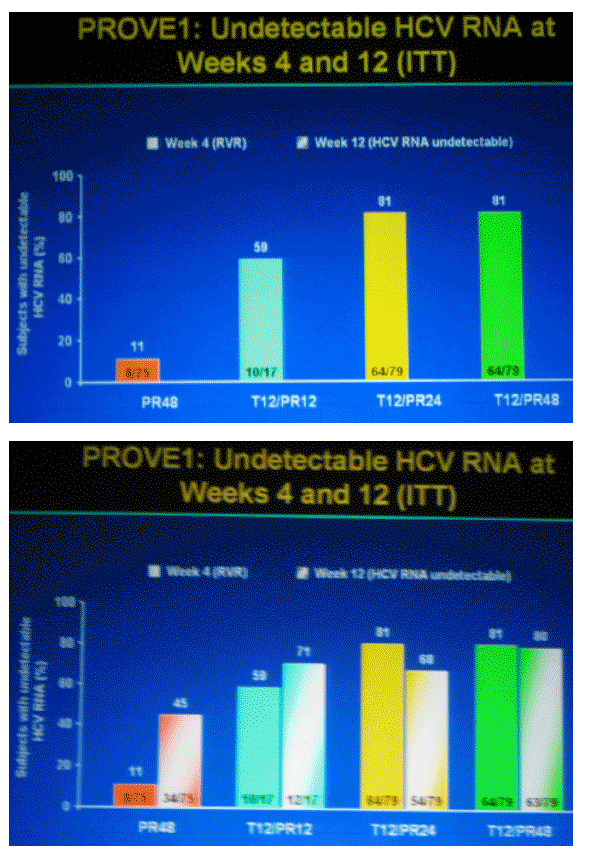
On-Treatment Virologic Failure
(During First 12 Weeks)
Low rates of virologic breakthrough in TVR-based treatment arms
--breakthrough was defined as having detectable HCV RNA after becoming undetectable, or a 1 log increase from nadir value
--most virologic breakthroughs occurred in the first 4 weeks of treatment
week 1 - week 4 was 5% (91/175)
week 1 - week12 was 7% (12/175, total)
--most breakthroughs occurred in subjects who never achieved undetectable HCV RNA
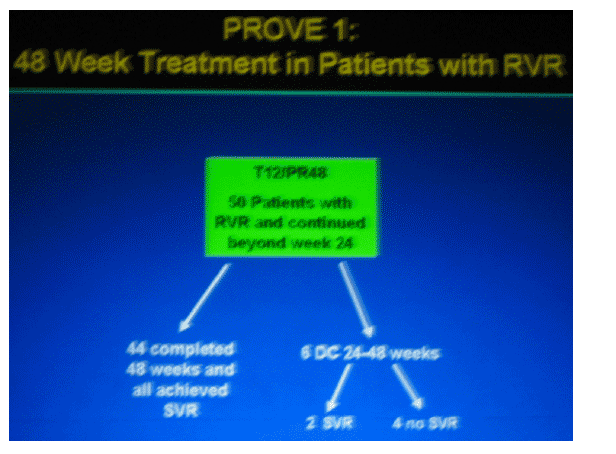
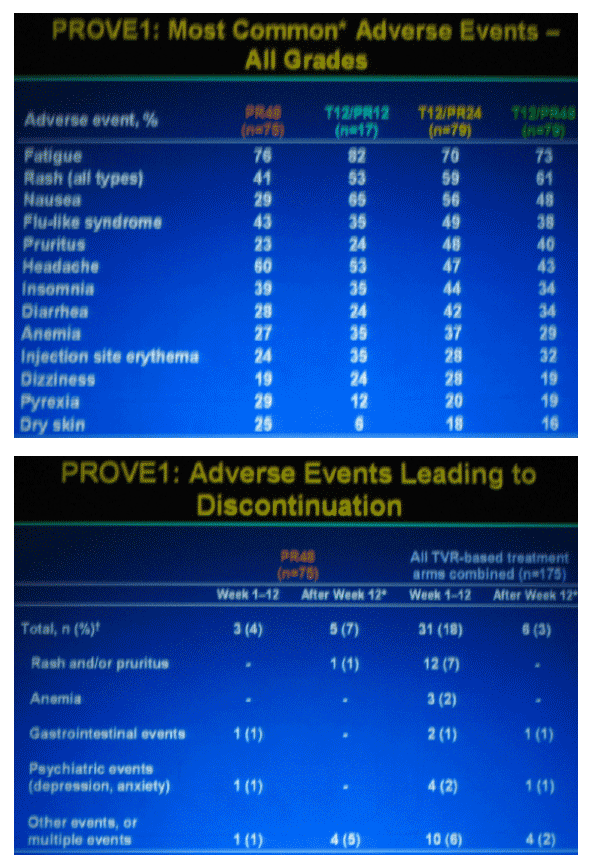
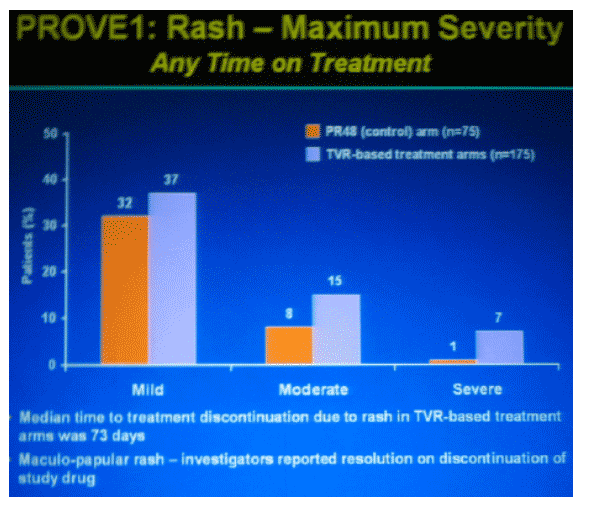
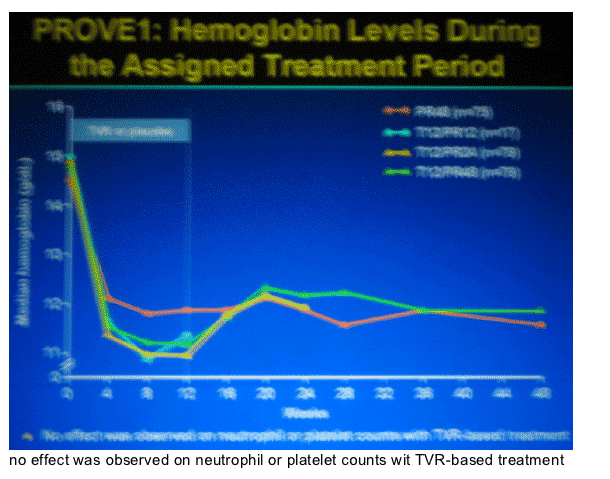
|
| |
|
 |
 |
|
|